Abstract
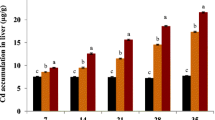
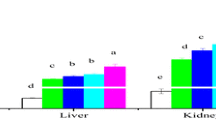
The aims of this study were to investigate the accumulation (15 days) and elimination (15 and 30 days) of cadmium (Cd) in the liver, gill, kidney and muscle of Oreochromis niloticus following exposures to different concentrations (1 and 2 mg/L) of Cd at different water temperatures (20, 25, 30 oC). Additionally, responses of oxidative stress biomarkers (superoxide dismutase, SOD; catalase CAT; glutathione peroxidase, GPx and malondialdehyde, MDA) of the liver were determined following Cd exposures. In accumulation period, Cd levels increased significantly in all the tissues at all temperatures and tissue accumulation order was kidney > liver > gill. All tissues, except the muscle, accumulated Cd in relation to exposure concentrations and water temperatures. There was no measurable level of Cd accumulation in the muscle, except in fish exposed to 2 mg Cd/L at 30 oC. Likewise, elimination of Cd from the tissues also increased in depends on periods and water temperatures. The order of Cd elimination from the tissues was gill > liver > kidney. The oxidative stress biomarkers also responded to both Cd exposure and temperature increases. The activities of antioxidant enzymes such as SOD, CAT, GPx and MDA levels in the liver increased in relation to increases in Cd concentrations and water temperatures.

Similar content being viewed by others
References
Abbas WT, Authman MMN, Darwish DA, Kenawy AM, Abumourad IMK, Ibrahim TB (2019) Cadmium toxicity-induced oxidative stress and genotoxic effects on nile tilapia (Oreochromis niloticus L.) fish: The protective role of fenugreek (Trigonella foenum-graecum) seeds. Egyp J Aquatic Biol Fisheries 23(5):193–215. DOI: https://doi.org/10.21608/EJABF.2019.64715
Abdel-Baki AS, Dkhil MA, Al-Quraishy S (2011) Bioaccumulation of some heavy metals in tilapia fish relevant to their concentration in water and sediment of Wadi Hanifah, Saudi Arabia. Afr J Biotechnol 10:2541–2547. DOI: https://doi.org/10.5897/AJB10.1772
Abdel-Tawwab M, Wafeek M (2017) Fluctuations in water temperature affected waterborne cadmium toxicity: hematology, anaerobic glucose pathway, and oxidative stress status of nile tilapia, Oreochromis niloticus (L.). Aquaculture 477:106–111. https://doi.org/10.1016/j.aquaculture.2017.05.007
Ahn TY, Park HJ, Kim JH, Kang JC (2020) Effects of antioxidant enzymes and bioaccumulation in eels (Anguilla japonica) by acute exposure of waterborne cadmium. Fisher Aquat Sci 23:23. https://doi.org/10.1186/s41240-020-00166-7
Almeida JA, Novelli ELB, Dal Pai Silva M, Junior RA (2001) Environmental cadmium exposure and metabolic responses of the nile tilapia, Oreochromis niloticus. Environ Pollut 114:169–175. https://doi.org/10.1016/S0269-7491(00)00221-9
Atli G, Canli M (2007) Enzymatic responses to metal exposures in a freshwater fish Oreochromis niloticus. Comp Biochem and Physiol Part C 145:282–287. https://doi.org/10.1016/j.cbpc.2006.12.012
Atli G, Canli EG, Eroglu A, Canli M (2016) Characterization of antioxidant system parameters in four freshwater fish species. Ecotox Environ Saf 126:30–37. https://doi.org/10.1016/j.ecoenv.2015.12.012
Banaee M, Haghi BN, Zoheiri F (2013) LC50 and bioaccumulation of lead nitrate (Pb(NO3)2) in goldfish (Carassius auratus). Intern J Aquatic Biol 1(5):233–239. https://doi.org/10.22034/ijab.v1i5.153
