Abstract
Objective
Minimally invasive approaches are subject to controversy in orthopedic surgery. The aim of the current study was to compare the radiographic parameters between two minimally invasive approaches in total hip arthroplasty.
Material and methods
Between January 2018 and February 2019, the radiographic parameters of 80 patients undergoing total hip arthroplasty via minimally invasive approaches (DAA: n = 40; SuperPath® SP: n = 40) have been measured. The radiographic analysis was performed with digital software tool mediCad® (HECTEC™ GmbH, Landshut, Germany).
Results
Patients treated with DAA showed significantly higher inclination (SP: 39.7° ± 7.3° vs. DAA: 44.7° ± 5.3°) and significantly lower cup anteversion values (SP: 31.2° ± 7.9° vs. DAA: 27. 5° ± 5.3°, p < 0.001) than patients undergoing THA via SP postoperatively. The horizontal femoral offset was neither preoperatively nor postoperatively higher in DAA than in SP cohort (preoperative: p = 0.71, postoperative: p = 0.25) (preoperative: SP:37.2 mm ± 7.3 vs. DAA 38.2 mm ± 7.5; postoperative: SP: 38.0 mm ± 7.2 vs. DAA: 40.5 mm ± 7.0). At both times, the acetabular offset was significantly higher in DAA cohort than in SP cohort (preoperative: SP: 32.9 mm ± 5.9 vs. DAA: 36.8 mm ± 4.9; postoperative: SP: 28.9 mm ± 4.2 vs. DAA: 33.4 mm ± 3.8) (preoperative: 0.001; postoperative: p < 0.001). The vertical height was preoperatively and postoperatively not significantly higher in SP cohort than in DAA cohort (preoperative: SP: 16.1 mm ± 4.1 vs. DAA: 15.5 mm ± 4.9; postoperative: SP: 16.6 mm ± 4.6 vs. DAA: 16.1 mm ± 4.6) (preoperative: p = 0.77; postoperative: p = 0.58). The preoperatively existing leg length discrepancy of the affected leg could be compensated via surgery without showing significant differences between the two cohorts (preoperative: SP: −3.2 mm ± 5.4 vs. DAA: 1.9 mm ± 4.9; postoperative: SP: 1.5 mm ± 5.4 vs. DAA: 4.8 mm ± 5.6) (preoperative: p = 0.34; postoperative: p = 0.09).
Conclusion
The current study demonstrates suitable cup positioning and stem alignment in the coronal plane using minimal-invasive approaches DAA and Superpath®.
Zusammenfassung
Hintergrund
Das Ziel dieser Studie war es die radiologischen Parameter von Patienten, die minimal-invasiv über einen direkten anterioren Zugang („direct anterior approach“ [DAA]) oder SuperPath® (Microport Orthopedics, Arlington, Virginia, USA) operiert wurden, zu vergleichen.
Material und Methoden
Zwischen Januar 2018 und Februar 2019 wurden prä- und postoperative radiologische Parameter von 80 Patienten, die sich einer Hüftendoprothesenimplantation über die beiden minimal-invasive Zugänge DAA (n = 40) und SP (n = 40) unterzogen, erhoben. Die radiologische Auswertung wurde mit der digitalen Software mediCad® (HECTEC™ GmbH, Landshut, Germany) vorgenommen. Die erhobenen radiologischen Parameter waren die Pfannenanteversion, die Pfanneninklination, das Drehzentrum, die vertikale Höhe, das vertikale femorale Offset, das horizontale femorale Offset, das azetabuläre Offset und die Beinlängendifferenz.
Ergebnisse
Patienten, die über einen DAA operiert wurden, zeigten signifikant höhere Pfanneninklinationswinkel (SP: 39,7° ± 7,3° vs. DAA: 44,7° ± 5,3°; p = 0,001) als Patienten, die über einen SP-Zugang operiert wurden. Die Pfannenanteversion war signifikant niedriger in der DAA-Gruppe als in der SP-Gruppe (SP: 31,2° ± 7,9° vs. DAA: 27,5° ± 5,3°; p = 0,001). Das horizontale femorale Offset war weder prä- noch postoperativ signifikant höher in der DAA-Gruppe als in der SP-Gruppe (präoperativ: SP: 37,2 mm ± 7,3 mm vs. DAA 38,2 mm ± 7,5 mm; postoperativ: SP: 38,0 mm ± 7,2 mm vs. DAA: 40,5 mm ± 7,0 mm, präoperativ: p = 0,71, postoperativ: p = 0,25). Prä- und postoperativ war das azetabuläre Offset in der DAA-Gruppe signifikant höher als in der SP-Gruppe (präoperativ: SP: 32,9 mm ± 5,9 mm vs. DAA: 36,8 mm ± 4,9 mm; postoperativ: SP: 28,9 mm ± 4,2 vs. DAA: 33,4 mm ± 3,8 mm; präoperativ: p = 0,001; postoperativ: p < 0,001). Die vertikale Höhe war prä- und postoperativ nicht signifikant höher in der SP-Gruppe als in der DAA-Gruppe (präoperativ: SP: 16,1 mm ± 4,1 mm vs. DAA: 15,5 mm ± 4,9 mm; postoperativ: SP: 16,6 mm ± 4,6 mm vs DAA: 16,1 mm ± 4,6 mm, präOP: p = 0,77; postOP: p = 0,58). Die präoperativ bestehende Beinlängendifferenz konnte durch die Operation in beiden Gruppen ausgeglichen werden, ohne jedoch signifikante Unterschiede zwischen den beiden Gruppen zu zeigen (präoperativ: SP: −3,2 mm ± 5,4 mm vs. DAA: −1,9 mm ± 4,9 mm; postoperativ: SP: 1,5 mm ± 5,4 mm vs. DAA: 4,8 mm ± 5,6 mm; präoperativ: p = 0.34; postoperativ: p = 0.09).
