Abstract
Beeswax is a naturally occurring product that worker bees produce. Beeswax is used in a variety of industries and pharmaceuticals. Humans utilize it extensively in cosmetics, medicinal formulations, and food manufacturing. Beeswax is an essential component of advanced contemporary beekeeping. Beekeepers, in particular, utilize significant amounts of beeswax to make beeswax comb foundation. In its natural condition, beeswax is white, but it becomes yellow then dark in color when it comes into touch with honey and pollen. The ongoing use of wax comb in bee activities (such as brood rearing, storage honey and bee bread), combined with environmental factors such as heavy metal and pesticide residues, resulted in a black color. Because of heavy metals can accumulate in wax for decades, beeswax can be a helpful tool for gathering data on hazardous contaminants in the environment. Because of their lipid-based chemical composition, beeswax combs act as a sink for numerous ambient pollutants as well as poisons when in the hive. The current study aims to measure nine heavy metals and important elements, including iron (Fe), chromium (Cr), zinc (Zn), copper (Cu), nickel (Ni), manganese (Mn), lead (Pb), cadmium (Cd), and cobalt (Co) in beeswax collected in the Behaira governorate region of Egypt between 2018 and 2022. Sample collection was conducted each year in triplicate. The samples were analyzed using an atomic absorption spectrophotometer. The quantity of metals in beeswax at different ages differed significantly. Depending on the wax age, Fe has the highest concentration in the range of 2.068 to 5.041 ppm, while Cd has the lowest ratio at 0.024 to 0.054 ppm from the first to fifth years old of comb age. The findings showed that as beeswax combs aged, the concentration of heavy metals rose. According to the study, it should gradually recycle beeswax combs each year and also adding new foundations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A naturally occurring honeybee product, beeswax is a highly complex mixture composed primarily of higher fatty acid esters, alcohols, hydrocarbons, proteins, and other minor components (Fratini et al. 2016). In addition to being utilized in the food, chemical, cosmetic, and pharmaceutical industries, beeswax is principally used in beekeeping to manufacture comb foundations (Al-Waili 2005; Amin et al. 2017; Svečnjak et al. 2019; Chaireh et al. 2020). It is fundamental for the honeybee because it is used to build the comb cells that store honey and bee bread. Beekeeping practices and quality depend largely on effective comb management, and beeswax manufacturing techniques have a significant impact on the sensory qualities of the wax (Smith et al. 2017).
Beeswax may be used to detect the presence of metals and pesticides in soil, water, and plants as a bio-indicator of pollution. Due to their unique lipid-based chemical composition, beeswax combs act as reservoir for a variety of environmental pollutants and toxins (Ngat et al. 2020; Ćirić et al. 2021).
Heavy metals can have a variety of hazardous consequences for consumers and bees and depend on how polluted the air, water, and soil (Kast and Kilchenmann 2022; Ullah et al. 2022). Long-term exposure to high levels of heavy metals may cause kidney damage, high lead levels in the blood (Zhang et al. 2012), carcinogenic risks for adults and children (Mao et al. 2019), gastrointestinal problems (Wang et al. 2020), as well as potential implications in impaired fertility (Wang et al. 2020). Heavy metals affect bee development, brood rearing, adult bee lifespan, pollen, nectar, and honey storage, and consumption behaviors (Bromenshenk et al. 1991; Hesketh et al. 2016; Burden et al. 2019).
Because heavy metals can accumulate over a long period of time, beekeepers regularly remove old, dark, and damaged combs during the active beekeeping season and transport them to the craft unit for processing into fresh wax foundations (Svečnjak et al. 2019).When compared to fresh combs, the quality and age of used combs can dramatically improve honeybees workers brood areas, worker population, worker life span, weights of freshly emerged workers and drones, and honey yield (Abd Al-Fattah et al. 2021). Heavy metals are transported to the hive as a result of bee physiological activity or human activity during routine beekeeping procedures (Borsuk et al. 2021). Heavy metal pollution of honeybees, honey, and pollen is extensively established. However, beeswax is rarely studied, most likely because it is not ingested, so it is typically out of the purview of scientific research. Heavy metal concentrations are not regularly tested in beeswax foundation manufacturing. Atomic absorption spectroscopy is extensively method used in assessing heavy metal levels. As a result, it is utilized to determine heavy metals in honeybees and their products (Pohl 2009; Gajger et al. 2016; Adugna et al. 2020).