Baykan U, Atli G, Canli M (2007) The effects of temperature and metal exposures on the profiles of metallothionein-like proteins in Oreochromis niloticus. Environ Toxicol Pharm 23(1):33–38. https://doi.org/10.1016/j.etap.2006.06.002
Beutler E (1984) Red cell metabolism: A manual of biochemical methods, 2nd edn. Grune and Stratton, New York, USA
Canli EG, Atli G, Canli M (2016) Responses of the antioxidant and osmoregulation systems of fish erythrocyte following copper exposures in differing calcium levels. Bull Environ Contam Toxicol 97(5):601–608. DOI https://doi.org/10.1007/s00128-016-1931-3
Canli EG (2021) Individual and combined effects of salinity and nanoparticles (Al2O3, TiO2) on the activity of antioxidant enzymes in freshwater fish (Oreochromis niloticus). Turkish J Fisher Aquat Sci 21(8):415–424. https://doi.org/10.4194/1303-2712-v21_8_05
Das D, Moniruzzaman M, Sarbajna A, Chakraborty SB (2017) Effect of heavy metals on tissue-specific antioxidant response in Indian major carps. Environ Sci Pollut Res 24:18010–18024. DOI https://doi.org/10.1007/s11356-017-9415-5
DaSilva GSF, Glass ML, Branco LGS (2013) Temperature and respiratory function in ectothermic vertebrates. J Therm Biol 38:55–63. https://doi.org/10.1016/j.jtherbio.2012.11.001
De Conto-Cinier C, Petit-Ramel M, Faure R, Garin D, Baouvet Y (1999) Kinetics of cadmium accumulation and elimination in carp Cyprinus carpio tissues. Comp Biochem Physiol C 122(3):345–352. https://doi.org/10.1016/S0742-8413(98)10132-9
De Smet H, Blust R (2001) Stress responses and changes in protein metabolism in carp Cyprinus carpio during cadmium exposure. Ecotox Environ Safety 48:255–262. https://doi.org/10.1006/eesa.2000.2011
Durmus M, Kosker AR, Ozogul Y, Aydın M, Ucar Y, Ayas D, Ozogul F (2018) The effects of sex and season on the metal levels and proximate composition of red mullet (Mullus barbatus Linnaeus 1758) caught from the Middle Black Sea. Hum Ecol Risk Assess 24(3):731–742. https://doi.org/10.1080/10807039.2017.1398071
Ebrahimi M, Taherianfard M (2011) The effects of heavy metals exposure on reproductive systems of cyprinid fish from Kor River. Iran J Fisher Sci 10(1):13–24. http://hdl.handle.net/1834/11305
El-Shenawy NS, EL-Hak HNG, Ghobashy MA, Mansour FA, Soliman MFM (2021) Using antioxidant changes in liver and gonads of Oreochromis niloticus as biomarkers for the assessment of heavy metals pollution at Sharkia province, Egypt. Region Stud Mar Science 46:1–8. https://doi.org/10.1016/j.rsma.2021.101863
Erdem C, Cicik B, Karayakar S, Karayakar F, Karaytuğ S (2005) Clarias gariepinus (Burchell, 1822)’da kadmiyum’un solungaç, karaciğer, böbrek, dalak ve kas dokularındaki birikim ve arıtımı SDÜ Eğirdir. Su Ürünleri Fakültesi Dergisi 1(2):18–26
Garcia-Santos S, Fontainhas-Fernandes A, Wilson JM (2006) Cadmium tolerance in the nile tilapia (Oreochromis niloticus) following acute exposure: assessment of some ionoregulatory parameters. Environ Toxicol 33–46. DOI https://doi.org/10.1002/tox.20152
Gupta P, Srivastava N (2006) Effects of sub-lethal concentrations of zinc on histological changes and bioaccumulation of zinc by kidney of fish Channa punctatus (Bloch). J Environ Biol 27(2):211–215 Free paper downloaded from: www.jeb.co.in
