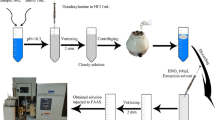
Abstract
The present study describes the development of a fit-for-purpose analytical procedure for the detection of Ag NPs in different marine organisms by Solid Sampling Continuous Source High Resolution Atomic Absorption Spectrometry (SS-CS-HR-AAS). The detection is based on the observation of the Ag absorption peak and its atomization delay tad which is different for ionic Ag and Ag NPs. The temperature program was optimized in order to achieve the maximum difference between the t ad (Δtad ). The method was first developed using biota CRMs spiked with different Ag NPs standard solutions or Ag+ , at the same concentration. Then, laboratory exposure experiments were performed on mussels and marine sponges. The results showed that the developed methodology is suitable for the detection of Ag NPs for both groups of organisms, showing Δtad up to 3.1 s. The developed method is therefore a promising tool to assess the presence of AgNPs in marine invertebrates.



Similar content being viewed by others
References
Abbott Chalew TE, Galloway JF, Graczyk TK (2012) Pilot study on effects of nanoparticle exposure on Crassostrea virginica hemocyte phagocytosis. Mar Pollut Bull 64:2251–2253. https://doi.org/10.1016/j.marpolbul.2012.06.026
Adamek D, Sliwinski J, Ostaszewska T et al (2018) Effect of Copper and Silver Nanoparticles on Trunk Muscles in Rainbow Trout (Oncorhynchus mykiss, Walbaum, 1792). Turkish J Fish Aquat Sci 18:781–788. https://doi.org/10.4194/1303-2712-v18_6_04
Artiaga G, Ramos K, Ramos L et al (2015) Migration and characterisation of nanosilver from food containers by AF4-ICP-MS. Food Chem 166:76–85. https://doi.org/10.1016/j.foodchem.2014.05.139
Batista D, Pascoal C, Cássio F (2020) The Increase in Temperature Overwhelms Silver Nanoparticle Effects on the Aquatic Invertebrate Limnephilus sp. Environ Toxicol Chem 39:1429–1437. https://doi.org/10.1002/etc.4738
Blasco C, Picó Y (2011) Determining nanomaterials in food. TrAC Trends Anal Chem 30:84–99. https://doi.org/10.1016/j.trac.2010.08.010
Echegoyen Y, Nerín C (2013) Nanoparticle release from nano-silver antimicrobial food containers. Food Chem Toxicol 62:16–22. https://doi.org/10.1016/j.fct.2013.08.014
Fabrega J, Luoma SN, Tyler CR et al (2011) Silver nanoparticles: Behaviour and effects in the aquatic environment. Environ Int 37:517–531. https://doi.org/10.1016/j.envint.2010.10.012
Feichtmeier NS, Leopold K (2014) Detection of silver nanoparticles in parsley by solid sampling high-resolution – continuum source atomic absorption spectrometry. 3887–3894
Feichtmeier NS, Ruchter N, Zimmermann S et al (2016) A direct solid sampling analysis method for the detection of silver nanoparticles in biological matrices. Anal Bioanal Chem 408:295–305. https://doi.org/10.1007/s00216-015-9108-1
Gagné F, Turcotte P, Gagnon C (2012) Screening test of silver nanoparticles in biological samples by graphite furnace-atomic absorption spectrometry. 404:2067–2072. https://doi.org/10.1007/s00216-012-6258-2
Genta-Jouve G, Cachet N, Oberhänsli F et al (2012) Comparative bioaccumulation kinetics of trace elements in Mediterranean marine sponges. Chemosphere 89:340–349. https://doi.org/10.1016/j.chemosphere.2012.04.052
Gomes T, Araújo O, Pereira R et al (2013) Genotoxicity of copper oxide and silver nanoparticles in the mussel Mytilus galloprovincialis. Mar Environ Res 84:51–59. https://doi.org/10.1016/j.marenvres.2012.11.009
Gruszka J, Martyna A, Godlewska-Żyłkiewicz B (2021) Chemometric approach to discrimination and determination of binary mixtures of silver ions and nanoparticles in consumer products by graphite furnace atomic absorption spectrometry. Talanta 230. https://doi.org/10.1016/j.talanta.2021.122319
Gruszka J, Zambrzycka-Szelewa E, Kulpa JS, Godlewska-Zyłkiewicz B (2018) Discrimination between ionic silver and silver nanoparticles in consumer products using graphite furnace atomic absorption spectrometry. J Anal At Spectrom 33:2133–2142. https://doi.org/10.1039/c8ja00310f
Handy RD, Henry TB, Scown TM et al (2008a) Manufactured nanoparticles: their uptake and effects on fish-a mechanistic analysis. Ecotoxicology 17:396–409. https://doi.org/10.1007/s10646-008-0205-1
Handy RD, Owen R, Valsami-Jones E (2008b) The ecotoxicology of nanoparticles and nanomaterials: current status, knowledge gaps, challenges, and future needs. Ecotoxicology 17:315–325. https://doi.org/10.1007/s10646-008-0206-0
Jokar M, Rahman RA (2014) Study of silver ion migration from melt-blended and layered-deposited silver polyethylene nanocomposite into food simulants and apple juice. Food Addit Contam Part A 31:734–742. https://doi.org/10.1080/19440049.2013.878812