As part of ongoing research on honeybees and bee products, nine heavy metal elements including the most dangerous ones were detected in the wax combs’ that were collected from locations in the Itay El Barud district of Egypt’s El-Behaira governorate using a validated atomic absorption spectrophotometer method and compared with results of beeswax sample analyses published in the literature (El-Wahed et al. 2021; Khalifa et al. 2021; El-Seedi et al. 2022a, b, c; Salama et al. 2022).
Materials and Methods
Samples Collection
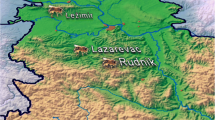
The honeybee wax samples were collected from the private apiary in Itay El Baroud region in El-Behaira governorate (Fig. 1, https://mapcarta.com/W312269819/Directions, accessed on November 13, 2022). The apiary is located behind a small road that carries agricultural tractors, cars, and tactics. The honey beeswax samples were collected from five chosen experimental years’ colonies from 2018 to 2022. In addition, from each colony, three honeybee wax frames were chosen, and with the knife, five wax pieces (5 × 5 cm) were randomly taken from each honeybee wax frame, and then saved in a plastic white bag in -5 Co freezer until analysis.
Samples Analysis for Heavy Metals
Samples were analyzed and determined by an atomic absorption spectrophotometer (Analyst, 400, Perkin Elmer, USA) according to AOAC guidelines (1995) in laboratories of in National Institute of Oceanography and Fisheries (NIOF) as describe herein (Lee 1995). The wax samples were digested then 1 g of each sample was taken and 5 ml of highly purified nitrogenous acid and 2 ml of oxygenated water were added to it, then they were placed in glass test tubes in a water bath at a temperature of 100 °C for about 18 h, until the digestion process was completed. The samples were left for the second day to cool completely, then transferred to volumetric flasks and completed the volume up to 50 ml using deionized water, then the samples were filtered using 0.4 μm filter paper. The samples were kept in polyethylene containers at (-4 °C) until the analysis process. The concentrations of iron (Fe), chromium (Cr), zinc (Zn), copper (Cu), nickel (Ni), manganese (Mn), lead (Pb), cadmium (Cd), and cobalt (Co) in ppm were measured in 15 samples wax using atomic absorption device at different ages of beeswax (Bosancic et al. 2020; Ullah et al. 2022). Limit of detection (LOD) values for the metals were Fe (0.08 mg/l), Cr (0.08 mg/l), Zn (0.01 mg/L), Cu (0.04 mg/l), Ni (0.08 mg/l), Mn (0.025 mg/l), Pb (0.2 mg/l), Cd (0.012 mg/l), and Co (0.06 mg/l).
Statistical Analysis
The statistical analysis of variance was carried out using the SPSS 26 program by following the one way - ANOVA method, and it was revealed that there were significant differences in the mean concentrations of the studied elements in the beeswax samples, and the statistical significance was estimated at a 95% confidence level.
Results
Elements Concentration in Beeswax
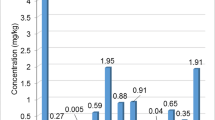
The mean concentrations of heavy metals were determined via analysis of wax samples, as listed in Table 1. A statistically significant difference was reported among the samples regarding the levels of heavy metals The mean concentrations of nine elements (Cr, Pb, Cu, Ni, Fe, Zn, Mn, Co, and Cd) were determined using an atomic absorption spectrophotometer. The results showed an influence of wax age on the heavy metal contents, where the mean concentrations of Fe, Cr, Zn, Cu, Ni, Mn, Pb, Cd, and Co were 5.041, 2.728, 2.480, 2.573, 2.547, 1.204, 0.185, 0.054, and 0.054 ppm, respectively, of samples aged 5 years old, while the mean concentrations of samples aged 1 year old were 2.068, 1.008, 0.364, 0.651, 0.890, 0.222, 0.040, 0.024, and 0.027 ppm, respectively. Beeswax samples from the first to fifth years old of comb age show a substantial difference in metal content. Pb, Cd, and Co were reported at the lowest levels in the ranges 0.040 to 0.185, 0.024 to 0.054, and 0.027 to 0.054 ppm, respectively.