Ha LT, Hoa BT, Dau PT (2021) Effects of heavy metals on the activity of catalase and glutathione-s-transferase in Nile tilapia fish (Oreochromis niloticus). VNU J Science: Nat Sci Tech 37(4):82–87. https://doi.org/10.25073/2588-1140/vnunst.5339
IAEA (2003) Trace elements and methylmercury in fish tissue.International Atomic Energy Agencyhttps://nucleus.iaea.org/rpst/Documents/rs_iaea-407.pdf. Retrieved 24 January 2003
Jezierska B, Witeska M (2006) The metal uptake and accumulation in fish living in polluted waters. Soil Water Poll Monitor Protec Remediation 3(23):107–113. DOI: https://doi.org/10.1007/978-1-4020-4728-2_6
Kanak EG, Dogan Z, Eroglu A, Atli G, Canli M (2014) Effects of fish size on the response of antioxidant systems of Oreochromis niloticus following metal exposures. Fish Physiol Biochem 40:1083–1091. DOI: https://doi.org/10.1007/s10695-014-9907-x
Karaytuğ S, Erdem C, Cicik B (2007) Accumulation of cadmium in the gill, liver, kidney, spleen, muscle and brain tissues of Cyprinus carpio. Ekoloji 16(63):16–22
Kim JH, Kim SK, Hur YB (2019) Hematological parameters and antioxidant responses in olive flounder Paralichthys olivaceus in biofloc depend on water temperature. J Therm Biol 82:206–212. https://doi.org/10.1016/j.jtherbio.2019.04.013
Kraal MH, Kraak MHS, De Groot CJ, Davids C (1995) Uptake and tissue distribution of dietary and aqueous cadmium by carp (Cyprinus carpio). Ecotoxicol Env Saf 31:179–183. https://doi.org/10.1006/eesa.1995.1060
Lee DC, Choi YJ, Kim JH (2022) Toxic effects of waterborne cadmium exposure on hematological parameters, oxidative stress, neurotoxicity, and heat shock protein 70 in juvenile olive flounder, Paralichthys olivaceus. Fish Shell Immun 122:476–483. https://doi.org/10.1016/j.fsi.2022.02.022
Lowry OH, Rosenbrough NJ, Farr A, Randall RJ (1951) Protein measurement with the folin-phenol reagent. J Biochem 193:265–277
McCords JM, Fridovich I (1969) Superoxide dismutase: An enzymic function for erythrocuprein (Hemocuprein). J Biol Chem 244(22):6049–6065. https://doi.org/10.1016/S0021-9258(18)63504-5
Morgan JD, Sakamoto T, Grau EG, Iwama GK (1997) Physiological and respiratory responses of the mozambique tilapia (Oreochromis mossambicus) to salinity acclimation. Comp Biochem Physiol A 117(3):391–398. https://doi.org/10.1016/S0300-9629(96)00261-7
Morshdy AEMA, Darwish WS, Hussein MAM, Mohamed MAA, Hussein MMA (2021) Lead and cadmium content in Nile tilapia (Oreochromis niloticus) from Egypt: A study for their molecular biomarkers. Scient Afr 12:e00794. https://doi.org/10.1016/j.sciaf.2021.e00794
Muramoto S (1983) Elimination of copper from cu-contaminated fish by long term exposure to EDTA and freshwater. J Environ Sci Health 18(3):455–461
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Olsvik PA, Softeland l, Hevroy EM, Rasinger JD, Waagbo R (2016) Fish pre-acclimation temperature only modestly affects cadmium toxicity in Atlantic salmon hepatocytes. J Therm Biol 57:21–34. https://doi.org/10.1016/j.jtherbio.2016.02.003
Pourkhabbaz A, Alipour H, Zarei I (2015) Bioaccumulation and depuration rates of zinc by Capoeta fusca under controlled conditions. Water Qual Expo Health 7:187–191. https://doi.org/10.1007/s12403-014-0139-1