Lee S, Bi X, Reed RB et al (2014) Nanoparticle Size Detection Limits by Single Particle ICP-MS for 40 Elements. Environ Sci Technol 48:10291–10300. https://doi.org/10.1021/es502422v
Leopold K, Brandt A, Tarren H (2017) Sizing gold nanoparticles using graphite furnace atomic absorption spectrometry. J Anal At Spectrom 32:723–730. https://doi.org/10.1039/c7ja00019g
Loeschner K, Navratilova J, Købler C et al (2013) Detection and characterization of silver nanoparticles in chicken meat by asymmetric flow field flow fractionation with detection by conventional or single particle ICP-MS. Anal Bioanal Chem 405:8185–8195. https://doi.org/10.1007/s00216-013-7228-z
Magesky A, Pelletier É (2018) Cytotoxicity and physiological effects of silver nanoparticles on marine invertebrates. Adv Exp Med Biol 1048:285–309. https://doi.org/10.1007/978-3-319-72041-8_17
Magesky A, Ribeiro CAO, Pelletier É (2016) Physiological effects and cellular responses of metamorphic larvae and juveniles of sea urchin exposed to ionic and nanoparticulate silver. Aquat Toxicol 174:208–227. https://doi.org/10.1016/j.aquatox.2016.02.018
Marambio-Jones C, Hoek EMV (2010) A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J Nanoparticle Res 12:1531–1551. https://doi.org/10.1007/s11051-010-9900-y
McCarthy MP, Carroll DL, Ringwood AH (2013) Tissue specific responses of oysters, Crassostrea virginica, to silver nanoparticles. Aquat Toxicol 138–139:123–128. https://doi.org/10.1016/j.aquatox.2013.04.015
Navratilova J, Praetorius A, Gondikas A et al (2015) Detection of engineered copper nanoparticles in soil using single particle ICP-MS. Int J Environ Res Public Health 12:15756–15768. https://doi.org/10.3390/ijerph121215020
Ntim SA, Thomas TA, Begley TH, Noonan GO (2015) Characterisation and potential migration of silver nanoparticles from commercially available polymeric food contact materials. Food Addit Contam Part A 32:1003–1011. https://doi.org/10.1080/19440049.2015.1029994
Peters R, Herrera-Rivera Z, Undas A et al (2015) Single particle ICP-MS combined with a data evaluation tool as a routine technique for the analysis of nanoparticles in complex matrices. J Anal At Spectrom 30:1274–1285. https://doi.org/10.1039/c4ja00357h
Resano M, Garcia-Ruiz E, Garde R (2016) High-resolution continuum source graphite furnace atomic absorption spectrometry for the monitoring of Au nanoparticles. J Anal At Spectrom 31:2233–2241. https://doi.org/10.1039/c6ja00280c
Resano M, Mozas E, Crespo C et al (2010) Solid sampling high-resolution continuum source graphite furnace atomic absorption spectrometry to monitor the biodistribution of gold nanoparticles in mice tissue after intravenous administration. J Anal At Spectrom 25:1864–1873. https://doi.org/10.1039/c0ja00086h
Shaw BJ, Handy RD (2011) Physiological effects of nanoparticles on fish: A comparison of nanometals versus metal ions. Environ Int 37:1083–1097. https://doi.org/10.1016/j.envint.2011.03.009
Silva BF, Pérez S, Gardinalli P et al (2011) Analytical chemistry of metallic nanoparticles in natural environments. TrAC Trends Anal Chem 30:528–540. https://doi.org/10.1016/j.trac.2011.01.008
Tiede K, Boxall ABA, Tiede D et al (2009) A robust size-characterisation methodology for studying nanoparticle behaviour in ‘real’ environmental samples, using hydrodynamic chromatography coupled to ICP-MS. J Anal At Spectrom 24:964–972. https://doi.org/10.1039/B822409A
von Goetz N, Fabricius L, Glaus R et al (2013) Migration of silver from commercial plastic food containers and implications for consumer exposure assessment. Food Addit Contam Part A 30:612–620. https://doi.org/10.1080/19440049.2012.762693
Zhang W, Xiao B, Fang T (2018) Chemical transformation of silver nanoparticles in aquatic environments: Mechanism, morphology and toxicity. Chemosphere 191:324–334. https://doi.org/10.1016/j.chemosphere.2017.10.016
Acknowledgements
The authors are grateful to Mr Angus Taylor and Mr François Oberhaensli for their assistance during exposure experiments with mussels and sponges. The agency is grateful for the support provided to its Marine Environment Laboratories by the Government of the Principality of Monaco.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and analysis were performed by AMO under the supervision of EV. The first draft of the manuscript was written by AMO. EV and OT reviewed and corrected it. All authors read and approved the final manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Orani, A.M., Vassileva, E. & Thomas, O.P. Application of SS-CS-HR-AAS measurements for the detection of Ag nanoparticles in marine invertebrates. Bull Environ Contam Toxicol 109, 865–871 (2022). https://doi.org/10.1007/s00128-022-03573-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-022-03573-9