Schlussfolgerung
Die aktuelle Studie zeigt eindeutig, dass über die beiden untersuchten minimal-invasiven Zugänge eine akkurate Implantatpositionierung möglich ist.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Total hip arthroplasty (THA) is one of the most common procedures in orthopedic surgery [1] and combines top level patient satisfaction with low complication rates [2]; however, traditional surgical techniques are associated with undesirable side effects including extensive skin incision, tissue damage, increased perioperative blood loss and delayed postoperative rehabilitation [3]. For those reasons, minimally invasive approaches, such as the direct anterior approach (DAA), have been introduced into THA [4]. First short-term results of DAA demonstrated promising results which were attributed to intermuscular approach [5]. Yet, there are several important problems with minimal-invasive approaches which have to be concerned, such as prolonged learning curves, posterior dislocation and component malposition due to limited overview and extensive posterior soft tissue release [6,7,8,9,10].
In 2011, an innovative minimal-invasive surgical technique, SuperPath® (supercapsular percutaneously assisted total hip), was introduced into THA [11]. The method unites the percutaneously assisted total hip (PATH, Wright Medical Technology, Memphis, TN, USA) and supercapsular (SuperCap, Wright Medical Technology) methods. First studies investigating SuperPath® approach showed encouraging results with shorter length of hospitalization stay and accurate implant positioning [7, 12,13,14,15]. The complication rates following joint replacement by the SuperPath® technique were described as very low [11, 16, 17].
The purpose of the present study was to compare the postoperative radiographic results of SuperPath® and DAA as minimal-invasive approaches in THA.
Material and methods
Between January and December 2018, 132 patients received THA due to primary arthritis using a minimal-invasive approach in a maximum care endoprosthesis center. Of the patients 80 could be contacted by telephone and gave the written consent to participate. The study excluded patients with femoral neck fractures, femoral head necrosis and posttraumatic arthritis as indications for hip joint replacement. Further exclusion criteria were the implantation of short stem femoral implants and cemented femoral stem implants. The medical history was collected by file research and the radiographic evaluation was made by two experienced surgeons. The observers were blinded and were not involved in the surgery.
Surgical techniques and evaluation of intraoperative data
All surgeries were performed by two experienced hip surgeons. MH used the DAA while DB performed THA via SP. A digitalized planning tool (mediCad®, HECTEC™ GmbH, Landshut, Germany) was used for preoperative planning. All patients underwent general anesthesia and received a single-shot antibiotic prophylaxis (cefazolin 2 g) before incision.
The patients in the DAA group were positioned supine on the operating table. The hip was positioned at the table break in order to allow extension during the procedure. The proximal starting point of skin incision was determined ~2 fingerbreadths lateral and ~1 fingerbreadth distal from anterior superior iliac spine (ASIS). A skin incision of suitable length was made along the straight line connecting proximal starting point and fibula head. The interval between the tensor fascia latae and the sartorius was opened and the circumflex vessels were coagulated. The anterior joint capsule could be visualized and incised longitudinally. A slight release of the proximal lateral hip capsule at the acetabular rim allowed a visualization of the joint space. A retractor can be positioned adjacent to the anterior inferior iliac spine (AIIS) to make medial joint space visible. To protect the trochanteric region another retractor was positioned next to the lateral femoral neck. From the exposed neck, a V-shaped piece of bone was cut out and the femoral head was removed. After head removal, the acetabulum could be directly inspected at the base of exposure. Anterior osteophytes and parts of the labrum were resected. The acetabulum was milled gradually in ascending order until all remnants of the cartilage are removed and microbleeding of the bone was visible. A press fit uncemented acetabular cup was then placed. If necessary, screws were inserted into acetabular cup. After palpatory and visual examination of the cup position, a polyethylene liner was impacted. To get access to the proximal femur, the leg was positioned with extension, external rotation and adduction in the ipsilateral hip joint. A slight knee flexion facilitates the external rotation. The remaining posterior joint capsule tissue was removed so that superior aspect of the femoral neck and piriformis fossa could be inspected. The release of remaining joint capsule soft tissue adhering to the femoral neck was described as crucial by some authors for obtaining appropriate access to proximal femur [18]. The femoral shaft should be external rotated for at least 90°. The proximal femur was closely positioned lateral to the acetabular component. The femoral neck was elevated with a support hook (femoral elevator) so that the access to femoral canal could be obtained using a box cutting osteotome.
