Abstract
Purpose
The purpose of this study is to describe the prevalence, intensity and distress of five symptoms in intensive care unit (ICU) patients and to investigate possible predictive factors associated with symptom intensity.
Methods
This is a prospective cohort study of ICU patients. A symptom questionnaire (i.e., Patient Symptom Survey) was used to describe the prevalence, intensity and distress of pain, thirst, anxiousness, tiredness, and shortness of breath over seven ICU days. Associations between symptom intensity and possible predictive factors were assessed using the general estimating equation (GEE) model.
Results
Out of 603 eligible patients, 353 (Sample 2) were included in the present study. On the first ICU day, 195 patients (Sample 1) reported thirst as the most prevalent symptom (66%), with the highest mean intensity score (6.13, 95% confidence interval (CI) [5.7–6.56]). Thirst was the most prevalent (64%) and most intense (mean score 6.05, 95%CI [5.81–6.3]) symptom during seven days in the ICU. Anxiousness was the most distressful (mean score 5.24, 95%CI [4.32–6.15]) symptom on the first day and during seven days (mean score 5.46, 95%CI [4.95–5.98]). During seven days, analgesic administration and sepsis diagnosis were associated with increased thirst intensity. Older age and being mechanically ventilated were associated with decreased pain intensity, and analgesic administration was associated with increased pain intensity. Family visits and female gender were associated with increased intensity of anxiousness and shortness of breath, respectively.
Conclusions
Self-reporting ICU patients experienced a high and consistent symptom burden across seven days. Certain variables were associated with the degree of symptom intensity, but further research is required to better understand these associations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Patients able to self- report experience a high and consistent symptom burden during treatment in the intensive care unit. Routine assessments of symptoms are warranted and clinicians should initiate symptom relief measures to improve patient comfort. |
Introduction
Treatments provided to severely ill or injured patients in intensive care units (ICUs) have increased patient survival [1,2,3]. Yet, current use of less sedation makes patients more awake [4], alert, and prone to bothersome symptoms. A more focused attention to patients’ symptoms may be the first step to improving recovery after ICU treatment or, when treatments fail, to providing a more peaceful death. As symptoms, by definition, are perceived experiences, providing patients the opportunity to report their symptoms is the gold standard for symptom assessment [5].
Recent studies have described various symptoms and discomforts in a variety of ICU populations [6, 7]. However some studies have only evaluated a single symptom or symptom intensity which does not capture the complexity of symptoms; some use retrospective designs, particularly problematic when attempting to elicit patients’ real-time experiences [6,7,8,9]. Other studies had extensive exclusion criteria, reducing the potential to generalize results to the general ICU patient population [10]. Overall, studies reporting symptoms from the first days in the ICU are scarce [8, 11]. ICU admissions are stressful in nature and associated with unclear clinical situations, several procedures, and therefore potentially represent a high risk of unrelieved symptoms [12,13,14].
Over 10 years ago, Puntillo and colleagues assessed the prevalence, intensity and distress of ten symptoms in a heterogeneous group of ICU patients, laying the groundwork for future, more extensive symptom studies [11]. What is unknown is the current status of ICU patient symptom experiences and factors that can potentially predict the intensity of certain symptoms [11, 15]. Therefore, the main aim of this study was to describe several dimensions (i.e., prevalence, intensity and distress) of five symptoms recommended for daily assessment [16] (i.e., thirst, pain, anxiousness, tiredness, shortness of breath) in a heterogeneous ICU population, over seven days. The secondary aim was to investigate associations between intensity of the five symptoms and selected demographic and clinical variables which we anticipated to be associated with these symptoms over seven days.
Methods
Study design and setting
This prospective cohort study was conducted as a multicenter observational study investigating symptoms in ICU patients and long-term outcomes during the first year after ICU admission (NCT 03714230). Patients were consecutively recruited from six medical and surgical ICU departments from two hospitals in Norway between October 2018 and June 2020.