Discussions
Because beeswax is not directly consumed by people, however, beeswax has significant biological and ecological significance as the honeybee brood nest because the comb cells are where adult bees store their food. Beeswax is a highly effective accumulator of heavy metals, such as Fe, Cr, Zn, Cu, Ni, Mn, Pb, Cd, and Co (Gajger et al. 2019). There is little research on the heavy metal content of beeswax, and there is not enough information on how it affects the quality of the comb (Bommuraj et al. 2019; Ullah et al. 2022).
The aim of the current study was to evaluate the heavy metals content of beeswax at different ages. Beeswax’s heavy metal composition depends on a number of variables, including its geography and beeswax age (Gajger et al. 2019). The wax comb samples were collected from an apiary in the Behaira governorate region of Egypt. The concentrations of heavy metals Fe, Cr, Zn, Cu, Ni, Mn, Pb, Cd, and Co were reported. The wax age had an effect on heavy metal concentrations, as shown in Table 1. Where the highest contents of heavy metals recorded for wax at the 5th year old and the lowest for wax at the 1st year old. Fe, Cr, Zn, Cu, Ni, and Mn were reported highly concentrations compared to Co, Cd, and Pb.
In the current study, the mean content of Fe ranged from 2.068 to 5.041 ppm, which is less than reported by Gajger et al. where the content of Fe ranged from 56.470 to 285 ppm (Gajger et al. 2019). The content of Fe from the various environmental sources was reported within the range of 1.080 to 334 µg/g (Formicki et al. 2013) and 5.972–18,516 mg/g (Aljedani 2020). The higher concentration of Fe may be due to different geographical locations where Fe may be co-occur in areas with similar environmental pollution loads. However, Fe is required not just for haemoglobin formation and oxygen transport but also for the activity of several enzymes. Fe metabolism disorders are among the most frequent illnesses in humans, ranging from anaemia to Fe excess and perhaps neurodegenerative disease. The amount of Fe in your diet depends on the age and gender (Abbaspour et al. 2014).
Cr is one of the 14 most dangerous heavy metals and one of the top priority pollutants, according to the Environmental Protection Agency. Cr is a naturally occurring element that is often used in industrial processes. Humans and other living things require trace levels of the element. Cr is an essential element that has beneficial effects on humans and plays a significant role in blood sugar regulation and lowering. A high dose of Cr can cause a variety of problems, including death in humans where the cytotoxic and genotoxic effects (Speer et al. 2019). The World Health Organization (WHO) set a daily limit of 250 g for Cr (ANS) 2010). In the current study, the content of Cr was 1.008 to 2.728 ppm, based the environmental conditions and regions, the Cr content were different. The presence of Cr accumulated in the wax could be considered an indicator of environmental pollution. The content of Cr ranged from 41.030 to 56.280 ppm (Gajger et al. 2019), 2.016, 2.300, and 3.920 mg/g (Aljedani 2020), 82 to 982 µg/kg (Zafeiraki et al. 2022), 432 µg/kg (Bommuraj et al. 2019), 131.200, 85.770, and 247.600 µg/kg based on the locations of beeswax from three different regions (Ćirić et al. 2021).
A similar pattern was also observed for Zn levels in beeswax, ranging from 1 to 81.200 µg/g (Formicki et al. 2013), 5.707 µg/g (Ullah et al. 2022), 19.699, 6.272, and 0.776 mg/g (Aljedani 2020). Zn is one of the most critical and necessary elements, with major public health implications. Zn is essential for the production of protein and collagen, which aid in wound healing and the maintenance of healthy skin. Growth and developmental delay, sensitivity to infections, delayed sexuality, mental fatigue, and skin toxicity are all possible consequences of the deficiency (Prasad 2020; Lu et al. 2023).