The preparation of the femoral canal was done with appropriate version and lateralization gradually in ascending order until press fit was reached. After trial reduction and careful fluoroscopy to evaluate the canal fit and offset of the femoral component, the femoral trial components was removed and the situs was irrigated carefully. If trial reduction showed no tendency of dislocation and an accurate range of motion, femoral component was implanted. After implantation of femoral component into femoral canal according to recommendation of manufacturer, the cone was cleaned from tissue material by irrigation and following drying with compresses. The final ceramic head was placed and seated. The acetabular component with its polyethylene inlay was irrigated. After reduction, position of components was assessed with final fluoroscopy and the stability of the hip was confirmed manually ([19, 20]; Fig. 1).
Photograph from a surgical site showing anatomical landmarks and incision line in DAA (direct anterior approach). (From Connolly and Kamath [21]). ASIS anterior superior iliac spine, GT greater trochanter
In the SuperPath® group, the patients were positioned in the standard lateral decubitus position with the involved leg in the “home position”, i.e., 45°–60° of flexion, 20°–30° of internal rotation, and slight adduction by elevating the foot on a padded Mayo stand according to Chow (2011) [11]. A skin incision of suitable length was done from the tip of the trochanter in line with femur axis in proximal direction. After transection of subcutis, fibers of the gluteus maximus were split in line with the skin incision. The bursa tissue has to be incised at the posterior rim of gluteus medius. The gluteus medius was retracted in anterior direction to identify the piriformis tendon. The gluteus minimus was dissected and moved anterior under use of Cobb elevator. After elevating the ipsilateral knee, sharp retractors were placed under piriformis tendon posterior and gluteus medius to visualize the joint capsule. After incision of joint capsule in line with the skin incision using an electrocautery, the labrum of the acetabulum was partially removed. At this point, hemostasis at the basis of the capsule was crucial [22]. The sharp elevators were removed and blunt Hohmann retractors were inserted around the neck, first posterior then anterior. After identifying the saddle of femoral neck as point of entry, the femoral preparation started with opening of the femoral canal using a sharp start reamer. At this point, it was important to confirm intramedullary reaming with a cortical feeler gauge. A channel was reamed at the upper margin of femoral neck from the femoral canal to the center of femoral head. Then, the femoral canal was broached in ascending order under protection of femoral head and neck. With the broach as a saw guide neck osteotomy was determined. After neck resection, a Romanelli retractor was placed inside the capsule just at the anterior and posterior wall to explore the acetabulum. After identifying the transverse ligament, the rest of the acetabular labrum was removed. To release the tension of the sciatic nerve, the leg was placed with flexed knee and extended hip. After palpation of the femur, a 1 cm incision was done 1–2 cm posterior to the femur that allowed the creation of a cannula through the gluteus maximus muscle ending posterior to the femoral neck inside the joint capsule.A bone hook was placed into the shoulder of the femoral broach to retract the femur anterior if necessary. Acetabular preparation and cup impaction were performed via a portal without needing release of the iliotibial tract or remaining external rotators using a sharp Romanelli self-retaining retractor (Innomed, Savannah, GA, USA) and a modified Zelpi selfretaining retractor (Life Instruments, Braintree, MA, USA). After cup impaction and inlay insertion trial reduction was performed under the use of a bone hook and a T-handle to gently manipulate the femur ([23]; Abb. 2).
A bone hook is placed into the shoulder of the femoral broach to retract the femur anterior if necessary. Acetabular preparation and cup impaction are performed via a portal without needing release of the iliotibial tract or remaining external rotators using a sharp Romanelli self-retaining retractor (Innomed, Savannah, GA, USA) and a modified Zelpi self-retaining retractor (Life Instruments, Braintree, MA, USA). After cup impaction and inlay insertion trial reduction is performed under the use of a bone hook and a T-handle to gently manipulate the femur [23].
All patients received press fit acetabular and femoral components. In the DAA group, the acetabular component was a cementless fixed Plasmacup DC® (Aesculap AG, Tuttlingen, Germany) which is a hemispheric cup with a microporous titanium coating (Plasmapore®). The femoral stem types used in the DAA group were Trendhip® (Aesculap AG). The bearing surfaces were ceramic heads on vitamin‑E blended highly cross-linked ultra-high molecular weight polyethylene inlays (UHMWPE-XE, Vitelene®). The head diameter ranged from 28–36 mm.
In Superpath group, all patients received Procotyl L® (Microport Orthopedics Arlington, TN, USA) cups with ultra-high molecular weight polyethylene (UHMWPE) inlays sized 28–36 mm internal diameter. Profemur L® (Microport Orthopedics) with a ceramic head was used in all patients of Superpath group as femoral stem.
Postoperative treatment and follow-up
All patients got therapeutic dose of low molecular weight heparin as thromboembolism prophylaxis for a minimum of 5 weeks. All patients received physiotherapy and were mobilized 1 day after surgery. The amount of weight bearing was dependent on individual decision of each surgeon and varied from full weight-bearing immediately after surgery to at least 20 kg weight-bearing for 6 weeks.
Radiographic measurements
We used preoperative and 7‑day postoperative digitalized radiographs with an anteroposterior and lateral view. For the templating and measurements, we used a digital image analysis system (mediCad™, HecTec GmbH, Niederviehbach, Germany). Scaling is carried out via 3‑point scale with a coin of standard size as reference level which positioned on imaging plate.