The study was performed in accordance with the ethical principles for medical research as written in the Declaration of Helsinki. Written informed consent for study participation was obtained from patients or temporarily from their family members until patients were able to consent. Patients who died before self-consent was obtained were retained in the study. Regional Committee for Medical Research Ethics approved the study (Approval number 2017/990-1), as did data protection officers at the respectively hospitals. The present study adheres to the reporting guidelines according to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) [17].
Study population
ICU patients were included if they were above 18 years of age and had one of these inclusion criteria; need for mechanical ventilation (MV), need for continuous vasoactive therapy or ICU stay greater than 24 h. All patients that met the inclusion criteria were consecutively enrolled in the study, and daily symptom assessments were started immediately using self-report when possible, or proxy-reporting by bed-side nurse. If patients did not want to participate when they were capable of self-report and give consent, all data on these patients were deleted. Only patient self-reported data were used in the present study. Patients were excluded if they had a predefined cognitive deficit as described in the medical chart (e.g., dementia), were homeless, unable to read and write Norwegian, admitted for organ preservation, or were re-admitted to the ICU within 72 h.
Outcome
The study outcome was self-reported symptom information measured on day one and during seven days using the Patient Symptom Survey (PSS) [11]. Sample 1 contains patients with only symptom assessments from day one; Sample 2 contains patients with assessments during seven days (including day 1). First day in the ICU was defined as the day of admission, or for those who were admitted late in the afternoon, the following day (first full day).
Data collection
Patient reported outcome measures (PROMs) on physical and mental health information prior to ICU (pre-ICU data) were retrieved from patients after their study inclusion in the ICU. During the ICU stay patients were asked to rate their symptoms once a day for seven days. Clinical variables were retrieved from medical records. Either patients or family members reported information about patients’ demographics.
Pre-ICU data
Frailty was measured with Clinical Frailty Scale (CFS) where patients gave a subjective judgement on their health status prior to ICU admission [18]. Frailty is described with nine classes, from very fit to terminally ill. This tool has shown to be valid and reliable and easy to use, even in young critically ill patients [19]. CFS was divided into three categories: non-frail, pre-frail and frail [20]. Charlson comorbidity index (CCI) was calculated and used to determine patients’ pre comorbidity profile [21].
Patients’ levels of anxiety and depression prior to ICU admission was measured using the Hospital Anxiety and Depression Scale (HADS) [22]. The scale consists of 14 items, with two subscales including seven questions each: HADS-Anxiety (HADS-A), and HADS Depression (HADS-D). Both are scored with a Likert scale (0–3). Total scores for each subscales range from 0 to 21. Each subscale indicates states of anxiety or depression, with a cut-off score ≥ 8 indicate states of anxiety and depression [23]. The HADS has shown good psychometric properties in patients admitted in an acute medical unit in Norway [24].
Demographic and clinical data
Demographic data were age, sex, education, co-habitation and work situation. Data on patient visitations from family members (within four hours of assessment) and medication use (i.e., opioids or sedatives, yes/no) administered within six hours before assessment were collected from medical charts. Level of alertness or agitation was classified according to the Richmond Agitation-Sedation Scale (RASS) completed during symptom assessments by the study nurse [25]. For intensity of illness we collected data on Simplified Acute Physiology Score II (SAPS II) [26] from medical records. Data on type of ICU admission (i.e., surgical or medical) and primary cause of ICU admission (e.g., respiratory, circulatory, gastroenterological) were collected from medical records in addition to data on invasive mechanical ventilation (IMV) and noninvasive mechanical ventilation (NIV).