Cu oral intake is usually not harmful to humans. Ingestion of excessive doses of soluble Cu salts can result in severe gastrointestinal discomfort, on rare occasions, and liver damage in vulnerable people exposed repeatedly (Taylor et al. 2020). According to the current work, age of wax influences in Cu content, which ranged from 0.651 to 2.573 ppm, were less significant content than those from different regions that contain high the levels of Cu: 12.8 to 40.93 ppm (Gajger et al. 2019), 1.034, 1.139, 1.913, and 0.095 mg/g (Aljedani 2020), and also 280 to 4244 µg/kg from three regions (Zafeiraki et al. 2022).
Ni is a trace mineral that is necessary for the survival of many animal species, microorganisms, and plants. Adults’ Ni requirements range from 25 to 35 g/day (Anke et al. 1995). Excessive Ni consumption and exposure can result in allergies, cardiovascular and renal illness, lung fibrosis, lung, and nasal cancer. In adults and adolescents, an oral reference dosage of 0.040 mg Cu/kg/day would be protective against acute or chronic toxicity (Genchi et al. 2020; Taylor et al. 2020). Ni is a heavy metal that is detected in wax at different ages, from 1 to 5 years old, at a ratio of 0.890 to 2.547 ppm. The previously published data revealed different concentrations of Ni in beeswax ranged from 194 to 1919 µg/kg (Zafeiraki et al. 2022), 12.170 to 17.830 ppm (Gajger et al. 2019), 15 to 313 µg/g (Formicki et al. 2013), 0.474, 0.598, and 0.678 mg/g (Aljedani 2020) and 0.251 µg/g (Ullah et al. 2022).
Also, Mn content varies based on factors including; geography. The values were 0.182 to 41,904 mg/kg (Zafeiraki et al. 2022), 16.630 to 32.870 ppm (Gajger et al. 2019), 22.200 to 450 µg/g (Formicki et al. 2013), 0.365, 0.501, 1.311, and 0.414 mg/g (Aljedani 2020). Mn is available in low amounts in a range of food sources, providing enough Mn content to support numerous physiological functions in the human body. However, as Mn’s value in a number of sectors grows, so does the risk of overexposure to this transition metal, which can have neurotoxic adverse consequences (Peres et al. 2016).
Pb, Cd, and Co were listed as minor metals in this study. Pb is considered to be one of the most serious environmental toxins, and various studies have shown that it is a cause of health issues. Pb poisoning can induce respiratory, neurological, digestive, cardiovascular, and urinary disorders (Boskabady et al. 2018). When compared to the data displayed in Table 1, the Pd incidence reported in earlier reports and records had a higher concentration (1.230 to 5.430 ppm) (Gajger et al. 2019). Lower Pd levels have been observed to vary between 29.250, 111.800, and 401.100 µg/kg (Ćirić et al. 2021), 255 µg/kg (Bommuraj et al. 2019), 104 to 313 µg/g (Formicki et al. 2013), 0.114, 0.137, and 0.215 mg/g (Aljedani 2020). The concentration of Pb detected in the wax samples depends on the region of the apiaries. Zafeiraki et al. reported the maximum concentrations of Pb from Poša was 3.193 mg/kg while the lowest is 59 µg/kg in Strážske.
Cd is a poisonous heavy metal that is non-essential, an environmental toxin, and harmful even at low concentrations, with no recognized positive role in the human body. Cd consumption has an especially severe impact on fertility and cardiovascular disease (Kumar and Sharma 2019; Genchi et al. 2020). Cd was in the range 0.024 to 0.054 ppm, and the value of Cd was reported by Zafeiraki et al. at 29 to 60 µg/kg (Zafeiraki et al. 2022), 470 to 9870 µg/g (Formicki et al. 2013), 0.059, 0.060, 0.075, and 0.00 mg/g (Aljedani 2020).
Co was also detected at the range of 29 to 129 µg/kg which is less than the data reported in the current study (Zafeiraki et al. 2022). Co is a trace element that is needed for human health and is absorbed through food. As a metal ingredient of vitamin B12, cobalt has a physiologically important function (Cooper, B.A. and Paranchych 1961). However, when consumed in excess, they cause a variety of negative health consequences, most notably neurological, cardiovascular, and endocrine deficiencies (Leyssens et al. 2017).