Rathore R, Khangarot B (2002) Effects of temperature on the sensitivity of sludge worm Tubifex tubifex Müller to selected heavy metals. Ecotoxicol Environ Saf 53:27–36. https://doi.org/10.1006/eesa.2001.2100
Sassi A, Annabi A, Kessabi K, Kerkeni A, Said K, Messaoudi I (2010) Influence of high temperature on cadmium-induced skeletal deformities in juvenile mosquitofish (Gambusia affinis). Fish Physiol Biochem 36:403–409. https://doi.org/10.1007/s10695-009-9307-9
Sun M, Hu F, Wang T, Zhang T, Jing Y, Guo W, Chen Q, Liu G (2022) Effect of temperature on the toxicokinetics and gene expression of the pacific cupped oyster Crassostrea gigas exposed to cadmium. Comp Biochem Physiol Part C 253:109252. https://doi.org/10.1016/j.cbpc.2021.109252
Suresh N (2009) Effect of cadmium chloride on liver, spleen and kidney melano macrophage centres in Tilapia mossambica. J Environ Biol 30(4):505–508
Thabet IA, Tawadrous WE, Samy AM (2019) Pollution induced change of liver of Oreochromis niloticus: metals accumulation and histopathological response. World J Adv Res Rev 2(2):25–35. https://doi.org/10.30574/wjarr.2019.2.2.0020
Tang UM, Muchlisin ZA, Syawal H, Masjudi H (2017) Effect of water temperature on the physiological stress and growth performance of tapah (Wallago leeri) during domestication. Arch Pol Fish 25:165–171. DOI https://doi.org/10.1515/aopf-2017-0016
Vadlamani SK, Basuri CH, Yallapragada PR (2018) Lipid peroxidation products in fry of Labeo rohita and Cyprinus carpio on exposure to sub lethal cadmium: A comparative study. Europ Acad Res 5(11):6048–6064. DOI:https://doi.org/10.22438/jeb/39/4/MRN-644
Verma Y, Rani V, Rana SVS (2020) Assessment of cadmium sulphide nanoparticles toxicity in the gills of a fresh water fish. Environ Nanotech Monitor&Manage 13:100280. https://doi.org/10.1016/j.enmm.2019.100280
Verma S, Batoye S, Jindal R (2021) Protective efficacy of naringenin against cadmiuminduced redox imbalance in Labeo rohita: an integrated biomarker approach. Environ Sci Poll Res. https://doi.org/10.1007/s11356-021-17703-z
Xiong X, Li H, Qiu N, Su L, Huang Z, Song L, Wang J (2020) Bioconcentration and depuration of cadmium in the selected tissues of rare minnow (Gobiocypris rarus) and the effect of dietary mulberry leaf supplementation on depuration. Environ Toxicol Pharm 73:103278. https://doi.org/10.1016/j.etap.2019.103278
Younis ESM, Abdel-Warith AWAM, Al-Asgah NA, Ebaid H, Mubarak M (2013) Histological changes in the liver and intestine of nile tilapia, Oreochromis niloticus, exposed to sublethal concentrations of cadmium. Pakistan J Zool 45(3):833–841. https://www.researchgate.net/publication/259568941
Acknowledgements
This work was supported by Mersin University, Scientific Research Projects Coordination Unit (Project Number: 2017-2-TP2-2555).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
The ethics committee approval was obtained from Mersin University Animal Experiments Local Ethical Committee by decision number 2017/06 dated 27/03/2017.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karayakar, F., Yurt, Ö., Cicik, B. et al. Accumulation and Elimination of Cadmium by the Nile Tilapia (Oreochromis niloticus) in differing Temperatures and Responses of Oxidative Stress Biomarkers. Bull Environ Contam Toxicol 109, 1126–1134 (2022). https://doi.org/10.1007/s00128-022-03616-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-022-03616-1