Symptom assessment
Symptoms were assessed using the PSS symptom checklist, a ten-item list of symptoms developed from the validated Edmonton Symptom Assessment Scale and adjusted for ICU populations [11]. Puntillo and colleagues tested face validity of the 10-item checklist in a previous pilot study, and responders were able to differentiate between symptoms and ranges of intensity and distress [27]. Later, face validity was established when delirious and nondelirious patients could differentiate between the presence or absence of symptoms and symptom intensity and distress [11]. For the present study, we selected five symptoms (i.e., pain, thirst, anxiousness, tiredness and shortness of breath) from the original PSS to increase feasibility of data collection, as recommended by Chanques et.al. [16]. The modified checklist with five symptoms was pilot-tested for face validity and feasibility in Norwegian ICU patients by the research group. Patients were asked to score their symptoms in the present moment. Intensity was measured on a Likert scale from 0 to 10 (0 = no intensity, 10 = worst intensity possible) and distress from 0–10 (0 = no distress, 10 = the most distress possible). All members of the research team were trained to ensure consistency of symptom assessment and reporting. The training was repeated during the data collection, to ensure compliance with the procedure.
Statistical analyses
Categorical data are described as counts and percentages, and continuous data are presented as mean and standard deviation (SD) (normally distributed data) or median and interquartile range (IQR) (data with skewed distribution).
As the same patients were able to report for up to seven days and thus there were statistical dependent, we fitted multivariate linear regressions models using the generalized estimating equation (GEE) method. GEE allows for randomly missing data at different time points, e.g. no imputation is necessary [28].
One model was fitted for each of the selected symptoms. The models examined the associations between each of the five symptoms (i.e., thirst, pain, anxiousness, tiredness and shortness of breath) as the dependent variables and the same list of possible predictive factors (i.e., age, sex, SAPS II, CCI, Frailty score, primary cause of ICU admission, type of ICU admission, HADS-A total score, HADS-D total score, level of education, MV treatment, analgesic administration, sedative administration, visits during ICU stay). The selection of these possible predictive factors was based on empirical and clinical considerations, and determined by consensus of the research team.
Our selected covariates and outcome variables were assessed over seven days. GEE models estimate the overall impact of a covariate assessed over a given time period on the outcome averaged over the same time period. Thus, we estimated an average effect of a covariate on an average level of the outcome.
Each symptom was coded zero if a patient did not have the symptom, and any assessment above zero (1–10) was coded as the symptom intensity. Further, all symptom assessments during seven days were collapsed to one mean intensity score for each patient. In step one, univariate analyses were performed for all independent variables. In step two, variables with p-values ≤ 0.10 from the univariate analyses were included in the multivariate models, and the models were further adjusted for age and gender [29]. The results from the GEE models are presented with regression coefficients (estimated β with 95% confidence intervals (CI)). P-values less than 0.05 were considered statistically significant in the multivariate models. Statistical analyses were performed using SPSS 28 for Windows (IBM SPSS, Chicago, IL, USA).
Results
Patient characteristics
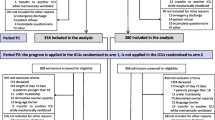
In total, 1234 patients were eligible for inclusion, and 603 met the inclusion criteria. Of these, 195 patients could self-report symptoms the first ICU day (sample 1), and 353 patents could self-report symptoms one or several times during their ICU stay (sample 2) (Fig. 1), and thereby constitutes the two samples in the present study. For the seven days assessments, median age was 63 years (IQR 48–73), 60.3% were males, and the majority of these patients had a medical reason for admission (63.5%). There were 38.8% treated with IMV and 22.4% with NIV. Of the IMV patients, 18 were orally intubated during symptom assessments, five of them providing full symptom assessments (prevalence, intensity, distress) and 13 patients were able to report only presence of symptoms (yes/no). The most frequent causes of ICU admission were respiratory (21.5%), circulatory (17.6%) and gastroenterological (16.7%) failure. Median ICU length of stay was 2.8 (IQR 1.7–5.2) days, and median SAPS II score was 38 (IQR 28–50). Sample 1 and Sample 2 were similar concerning clinical and demographic variables except for primary cause of ICU admission and time on IMV (Table 1).
Prevalence, intensity and distress of symptoms on the first day in ICU (study sample 1)
During the first day in the ICU, 195 patients were able to self-report symptoms (Table 2). Thirst was the most prevalent symptom (66%) and had the highest mean intensity score (6.13, 95%CI [5.7–6.56]). Anxiousness was the least prevalent symptom (24%), but it had the highest mean distress score (5.24, 95%CI [4.32–6.15]).