Conclusion
The purpose of the current research was to determine whether samples of beeswax taken at various ages may include substantial differences in the values of heavy metal concentrations. Beeswax taken from Egypt’s Behaira governorate was tested for nine heavy metals. According to the study, new beeswax combs only contain fewer amounts of heavy metals. The highest metal concentrations were reported in wax aged 5 years old.
Beekeepers should adopt a new philosophy towards beeswax combs and regularly utilize fresh wax foundation to help decrease heavy metal contamination of honeybee hives and their products, which is also preferable for consumers and the performance of honeybee colonies.
References
Abbaspour N, Hurrell R, Kelishadi R (2014) Review on iron and its importance for human health. J Res Med Sci 19:164–174
Abd Al-Fattah MAAW, Yehia Ibrahim Y, Ibrahim Haggag M (2021) Some biological aspects of honey bee colonies in relation to the age of beeswax combs. J Apic Res 60:405–413. https://doi.org/10.1080/00218839.2021.1899657
Adugna E, Hymete A, Birhanu G, Ashenef A (2020) Determination of some heavy metals in honey from different regions of Ethiopia. Cogent Food Agric 6:1764182–1764196. https://doi.org/10.1080/23311932.2020.1764182
Al-Waili N (2005) Clinical and mycological benefits of topical application of honey, olive oil and beeswax in diaper dermatitis. Clin Microbiol Infect 11:160–163
Aljedani DM (2020) Revealing some elements and heavy metals in honeybee and beeswax samples collected from different environments. Entomol Appl Sci Lett 7:89–101
Amin M, Putra N, Kosasih EA et al (2017) Thermal properties of beeswax/graphene phase change material as energy storage for building applications. Appl Therm Eng 112:273–280. https://doi.org/10.1016/j.applthermaleng.2016.10.085
Anke M, Angelow L, Glei M et al (1995) The biological importance of nickel in the food chain. Fresenius J Anal Chem 352:92–96. https://doi.org/10.1007/BF00322304
Bommuraj V, Chen Y, Klein H et al (2019) Pesticide and trace element residues in honey and beeswax combs from Israel in association with human risk assessment and honey adulteration. Food Chem 299:125123. https://doi.org/10.1016/j.foodchem.2019.125123
Borsuk G, Sulborska A, Stawiarz E et al (2021) Capacity of honeybees to remove heavy metals from nectar and excrete the contaminants from their bodies. Apidologie 52:1098–1111. https://doi.org/10.1007/s13592-021-00890-6
Bosancic B, Zabic M, Mihajlovic D et al (2020) Comparative study of toxic heavy metal residues and other properties of honey from different environmental production systems. Environ Sci Pollut Res 27:38200–38211. https://doi.org/10.1007/s11356-020-09882-y
Boskabady M, Marefati N, Farkhondeh T et al (2018) The effect of environmental lead exposure on human health and the contribution of inflammatory mechanisms, a review. Environ Int 120:404–420. https://doi.org/10.1016/j.envint.2018.08.013
Bromenshenk JJ, Gudatis JL, Carlson SR et al (1991) Population dynamics of honey bee nucleus colonies exposed to industrial pollutants. Apidologie 22:359–369. https://doi.org/10.1051/apido:19910401
Burden CM, Morgan MO, Hladun KR et al (2019) Acute sublethal exposure to toxic heavy metals alters honey bee (Apis mellifera) feeding behavior. Sci Rep 9:4253–4262. https://doi.org/10.1038/s41598-019-40396-x
Chaireh S, Ngasatool P, Kaewtatip K (2020) Novel composite foam made from starch and water hyacinth with beeswax coating for food packaging applications. Int J Biol Macromol 165:1382–1391. https://doi.org/10.1016/j.ijbiomac.2020.10.007
Ćirić J, Spirić D, Baltić T et al (2021) Honey bees and their products as indicators of environmental element deposition. Biol Trace Elem Res 199:2312–2319. https://doi.org/10.1007/s12011-020-02321-6