The hip center of rotation (COR) was specified as the center of a circle drawn around the edge of the femoral head and its center [24,25,26]. The horizontal femoral offset (HFO) was determined as the perpendicular distance between the COR and the proximal femoral shaft axis (FSA) [17, 18]. The vertical femoral offset (VFO) was defined as the distance between the trans-teardrop line and the medial apex of the lesser trochanter [27]. The vertical height (VH) of the COR was defined as the distance between the preoperative or postoperative COR and the inter-teardrop line [28]. Acetabular offset (AO) was measured as the perpendicular distance between the COR and line at the inner margin of ipsilateral teardrop figure [17, 19, 20]. The limb length differences were then determined by measuring the perpendicular distance (mm) between the teardrop and the apex of the lesser trochanter on either side. The Widmer and McLaren methods were used to evaluate the cup inclination and anteversion [29,30,31]. The caput-collum-diaphyseal angle (CCD angle) was determined as the angle between the proximal femoral shaft axis and the axis of the femoral neck (preoperative) and the neck of the prosthesis (postoperative).
Statistical analysis
Summary statistics of the data were expressed as mean ± SD. The Shapiro-Wilk test was used to test for normal distribution. The paired Student’s t test was used for comparison of the normal distribution of preoperative and postoperative means and the Wilcoxon signed-rank test for non-normal distribution. The comparisons with p-values < 0.05 were considered to be significant. The software SPSS 19 (IBM, Armonk, NY, USA) was used to carry out the statistical computations.
Results
Patient demographics
In the present study, postoperative radiographic images of 80 patients were evaluated. The evaluated demographics were homogeneous in both groups (Table 1). In the DAA group, gender ratio was 16:24 (male:female) and 9:31 in the in the SuperPath® group. The mean age at the time of arthroplasty was 68.6 years ± 8.7 (48–82) in the DAA group and 68.8 years ± 8.7 (51–85) in the Superpath® group (p = 0.92). The average body mass index (BMI) was measured 26.1 kg/m2 ± 2.1 (22–30) and 25.2 kg/m2 ± 1.8 (21–30) in the SuperPath® group (p = 0.06).
Radiographic evaluation
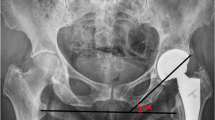
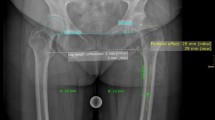
Patients being operated on via DAA showed significantly higher inclination and lower anteversion values than patients from the SP cohort (p = 0.001) (see Table 1 and Fig. 1). Patients from DAA cohort had preoperatively and postoperatively non-significantly higher horizontal femoral offset levels (preoperative: p = 0.71; post: p = 0.25). At both times, patients who were treated via DAA had significantly higher acetabular offset values (preoperative: p < 0.001; postoerative: p < 0.001). The values for vertical height were at both times non-significantly higher in SP cohort than in DAA cohort (preoperative: p = 0.77; postoerative: p = 0.58). The leg length reduction of the affected limb converted to increased leg length postoperatively without showing significant differences between the two cohorts (preoperative: p = 0.34; postoerative: p = 0.09). There was no difference in CCD pre- and postoperatively between the two cohorts (preoerative: p = 0.55; postoerative: p = 0.65). Fig. 3 shows scatterplot of inclination and anteversion in DAA (direct anterior approach) and SP (SuperPath®). Fig. 4 illustrates COR Reconstruction pre- and postoperative over with both techniques. Figs. 5, 6 and 7 show postoperative radiographs from patients who underwent MIS (minimal-invasive surgery) THA via DAA (direct anterior approach) or SuperPath®.
Discussion
The application of highly specialized minimally invasive surgical approaches in THA has increased over the last years [32, 33]. Especially, the direct anterior approach (DAA) has gained in popularity [34]. A meta-analysis by Higgins et al. (2015) about the clinical and surgical results of THA via DAA revealed better results regarding postoperative pain, functional outcomes, length of hospitalization, hip stability and postoperative narcotic consumption in comparison with traditional posterior approaches [35, 36]. Similarly, the PRISMA meta-analysis comparing DAA with lateral approach showed superior results of DAA with regard to early postoperative functional rehabilitation, lower levels of perceived pain, and shorter hospitalization time [37]; however, some serious complications such as higher rates of posterior dislocation due to implant malposition and posterior soft tissue release were reported in association with DAA [34,35,36].
Recently, SuperPath® was introduced as an alternative minimal-invasive approach in THA [11]. The first reports about clinical and radiographic results of SuperPath® were highly promising with accurate implant positioning and low complication rates [12,13,14,15,16,17,18].
The purpose of the current study was to reveal the differences in postoperative radiographic outcome of two minimal-invasive surgical approaches in THA (DAA vs. SuperPath®).