Prevalence, intensity and distress of symptoms during seven days (study sample 2)
During seven days, 353 patients self-reported symptoms 668 times (median 2 assessments per patient) (Table 2). The majority of patients were discharged before day three (median ICU lenght of stay (LOS): 2.8 days) explaining the low number of assessments per patients.
Thirst was the most prevalent (64%) and most intense (6.05, 95%CI [5.81–6.3]) symptom. Anxiousness was the symptom with the lowest prevalence (25%) during seven days but with the highest mean distress score (5.46, 95%CI [4.95–5.98]). Pain had the lowest mean intensity score (4.61, 95%CI [4.31–4.9]) and tiredness the lowest mean distress score (3.5, 95%CI [3.13–3.87]).
Associations between intensity and possible predictive factors (study sample 2)
The results from the multivariate GEE analysis of possible predictive factors for all five symptoms are listed in Tables 3a–e. The GEE model for thirst intensity revealed that both a sepsis diagnosis (B = 1.71 95%CI [0.58–2.84]) and analgesic administration (B = 0.88, 95%CI [0.18–1.59]) were associated with increased thirst intensity (Table 3a).
Increased age (B = − 0.04, 95%CI [− 0.07 to − 0.02]) was associated with decreased pain intensity. Receiving MV was associated with lower pain intensity scores (B = − 0.9, 95%CI [− 1.48 to − 0.32]), and analgesic administration during the last six hours before symptom assessment was associated with higher pain intensity scores (B = 1, 95%CI [0.49–1.5]) (Table 3b).
Having visits from family members during ICU stay was associated with more anxiousness (B = 0.64, 95%CI [0.04–1.24]) (Table 3c). Female sex was associated with increased shortness of breath (B = 0.56, 95%CI [0–1.11]) (Table 3e). None of the selected possible predictive factors were associated with the intensity of tiredness in the multivariate analyses (Table 3d).
Discussion
The main finding of the present study was that the patients reported a consistently high symptom burden for up to seven days of ICU stay. The most prevalent symptom was thirst, followed by tiredness, pain, shortness of breath and anxiousness. Thirst was the most intense symptom, while being anxious was the most distressing symptom. Indeed, the almost equal symptom intensity score of 5 for all symptoms is believed to depict at least a moderate degree of intensity [30].
Previous thirst studies have reported thirst prevalence from 70% to 76% [11, 31,32,33], versus 64–67% in the present study. We found mean intensity thirst score to be 6.13 for each of the seven days. Different scales have been used to measure intensity scores for thirst in different studies. Puntillo et al. found a mean intensity scores at 2.16 of 3 [11], while a study by Negro et al. found a mean intensity score of 5.4 of 10 for thirst [33]. It is a paradox that thirst is the most prevalent and most intense symptom in ICU patients and still not routinely assessed or treated. One study found that oral care only affected the perception of thirst for one hour, underscoring the importance of offering frequent relief of thirst [34]. Simple thirst bundles have shown to be efficient in increasing patient comfort [8, 35, 36]. Therefore, routine thirst assessment and treatment should be implemented in daily care [8, 37, 38].
Analgesic administration was associated with higher thirst intensity in this study. This finding is similar to a study by Stotts et al. that found thirst presence to be predicted by high opioid doses [15]. However, Lin et al. found that use of opioids was not a risk factor for thirst [31]. This discrepancy may be that different doses of opioids were used for pain management or that thirst was managed differently in those two studies.
A surprising, and unexplained, result was that the diagnosis of sepsis was associated with increased thirst intensity. We could not investigate the effect of patient’s fluid balance on thirst due to incomplete data. However, Stotts et al. found no statistically significant differences in fluid balance between patients who reported thirst and those who did not [15].
Anxiousness was the least prevalent symptom in the present study, but it had the highest mean distress scores. According to the theory of unpleasant symptoms (TOUS), distress is the degree to which the person is bothered by a symptom [39]. This finding reinforces the importance of investigating multiple dimensions of symptoms and appreciate psychological aspects of a patient’s symptom experience.