Cooper BA, Paranchych W (1961) Selective uptake of specifically bound cobalt-58 vitamin 812 by human and mouse tumour cells. Nature 191:393–395
EFSA Panel on Food Additives and Nutrient Sources added to Food (2010) Scientific opinion on the safety of trivalent chromium as a nutrient added for nutritional purposes to foodstuffs for particular nutritional uses and foods intended for the general population (including food supplements). EFSA J 8:1882–1927. https://doi.org/10.2903/j.efsa.2010.1882
El-Seedi HR, Ahmed HR, El-Wahed AAA et al (2022a) Bee stressors from an immunological perspective and strategies to improve bee health. Vet Sci 9:199–221
El-Seedi HR, El-Wahed AAA, Naggar Y, Al et al (2022b) Insights into the role of natural products in the control of the honey bee gut parasite (nosema spp). Animals 12:3062. https://doi.org/10.3390/ani12213062
El-Seedi HR, El-Wahed AAA, Zhao C et al (2022c) A spotlight on the egyptian honeybee (Apis mellifera lamarckii). Animals 12:2749. https://doi.org/10.3390/ani12202749
El-Wahed AAA, Farag MA, Eraqi WA et al (2021) Unravelling the beehive air volatiles profile as analysed via solid-phase microextraction (SPME) and chemometrics. J King Saud Univ - Sci 33:101449–101456. https://doi.org/10.1016/j.jksus.2021.101449
Formicki G, Greń A, Stawarz R et al (2013) Metal content in honey, propolis, wax, and bee pollen and implications for metal pollution monitoring. Pol J Environ Stud 22:99–106
Fratini F, Cilia G, Turchi B, Felicioli A (2016) Beeswax: a minireview of its antimicrobial activity and its application in medicine. Asian Pac J Trop Med 9:839–843. https://doi.org/10.1016/j.apjtm.2016.07.003
Gajger IT, Kosanović M, Bilandžić N et al (2016) Variations in lead, cadmium, arsenic, and mercury concentrations during honeybee wax processing using casting technology. Arh Hig Rada Toksikol 67:223–228. https://doi.org/10.1515/aiht-2016-67-2780
Gajger IT, Kosanović M, Oreščanin V et al (2019) Mineral content in honeybee wax combs as a measurement of the impact of environmental factors. Bull Environ Contam Toxicol 103:697–703. https://doi.org/10.1007/s00128-019-02713-y
Genchi G, Carocci A, Lauria G, Sinicropi MS (2020) Nickel: human health and environmental toxicology. Int J Environ Res Public Health 17:679–700
Hesketh H, Lahive E, Horton AA et al (2016) Extending standard testing period in honeybees to predict lifespan impacts of pesticides and heavy metals using dynamic energy budget modelling. Sci Rep 6:37655–37666. https://doi.org/10.1038/srep37655
Kast C, Kilchenmann V (2022) An in vitro model for assessing the toxicity of pesticides in beeswax on honey bee larvae. Chemosphere 287:132214–132221. https://doi.org/10.1016/j.chemosphere.2021.132214
Khalifa SAM, Elshafiey EH, Shetaia AA et al (2021) Overview of bee pollination and its economic value for crop production. Insects 12:688–703. https://doi.org/10.3390/insects12080688
Kumar S, Sharma A (2019) Cadmium toxicity: Effects on human reproduction and fertility. Rev Environ Health 8:47–58. https://doi.org/10.1515/reveh-2019-0016
Lee MH (1995) Official methods of analysis of AOAC International (16th edn): edited by Patricia A. Cunniff, AOAC International, 1995. $359.00 (North America)/$399.00 (elsewhere)(xxvi + 1899 pages) ISBN 0 935 584 54 4
Leyssens L, Vinck B, Van Der Straeten C et al (2017) Cobalt toxicity in humans—A review of the potential sources and systemic health effects. Toxicology 387:43–56. https://doi.org/10.1016/j.tox.2017.05.015
Lu CW, Pang JHS, Ko YS, Chang CJ, Wang CW, Chen WT, Chen CB, Hui RCY, Hung SI, Lu LY, Lu KL (2023) Zinc deficiency associated with cutaneous toxicities induced by epidermal growth factor receptor tyrosine kinase inhibitor therapy in patients with lung adenocarcinoma. J Eur Acad Dermatology Venereol 37:328–339