Hip dislocation is a frequent complication after THA. It is associated with a multitude of reasons [38, 39]. Most hip dislocations occur in the immediate postoperative period (30–60%) [40,41,42]. Acetabular cup positioning seems to be crucial for hip joint stability and function after THA. Inclination and anteversion angles are reliable parameters to assess postoperative cup positioning in THA [43]. The “safe zone” first described by Lewinnek et al. (1978), indicates the critical values for inclination (30–50°) and anteversion (5–25°) [44]; however, the “death” of the zone was recently proclaimed by Tezuka et al. (2019) with the hypothesis that patient’s individual functional stability is more important to prevent dislocation than rigid limiting values [45]. Rasuli et al. (2015) reported in an article about the radiographic outcomes of the PATH and SuperPath® approaches that cups were significantly more anteverted in the SuperPath® cohort without increased dislocations rates [46]. In the current study, in SP cohort there were significantly higher anteversion and significantly lower inclination angles in comparison with DAA (see Table 1 and Fig. 1). The impact of these is still not fully clear. We know that higher inclination leads to higher edge loading and wear rates and that a higher anteversion leads to higher dislocation rates to the front and lower anteversion to the back [47]. In our data, the inclination and anteversion differ between the two techniques but is in mean within the safe zones from the literature and there were no cases of dislocation. A possible explanation for the significant differences in the anteversion and inclination values could be the different position of the patients on the operating table. In DAA the patients are place in supine position and in SP in standard lateral decubitus position.
In several studies it was demonstrated that dislocation is associated with superolateral or inferolateral position of COR at distances > 5 mm away in comparison with the native COR [8, 48, 49]. Our data demonstrate that the COR was equally reconstructed with both techniques (Fig. 2). The significant difference in the acetabular offset (AO) is explained with the also existing preoperative significant difference (Table 1). With this in mind it would be of concern if the preoperative difference could not be found postoperatively. All other parameters regarding the COR were not statistically significant.
Leg length discrepancy (LLD) is a common cause for patient dissatisfaction after THA [35] and frequently causes gait disorders, nerve injury, lower back pain and hip instability. The current data situation concerning LLD is not clear. There are reports on LLD up to 10 mm without symptoms while others claimed that even small discrepancies could produce dissatisfaction [9, 35]. One of the first reports about radiographic outcome of SuperPath® in 150 patients by Quitmann (2019) did not reveal LLD above 5 mm in any patient [20]. There are only a few data on LLD in DAA available. Lv et al. (2017) and Lee et al. (2017) reported on LLD in patients treated with DAA not exceeding 5 mm [10, 50]. In the current study, patients treated via DAA had an LLD from 4.8 ± 5.6 mm and patients treated via SuperPath® 1.5 ± 5.4 mm. Accordingly there was no statistical difference between both treatments regarding the LLD (p = 0.09). In total, our results are in accordance with the current literature demonstrating good reconstruction of LLD with DAA and SuperPath®.
Stem alignment is usually placed neutrally and parallel to the femoral shaft axis. In contrast, intraoperative stem malpositioning in the coronal plane may affect offset or leg length restoration and can impair optimum load transfer between the implant and the natural bone [51]. Especially, varus stem alignment can lead to femoral cortical hypertrophy and thigh pain [52]. Low centrum-collum-diaphysis(CCD)-angles, a long neck and a trochanter overhang enhance the risk for intraoperative varus stem positioning [53]. In our study, we used the Trendhip® (Aesculap AG, Tuttlingen, Germany) and the Profemur L® (Microport Orthopedics Arlington, TN, USA) stems. The Trendhip® stem which was used in DAA has a CCD angle of 134°. The Profemur L® stem being used SP has a CCD angle of 135°. Neither preoperatively nor postoperatively could we find significant differences between the two cohorts. The slightly higher CCD angles in SP cohorts might results from the implant design with 1° higher CCD angle of the Profemur® L stem.
One of the major problems of DAA is the limited overview due to small skin incision length. To get an accurate access to the femoral canal, it is often necessary to elevate the femur with previous slight release of posterior joint capsule and muscle tendons from the femoral bone [54]. As a result, the risk of posterior dislocation might be increased, if damage of posterior structures is too large [55, 56]. Especially, the short external rotators have to be preserved to provide dorsal hip stability [57, 58]. For this reason, capsule and posterior muscle tissue-preserving techniques were devised. In these techniques, the posterior joint capsule is cut in a proximal-distal direction at the midpoint of the attachment site on the femur and the attachment site on the posterior acetabular roof [31, 50, 59,60,61,62].
We acknowledge that the current study has several limitations. Firstly, this is a retrospective control cohort study. The bias in patient selection, lack of randomization, and difference in operative techniques between surgeons might exist in this study. Secondly, the use of plain radiographs rather than CT scans to measure cup positioning may have led to slight variations based on patient pelvic tilt and rotation.
A major strength of our study is that we report for the first time on a comparison between the recently introduced MIS hip approach (SuperPath®) and the established MIS-DAA approach. Furthermore, surgery was performed by only two experienced hip surgeons from a level one endoprosthetic center. Both surgeons are familiar with minimal-invasive approaches in THA for years. The impact of potential differences due to age, gender and BMI has been considered. Only patients with osteoarthritis were included as other diagnoses such as rheumatoid arthritis and femoral neck fractures were excluded.
Conclusion
Regardless of the used minimal-invasive technique (DAA/SuperPath®) it is possible to achieve suitable cup positioning and stem alignment in the coronal plane. The restoration of center of rotation was feasible with both minimal-invasive approaches. Neither DAA nor SP provoked leg length discrepancy. The impact of this observation on the durability of the prostheses needs to be investigated in the future by long term follow-up studies.