Surprisingly, we found that receiving visits from family members was associated with higher intensity score for anxiousness. In contrast, a recent systematic review and meta-analysis found that flexible visiting hours might reduce anxious symptoms [40]. Others found that being separated from family members were patients’ second most stressful experience [10]. We noted visits from family members in the four hours prior to data collection. Possibly patients felt more anxious after family had left, and this experience was associated with increased scores of anxiousness, or that anxious patients were those who had most visits. Another reason may be that patients have witnessed family members being worried by seeing the patients as sick. Because of this, the patient’s feeling of anxiousness increased. However, our unexplained finding about family visits should not encourage limitation of family visits, but should be further explored in future studies.
Pain was reported by 36% of the patients in the present study, and patients who received analgesics reported higher pain intensity score than patients without such medications. One study found that being given opioids prior to a procedure was a risk factor for higher procedural pain intensity [12]. Yet, a recent study of procedural pain found contrary results [41]. We had only information about analgesic administered within six hours prior to pain assessment. Therefore, it is possible that pain had increased in the time between administration of analgesics and symptom assessments, the administration could have been too close to the symptom assessment, or the pain treatments could be inadequate. We also found that patients on MV reported less pain intensity. This finding is consistent with a recent study of ICU patients in Norway [41]. In the present study, 66% of the patients on MV received analgesics (data not shown), suggesting an association between MV and pain beyond the administration of analgesics. However, this relationship needs further exploration.
Finally, we found that higher age was associated with less pain intensity. This finding is compatible with recent studies of ICU patients [14, 41]. However, two other studies showed conflicting results, where higher age was not associated with less pain intensity, for procedural pain and for critically ill patients [42, 43]. In summary, study populations and pain management practices differ, perhaps accounting for different results.
Breathlessness is frequently reported as a distressing symptom by ICU patients and was reported by one third of the patients. Further, it is associated with anxiety in MV patients [44, 45]. Female sex was the only possible predictive factor associated with shortness of breath.
Strength and limitations
The study has some limitations. The symptoms were only assessed once a day; therefore, we do not know if there were variations of symptoms throughout 24 h. Further, using the same list of possible predictive factors in the GEE models limited our investigation of possible predictive factors specific to each symptom. Other factors may be investigated that explore treatment and nursing actions such as fluid balance, fluid restrictions and mobilization and the impact of symptoms on each other. Since this was a selected cohort of patients able to self-report symptoms one or several times during seven days, the result from the present study may not be generalized to all ICU patients. Nevertheless, we collected data prospectively from a large, heterogeneous sample of adult ICU patients in several ICUs and had a longer follow up of up to seven days.
Conclusions
Our study found that self-reporting ICU patients experienced a high and consistent symptom burden across seven days. Thirst was the most prevalent and intense symptom, but the other symptoms examined were also high in intensity. Certain factors are associated with the degree of symptom intensity and distress. Further research is warranted to better understand these associations and investigate symptom relief measures.
Data availability
Data used in the analysis for the present study are available from the corresponding author upon reasonable request.