Mao C, Song Y, Chen L et al (2019) Human health risks of heavy metals in paddy rice based on transfer characteristics of heavy metals from soil to rice. CATENA 175:339–348. https://doi.org/10.1016/j.catena.2018.12.029
Ngat TT, Xuan Lam T, Gia Minh H, Thi Phuong Lien N (2020) Honeybee products as potential bioindicators of heavy metals contamination from Northern Vietnam. Vietnam J Biotechnol 18:373–384. https://doi.org/10.15625/1811-4989/18/2/15086
Peres TV, Schettinger MRC, Chen P et al (2016) Manganese-induced neurotoxicity: a review of its behavioral consequences and neuroprotective strategies. BMC Pharmacol Toxicol 17:57–76. https://doi.org/10.1186/s40360-016-0099-0
Pohl P (2009) Determination of metal content in honey by atomic absorption and emission spectrometries. TrAC - Trends Anal Chem 28:117–128. https://doi.org/10.1016/j.trac.2008.09.015
Prasad AS (2020) Lessons learned from experimental human model of zinc deficiency. J Immunol Res 2020:. https://doi.org/10.1155/2020/9207279
Salama S, Shou Q, Abd El-Wahed AA et al (2022) Royal jelly: beneficial properties and synergistic effects with chemotherapeutic drugs with particular emphasis in anticancer strategies. Nutrients 14:4166–4177. https://doi.org/10.3390/nu14194166
Smith ML, Koenig PA, Peters JM (2017) The cues of colony size: how honey bees sense that their colony is large enough to begin to invest in reproduction. J Exp Biol 220:1597–1605. https://doi.org/10.1242/jeb.150342
Speer RM, Wise SS, Croom-Perez TJ et al (2019) A comparison of particulate hexavalent chromium cytotoxicity and genotoxicity in human and leatherback sea turtle lung cells from a one environmental health perspective. Toxicol Appl Pharmacol 376:70–81. https://doi.org/10.1016/j.taap.2019.05.013
Svečnjak L, Chesson LA, Gallina A et al (2019) Standard methods for Apis mellifera beeswax research. J Apic Res 58:1–108. https://doi.org/10.1080/00218839.2019.1571556
Taylor AA, Tsuji JS, Garry MR et al (2020) Critical review of exposure and effects: implications for setting regulatory health criteria for ingested copper. Environ Manage 65:131–159. https://doi.org/10.1007/s00267-019-01234-y
Ullah R, Jan FA, Gulab H et al (2022) Metals contents in honey, beeswax and bees and human health risk assessment due to consumption of honey: a Case study from selected districts in Khyber Pakhtunkhwa, Pakistan. Arch Environ Contam Toxicol 82:341–354. https://doi.org/10.1007/s00244-021-00910-7
Wang L, Yin X, Gao S et al (2020) In vitro oral bioaccessibility investigation and human health risk assessment of heavy metals in wheat grains grown near the mines in North China. Chemosphere 252:126522. https://doi.org/10.1016/j.chemosphere.2020.126522
Zafeiraki E, Sabo R, Kasiotis KM et al (2022) Adult honeybees and beeswax as indicators of trace elements pollution in a vulnerable environment: distribution among different apicultural compartments. Molecules 27:6629–6654. https://doi.org/10.3390/molecules27196629
Zhang X, Yang L, Li Y et al (2012) Impacts of lead/zinc mining and smelting on the environment and human health in China. Environ Monit Assess 184:2261–2273. https://doi.org/10.1007/s10661-011-2115-6
Acknowledgements
Aida A. Abd El-Wahed thanks Department of Bee Research, Plant Protection Research Institute, Agricultural Research Centre, Egypt for support.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Nadia M. Hassona and Aida A. Abd El-Wahed. The first draft of the manuscript was written by Aida A. Abd El-Wahed and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
No potential conflict of interest was reported by the author.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hassona, N.M., El-Wahed, A.A.A. Heavy Metal Concentrations of Beeswax (Apis mellifera L.) at Different Ages. Bull Environ Contam Toxicol 111, 26 (2023). https://doi.org/10.1007/s00128-023-03779-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00128-023-03779-5