Abbreviations
- AIIS:
-
Anterior inferior iliac spine
- AO:
-
Acetabular offset
- ASIS:
-
Anterior superior iliac spine
- BMI:
-
Body mass index
- CCD:
-
centrum-collum-diaphyseal
- COR:
-
Center of rotation
- CT:
-
Computer tomography
- DAA:
-
direct anterior approach
- HFO:
-
Horizontal femoral offset
- LLD:
-
Leg length discrepancy
- MIS:
-
Minimal invasive surgery
- ROM:
-
Range of motion
- SD:
-
Standard deviation
- SP:
-
SuperPATH®
- THA:
-
Total hip arthroplasty
- VFO:
-
Vertical femoral offset
- VH:
-
Vertical height
References
Xie J, Zhang H, Wang L, Yao X, Pan Z, Jiang Q (2017) Comparison of supercapsular percutaneously assisted approach total hip versus conventional posterior approach for total hip arthroplasty: a prospective, randomized controlled trial. J Orthop Surg Res 12(1):138. https://doi.org/10.1186/s13018-017-0636-6
Learmonth ID, Young C, Rorabeck C (2007) The operation of the century: total hip replacement. Lancet 370(9597):1508–1519
Schofield SF (2004) Minimally invasive surgery for hip replacement. ANZ J Surg 74:287–288. https://doi.org/10.1111/j.1445-2197.2004.02971.x
Hoell S, Sander M, Gosheger G, Ahrens H, Dieckmann R, Hauschild G (2014) The minimal invasive direct anterior approach in combination with large heads in total hip arthroplasty—is dislocation still a major issue? a case control study. BMC Musculoskelet Disord 15:80. https://doi.org/10.1186/1471-2474-15-80
Sariali E, Leonard P, Mamoudy P (2008) Dislocation after total hip arthroplasty using Hueter anterior approach. J Arthroplasty 23(2):266–272
Lilikakis AK, Villar RN (2005) The influence of incision length on immediate postoperative rehabilitation after total hip replacement. Hip Int 15(2):102–107
Yang C, Zhu Q, Han Y, Zhu J, Wang H, Cong R, Zhang D (2010) Minimally-invasive total hip arthroplasty will improve early postoperative outcomes: a prospective, randomized, controlled trial. Ir J Med Sci 179(2):285–290
Dearborn JT, Harris WH (2000) Acetabular revision arthroplasty using so-called jumbo cementless components: an average 7‑year follow-up study. J Arthroplasty 15(1):8–15
Keršič M, Dolinar D, Antolič V, Mavčič B (2014) The impact of leg length discrepancy on clinical outcome of total hip arthroplasty: comparison of four measurement methods. J Arthroplasty 29(1):137–141
Lv M, Zhang JQ, Wang XS, Huang Y, Li W, Zhang CY (2017) Surgical technique and early clinical outcomes of direct anterior approach to total hip arthroplasty. Beijing Da Xue Xue Bao Yi Xue Ban 49(2):206–213
Chow JP (2011) Modified micro-superior percutaneously-assisted total hip: early experiences & case reports. Curr Rev Musculoskelet Med 4:146
Cardenas-Nylander C et al (2016) Innovative approach in total hip arthroplasty: supercapsular percutaneously-assisted. Hip Int 1:34–37
Soderquist MC et al (2017) Acetabular placement accuracy with the direct anterior approach freehand technique. J Arthroplasty 32(9):2748–2754
Zhao HY et al (2017) Comparison of early functional recovery after total hip arthroplasty using a direct anterior or posterolateral approach: a randomized controlled trial. J Arthroplasty 32(11):3421–3428
Gofton W et al (2015) Thirty-day readmission rate and discharge status following total hip arthroplasty using the supercapsular percutaneously-assisted total hip surgical technique. Int Orthop 39(5):847–851
Bierbaum BE, Callaghan JJ, Galante JO, Rubash HE, Tooms RE, Welch RB (1999) An analysis of blood management in patients having a total hip or knee arthroplasty. J Bone Joint Surg Am 81(1):2–10
Liu B, Ma W, Li H, Wu T, Huo J, Han Y (2019) Incidence, classification, and risk factors for Intraoperative periprosthetic femoral fractures in patients undergoing total hip arthroplasty with a collum femoris preserving stem: a retrospective study. J Arthroplasty. https://doi.org/10.1016/j.arth.2019.03.031
Kreuzer S, Leffers K, Kumar S (2011) Direct anterior approach for hip resurfacing: surgical technique and complications. Clin Orthop Relat Res 469(6):1574–1581. https://doi.org/10.1007/s11999-010-1698-5
Haversath M, Lichetzki M, Serong S, Busch A, Landgraeber S, Jäger M, Tassemeier T (2021) The direct anterior approach provokes varus stem alignment when using a collarless straight tapered stem. Arch Orthop Trauma Surg 141(6):891–897. https://doi.org/10.1007/s00402-020-03457-9
Lovell TP (2008) Single-incision direct anterior approach for total hip arthroplasty using a standard operating table. J Arthroplasty 23(7):64–68. https://doi.org/10.1016/j.arth.2008.06.027
Connolly KP, Kamath AF (2016) Direct anterior total hip arthroplasty: literature review of variations in surgical technique. World J Orthop 7(1):38–43. https://doi.org/10.5312/wjo.v7.i1.38