References
Mehta S, McCullagh I, Burry L (2011) Current sedation practices: lessons learned from international surveys. Anesthesiol Clin 29(4):607–624. https://doi.org/10.1016/j.anclin.2011.09.003
Lerolle N, Trinquart L, Bornstain C, Tadié JM, Imbert A, Diehl JL et al (2010) Increased intensity of treatment and decreased mortality in elderly patients in an intensive care unit over a decade. Crit Care Med 38(1):59–64. https://doi.org/10.1097/CCM.0b013e3181b088ec
Lindemark F, Haaland ØA, Kvåle R, Flaatten H, Johansson KA (2015) Age, risk, and life expectancy in norwegian intensive care: a registry-based population modelling study. PLOS ONE 10(5):e0125907. https://doi.org/10.1371/journal.pone.0125907
Devabhakthuni S, Armahizer MJ, Dasta JF, Kane-Gill SL (2012) Analgosedation: a paradigm shift in intensive care unit sedation practice. Ann Pharmacother 46(4):530–540. https://doi.org/10.1590/0034-7167.2016690418i
Barr J, Fraser GL, Puntillo K, Ely EW, Gélinas C, Dasta JF et al (2013) Clinical practice guidelines for the management of pain, agitation, and delirium in adult patients in the intensive care unit. Crit Care Med 41(1):263–306. https://doi.org/10.1097/CCM.0b013e3182783b72
Kalfon P, Mimoz O, Auquier P, Loundou A, Gauzit R, Lepape A et al (2010) Development and validation of a questionnaire for quantitative assessment of perceived discomforts in critically ill patients. Intensive Care Med 36(10):1751–1758. https://doi.org/10.1007/s00134-010-1902-9
Dessotte CA, Rodrigues HF, Furuya RK, Rossi LA, Dantas RA (2016) Stressors perceived by patients in the immediate postoperative of cardiac surgery. Rev Bras Enferm 69(4):741–750. https://doi.org/10.1590/0034-7167.2016690418i
Tsai H-Y, Chao A, Hsiao W-L (2023) The effectiveness of cold oral stimuli in quenching postoperative thirst: a systematic review and meta-analysis. Intensive Critical Care Nurs 75:103359. https://doi.org/10.1016/j.iccn.2022.103359
Granja C, Lopes A, Moreira S, Dias C, Costa-Pereira A, Carneiro A (2005) Patients’ recollections of experiences in the intensive care unit may affect their quality of life. Crit Care 9(2):R96. https://doi.org/10.1186/cc3026
Zengin N, Ören B, Üstündag H (2020) The relationship between stressors and intensive care unit experiences. Nurs Crit Care 25(2):109–116. https://doi.org/10.1111/nicc.12465
Puntillo KA, Arai S, Cohen NH, Gropper MA, Neuhaus J, Paul SM et al (2010) Symptoms experienced by intensive care unit patients at high risk of dying. Crit Care Med 38(11):2155–2160. https://doi.org/10.1097/CCM.0b013e3181f267ee
Puntillo KA, Max A, Timsit J-F, Vignoud L, Chanques G, Robleda G, et al (2014) Determinants of procedural pain intensity in the intensive care unit. The Europain® study. Am J Respir Crit Care Med 189(1):39–47. https://doi.org/10.1164/rccm.201306-1174OC
Devlin JW, Skrobik Y, Gélinas C, Needham DM, Slooter AJC, Pandharipande PP et al (2018) Clinical Practice Guidelines for the Prevention and Management of Pain, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption in Adult Patients in the ICU. Crit Care Med 46(9):e825–e873. https://doi.org/10.1097/ccm.0000000000003299
Kalfon P, Boucekine M, Estagnasie P, Geantot MA, Berric A, Simon G et al (2020) Risk factors and events in the adult intensive care unit associated with pain as self-reported at the end of the intensive care unit stay. Crit Care 24(1):685. https://doi.org/10.1186/s13054-020-03396-2
Stotts NA, Arai SR, Cooper BA, Nelson JE, Puntillo KA (2015) Predictors of thirst in intensive care unit patients. J Pain Symptom Manage 49(3):530–538. https://doi.org/10.1016/j.jpainsymman.2014.07.001
Chanques G, Nelson J, Puntillo K (2015) Five patient symptoms that you should evaluate every day. Intensive Care Med 41(7):1347–1350. https://doi.org/10.1007/s00134-015-3729-x