Quitmann H (2019) Supercapsular percutaneously assisted (SuperPath) approach in total hip arthroplasty : surgical technique and preliminary results. Oper Orthop Traumatol. https://doi.org/10.1007/s00064-019-0597-5
Chow J (2017) SuperPath: the direct superior portal-assisted total hip approach. JBJS Essent Surg Tech 7(3):e23. https://doi.org/10.2106/JBJS.ST.16.00061
Innmann MM, Streit MR, Kolb J, Heiland J, Parsch D, Aldinger PR, Königshausen M, Gotterbarm T, Merle C (2015) Influence of surgical approach on component positioning in primary total hip arthroplasty. BMC Musculoskelet Disord 16:180. https://doi.org/10.1186/s12891-015-0623-1
Merle C, Waldstein W, Pegg E, Streit MR, Gotterbarm T, Aldinger PR, Murray DW, Gill HS (2012) Femoral offset is underestimated on anteroposterior radiographs of the pelvis but accurately assessed on anteroposterior radiographs of the hip. J Bone Joint Surg Br 94(4):477–482
Dastane M, Dorr LD, Tarwala R, Wan Z (2011) Hip offset in total hip arthroplasty: quantitative measurement with navigation. Clin Orthop Relat Res 469(2):429–436
Dolhain P, Tsigaras H, Bourne RB, Rorabeck CH, MacDonald S, Mc Calden R (2002) The effectiveness of dual offset stems in restoring offset during total hip replacement. Acta Orthop Belg 68:490–499
Kim SC, Lim YW, Kwon SY, Jo WL, Ju SH, Park CJ, Lee CW, Kim YS (2017) Level of surgical experience is associated with change in hip center of rotation following cementless total hip arthroplasty: a radiographic assessment. PLoS ONE 12(5):e178300. https://doi.org/10.1371/journal.pone.0178300
Widmer KH (2004) A simplified method to determine acetabular cup anteversion from plain radiographs. J Arthroplasty 19:387–390
McLaren RH (1973) Prosthetic hip angulation. Radiology 107:705–706
Kim SC, Lim YW, Kwon SY, Lee JK, Park IK, Kim YS (2016) Comparative analysis of radiographic hip joint geometry using measurement tools on picture archiving and communication system: a prospective study of 100 pelvic radiographs of Koreans. J Arthroplasty 31(11):2597–2602
Alecci V, Valente M, Crucil M, Minerva M, Pellegrino CM, Sabbadini DD (2011) Comparison of primary total hip replacements performed with a direct anterior approach versus the standard lateral approach: perioperative findings. J Orthop Traumatol 12(3):123–129
Hürlimann M, Schiapparelli FF, Rotigliano N, Testa E, Amsler F, Hirschmann MT (2017) Influence of surgical approach on heterotopic ossification after total hip arthroplasty—is minimal invasive better? A case control study. BMC Musculoskelet Disord 18(1):27. https://doi.org/10.1186/s12891-017-1391-x
Graves SC, Dropkin BM, Keeney BJ, Lurie JD, Tomek IM (2016) Does surgical approach affect patient-reported function after primary THA? Clin Orthop Relat Res 474(4):971–981. https://doi.org/10.1007/s11999-015-4639-5
Higgins BT, Barlow DR, Heagerty NE, Lin TJ (2015) Anterior vs. posterior approach for total hip arthroplasty, a systematic review and meta-analysis. J Arthroplasty 30(3):419–434
Debi R, Slamowicz E, Cohen O, Elbaz A, Lubovsky O, Lakstein D, Tan Z, Atoun E (2018) Acetabular cup orientation and postoperative leg length discrepancy in patients undergoing elective total hip arthroplasty via a direct anterior and anterolateral approaches. BMC Musculoskelet Disord 19(1):188. https://doi.org/10.1186/s12891-018-2097-4
Yue C, Kang P, Pei F (2015) Comparison of direct anterior and lateral approaches in total hip arthroplasty: a systematic review and meta-analysis (PRISMA). Medicine 94(50):e2126
Widmer K‑H (2006) Is there really a “safe zone” for the placement of total hip components? Bioceramics and Alternative Bearings in Joint Arthroplasty, pp 249–252
Soong M, Rubash HE, Macaulay W (2004) Dislocation after total hip arthroplasty. J Am Acad Orthop Surg 12(5):314–321
Karachalios T, Komnos G, Koutalos A (2018) Total hip arthroplasty: survival and modes of failure. EFORT Open Rev 3(5):232–239. https://doi.org/10.1302/2058-5241.3.170068
Bozic KJ, Kurtz SM, Lau E, Ong K, Vail TP, Berry DJ (2009) The epidemiology of revision total hip arthroplasty in the United States. J Bone Joint Surg Am 91(1):128–133
Ullmark G (2016) The unstable total hip arthroplasty. EFORT Open Rev 1(4):83–88
Fabeck L, Farrokh D, Tolley M, Descamps PY, Gebhart M, Delincé P (1999) A method to measure acetabular cup anteversion after total hip replacement. Acta Orthop Belg 65(4):485–491
Lewinnek GE, Lewis JL, Tarr R, Compere CL, Zimmerman JR (1978) Dislocations after total hip-replacement arthroplasties. J Bone Joint Surg Am 60:217–220
Tezuka T, Heckmann ND, Bodner RJ, Dorr LD (2019) Functional safe zone is superior to the Lewinnek safe zone for total hip arthroplasty: why the Lewinnek safe zone is not always predictive of stability. J Arthroplasty 34(1):3–8. https://doi.org/10.1016/j.arth.2018.10.034
Rasuli KJ, Gofton W (2015) Percutaneously assisted total hip (PATH) and supercapsular percutaneously assisted total hip (SuperPATH) arthroplasty: learning curves and early outcomes. Ann Transl Med 3(13):179. https://doi.org/10.3978/j.issn.2305-5839.2015.08.02
O’Dwyer Lancaster-Jones O, Williams S, Jennings LM, Thompson J, Isaac GH, Fisher J, Al-Hajjar M (2018) An in vitro simulation model to assess the severity of edge loading and wear, due to variations in component positioning in hip joint replacements. J Biomed Mater Res Part B Appl Biomater 106(5):1897–1906
Asayama I, Chamnongkich S, Simpson KJ, Kinsey TL, Mahoney OM (2005) Reconstructed hip joint position and abductor muscle strength after total hip arthroplasty. J Arthroplasty 20(4):414–420
Khlopas A, Chughtai M, Elmallah RK, Hip-Flores D, Malkani AL, Harwin SF, Mont MA, Ries MD (2018) Novel acetabular cup for revision THA improves hip center of rotation: a radiographic evaluation. Clin Orthop Relat Res 476(2):315–322. https://doi.org/10.1007/s11999.0000000000000034
Lee GC, Marconi D (2015) Complications following direct anterior hip procedures: costs to both patients and surgeons. J Arthroplasty 30(9):98–101. https://doi.org/10.1016/j.arth.2015.03.043
Haversath M, Busch A, Jäger M, Tassemeier T, Brandenburger D, Serong S (2019) The ‘critical trochanter angle’: a predictor for stem alignment in total hip arthroplasty. J Orthop Surg Res 14(1):165. https://doi.org/10.1186/s13018-019-1206-x
Evola FR, Evola G, Graceffa A, Sessa A, Pavone V, Costarella L et al (2014) Performance of the CLS Spotorno uncemented stem in the third decade after implantation. Bone Joint J 96(4):455–461. https://doi.org/10.1302/0301-620X.96B4.32607
Murphy CG, Bonnin MP, Desbiolles AH, Carrillon Y, Aїt Selmi ST (2016) Varus will have varus; a radiological study to assess and predict varus stem placement in uncemented femoral stems. Hip Int 26(6):554–560. https://doi.org/10.5301/hipint.5000412
Kanda A, Kaneko K, Obayashi O, Mogami A, Morohashi I (2018) Preservation of the articular capsule and short lateral rotator in direct anterior approach to total hip arthroplasty. Eur J Orthop Surg Traumatol 28(6):1111–1116. https://doi.org/10.1007/s00590-018-2166-2
Meneghini RM, Pagnano MW, Trousdale RT, Hozack WJ (2006) Muscle damage during MIS total hip arthroplasty: Smith-Petersen versus posterior approach. Clin Orthop Relat Res 453:293–298
Barton C, Kim PR (2009) Complications of the direct anterior approach for total hip arthroplasty. Orthop Clin North Am 40(3):371–375
Ito Y, Matsushita I, Watanabe H, Kimura T (2012) Anatomic mapping of short external rotators shows the limit of their preservation during total hip arthroplasty. Clin Orthop Relat Res 470(6):1690–1695
De Geest T, Fennema P, Lenaerts G, De Loore G (2015) Adverse effects associated with the direct anterior approach for total hip arthroplasty: a Bayesian meta-analysis. Arch Orthop Trauma Surg 135(8):1183–1192
Tsai SJ, Wang CT, Jiang CC (2008) The effect of posterior capsule repair upon post-operative hip dislocation following primary total hip arthroplasty. BMC Musculoskelet Disord 9:29. https://doi.org/10.1186/1471-2474-9-29
Prietzel T, Hammer N, Schleifenbaum S, Adler D (2014) The impact of capsular repair on the dislocation rate after primary total hip arthroplasty: a retrospective analysis of 1972 cases. Z Orthop Unfall 152(2):130–143. https://doi.org/10.1055/s-0034-1368209
White RE Jr, Forness TJ, Allman JK, Junick DW (2001) Effect of posterior capsular repair on early dislocation in primary total hip replacement. Clin Orthop Relat Res 393:163–167. https://doi.org/10.1097/00003086-200112000-00019
Kwon MS, Kuskowski M, Mulhall KJ (2006) Does surgical approach affect total hip arthroplasty dislocation rates? Clin Orthop Relat Res 447:34–38. https://doi.org/10.1097/01.blo.0000218746.84494.df
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. Busch, A. Wegner, D. Wassenaar, D. Brandenburger, M. Haversath, and M. Jäger declare that they have no competing interests.
Ethical standards.
For this article no studies with human participants or animals were performed by any of the authors. All studies mentioned were in accordance with the ethical standards indicated in each case. This retrospective study was performed after consultation with the institutional ethics committee and in accordance with national legal requirements.
Additional information
The authors André Busch and Alexander Wegner contributed equally to the manuscript.

Scan QR code & read article online
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Busch, A., Wegner, A., Wassenaar, D. et al. SuperPath® vs. direct anterior approach. Orthopädie 51, 986–995 (2022). https://doi.org/10.1007/s00132-022-04310-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00132-022-04310-0