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP (2008) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol 61(4):344–349. https://doi.org/10.1016/j.jclinepi.2007.11.008
Rockwood K, Song X, MacKnight C, Bergman H, Hogan DB, McDowell I et al (2005) A global clinical measure of fitness and frailty in elderly people. CMAJ 173(5):489–495. https://doi.org/10.1503/cmaj.050051
Bagshaw SM, Majumdar SR, Rolfson DB, Ibrahim Q, McDermid RC, Stelfox HT (2016) A prospective multicenter cohort study of frailty in younger critically ill patients. Crit Care 20(1):175. https://doi.org/10.1186/s13054-016-1338-x
Flaatten H, De Lange DW, Morandi A, Andersen FH, Artigas A, Bertolini G et al (2017) The impact of frailty on ICU and 30-day mortality and the level of care in very elderly patients (≥ 80 years). Intensive Care Med 43(12):1820–1828. https://doi.org/10.1007/s00134-017-4940-8
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383. https://doi.org/10.1016/0021-9681(87)90171-8
Zigmond AS, Snaith RP (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand 67(6):361–370. https://doi.org/10.1111/j.1600-0447.1983.tb09716.x
Snaith RP (2003) The hospital anxiety and depression scale. Health Qual Life Outcomes 1(1):29. https://doi.org/10.1186/1477-7525-1-29
Helvik AS, Engedal K, Skancke RH, Selbæk G (2011) A psychometric evaluation of the Hospital Anxiety and Depression Scale for the medically hospitalized elderly. Nord J Psychiatry 65(5):338–344. https://doi.org/10.3109/08039488.2011.560684
Sessler CN, Grap MJ, Brophy GM (2001) Multidisciplinary management of sedation and analgesia in critical care. Semin Respir Crit Care Med 22(2):211–226. https://doi.org/10.1055/s-2001-13834
Le Gall J-R, Lemeshow S, Saulnier F (1993) A New Simplified Acute Physiology Score (SAPS II) Based on a European/North American Multicenter Study. JAMA 270(24):2957–2963. https://doi.org/10.1001/jama.1993.03510240069035
Li DT, Puntillo K (2006) A pilot study on coexisting symptoms in intensive care patients. Appl Nurs Res 19(4):216–219. https://doi.org/10.1016/j.apnr.2006.01.003
Zeger SL, Liang K-Y, Albert PS (1988) Models for longitudinal data: a generalized estimating equation approach. Biometrics 44(4):1049–1060. https://doi.org/10.2307/2531734
Bursac Z, Gauss CH, Williams DK, Hosmer DW (2008) Purposeful selection of variables in logistic regression. Source Code Biol Med 3:17. https://doi.org/10.1186/1751-0473-3-17
Serlin RC, Mendoza TR, Nakamura Y, Edwards KR, Cleeland CS (1995) When is cancer pain mild, moderate or severe? Grading pain severity by its interference with function. Pain 61(2):277–284. https://doi.org/10.1016/0304-3959(94)00178-h
Lin R, Li H, Chen L, He J (2022) Prevalence of and risk factors for thirst in the intensive care unit: an observational study. J Clin Nurs. https://doi.org/10.1111/jocn.16257
Nelson JE, Meier DE, Oei EJ, Nierman DM, Senzel RS, Manfredi PL et al (2001) Self-reported symptom experience of critically ill cancer patients receiving intensive care. Crit Care Med 29(2):277–282. https://doi.org/10.1097/00003246-200102000-00010
Negro A, Villa G, Greco M, Ciriolo E, Luraschi EL, Scaramuzzi J et al (2021) Thirst in patients admitted to intensive care units: an observational study. Ir J Med Sci. https://doi.org/10.1007/s11845-021-02817-7
Doi S, Nakanishi N, Kawahara Y, Nakayama S (2021) Impact of oral care on thirst perception and dry mouth assessments in intensive care patients: an observational study. Intensive Crit Care Nurs 66:103073. https://doi.org/10.1016/j.iccn.2021.103073
Puntillo K, Arai SR, Cooper BA, Stotts NA, Nelson JE (2014) A randomized clinical trial of an intervention to relieve thirst and dry mouth in intensive care unit patients. Intensive Care Med 40(9):1295–1302. https://doi.org/10.1007/s00134-014-3339-z
VonStein M, Buchko BL, Millen C, Lampo D, Bell T, Woods AB (2019) Effect of a scheduled nurse intervention on thirst and dry mouth in intensive care patients. Am J Crit Care 28(1):41–46. https://doi.org/10.4037/ajcc2019400
Arai S, Stotts N, Puntillo K (2013) Thirst in critically ill patients: from physiology to sensation. Am J Crit Care 22(4):328–335. https://doi.org/10.4037/ajcc2013533
Leemhuis A, Shichishima Y, Puntillo K (2019) Palliation of thirst in intensive care unit patients: translating research into practice. Crit Care Nurse 39(5):21–28. https://doi.org/10.4037/ccn2019544
Lenz ER, Pugh LC, Milligan RA, Gift A, Suppe F (1997) The middle-range theory of unpleasant symptoms: an update. Adv Nurs Sci 19(3):14–27. https://doi.org/10.1097/00012272-199503000-00003
Nassar Junior AP, Besen B, Robinson CC, Falavigna M, Teixeira C, Rosa RG (2018) Flexible versus restrictive visiting policies in ICUs: a systematic review and meta-analysis. Crit Care Med 46(7):1175–1180. https://doi.org/10.1097/ccm.0000000000003155
Olsen BF, Valeberg BT, Jacobsen M, Småstuen MC, Puntillo K, Rustøen T (2021) Pain in intensive care unit patients—a longitudinal study. Nurs Open 8(1):224–231. https://doi.org/10.1002/nop2.621
van Dijk JFM, Zaslansky R, van Boekel RLM, Cheuk-Alam JM, Baart SJ, Huygen FJPM et al (2021) Postoperative pain and age: a retrospective cohort association study. Anesthesiology 135(6):1104–1119. https://doi.org/10.1097/aln.0000000000004000
Stotts NA, Puntillo K, Stanik-Hutt J, Thompson CL, White C, Rietman Wild L (2007) Does age make a difference in procedural pain perceptions and responses in hospitalized adults? Acute Pain 9(3):125–134. https://doi.org/10.1016/j.acpain.2007.07.001
Schmidt M, Demoule A, Polito A, Porchet R, Aboab J, Siami S et al (2011) Dyspnea in mechanically ventilated critically ill patients*. Crit Care Med 39(9):2059–2065. https://doi.org/10.1097/CCM.0b013e31821e8779
Demoule A, Hajage D, Messika J, Jaber S, Diallo H, Coutrot M et al (2022) Prevalence, intensity, and clinical impact of dyspnea in critically Ill patients receiving invasive ventilation. Am J Respir Crit Care Med 205(8):917–926. https://doi.org/10.1164/rccm.202108-1857OC
Acknowledgements
We would like to thank the Norwegian Symptom Management Network for the funding of a one-week international research stay and two writing seminars for the PhD-student. The TSD (Tjeneste for Sensitive Data) facilities, owned by the University of Oslo, operated and developed by the TSD service group at the University of Oslo, IT department (USIT), were used to conduct this study.
Funding
Open access funding provided by University of Oslo (incl Oslo University Hospital). Department of Postoperative and Intensive Care Nursing and Department of Research and Development at Division of Emergencies and Critical Care, Oslo University Hospital, and South Eastern Norway Regional Health Authority funded this study.
Author information
Authors and Affiliations
Contributions
Study conception and design: TR, AL, KH, KP, SB. Material preparation and data collection: CS-L, KH. First draft of the manuscript: CS-L, KH, TR, SB, KP, MH. Formal analysis: CS-L, MH. Review and editing of the manuscript: all authors. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
All authors declare that they have no have no commercial or financial interests or conflict of interest.
Ethical approval
This study complies with ethical standards. Regional Committee for Medical Research Ethics approved the study (Approval number 2017/990-1).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Saltnes-Lillegård, C., Rustøen, T., Beitland, S. et al. Self-reported symptoms experienced by intensive care unit patients: a prospective observational multicenter study. Intensive Care Med 49, 1370–1382 (2023). https://doi.org/10.1007/s00134-023-07219-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-023-07219-0