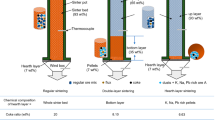
Abstract
The hydrothermal hydrogen reduction process for treating high-iron bauxite residue (red mud) was investigated, and the optimum conditions of alumina extraction as well as the enrichment of iron minerals were verified by experiments. Results show that the surface magnetization of Al-goethite under the function of hydrogen reduction accelerates its conversion to hematite and/or magnetite. This conversion releases the substituted Al in goethite as well as the undigested gibbsite/boehmite and further enriches the iron content in residue. After hydrothermal hydrogen reduction with H2/Red mud ratio of 0.085 mol/20 g at 270°C for 60 min, the alumina relative recovery ratio reaches 95.40% and the grade of iron (total iron in the form of iron element) in the residue can be enriched to 55.85%. Further, co-processing of the obtained iron-rich residue in the steel industry can achieve a significant reduction of red mud discharge.





Similar content being viewed by others
References
Cardenia C, Efthymios B, Dimitris P (2018) Iron recovery from bauxite residue through reductive roasting and wet magnetic separation. J Sustain Metall 5(1):9–19. https://doi.org/10.1007/s40831-018-0181-5
Cardenia C, Balomenos E, Tam PWY et al (2021) A combined soda sintering and microwave reductive roasting process of bauxite residue for iron recovery. Minerals 11(2):222. https://doi.org/10.3390/min11020222
Chen MS, Ding SM, Chen X et al (2018) Mechanisms driving phosphorus release during algal blooms based on hourly changes in iron and phosphorus concentrations in sediments. Water Res 133:153–164. https://doi.org/10.1016/j.watres.2018.01.040
Evans K (2016) The history, challenges, and new developments in the management and use of bauxite residue. J Sustain Metall 2(4):316–331. https://doi.org/10.1007/s40831-016-0060-x
Fei L, Hu Y, Wang L et al (2020) Dealkalization processes of bauxite residue: a comprehensive review. J Hazard Mater 403:123671. https://doi.org/10.1016/j.jhazmat.2020.123671
International Aluminium Institute (2020) https://www.world-aluminium.org
Li LY (2001) A study of iron mineral transformation to reduce red mud tailings. Waste Manag 21(6):525–534. https://doi.org/10.1016/S0956-053X(00)00107-0
Li XB, Wang YL, Zhou QS et al (2018) Reaction behaviors of iron and hematite in sodium aluminate solution at elevated temperature. Hydrometallurgy 175:257–265. https://doi.org/10.1016/j.hydromet.2017.12.004
Li XB, Zhou ZY, Wang YL et al (2020a) Enrichment and separation of iron minerals in gibbsitic bauxite residue based on reductive Bayer digestion. Trans Nonferrous Met Soc China 30(7):1980–1990. https://doi.org/10.1016/S1003-6326(20)65355-9
Li B, Wu H, Liu XL et al (2020b) Simultaneous removal of SO2 and NO using a novel method with red mud as absorbent combined with O3 oxidation. J Hazard Mater 392:122270. https://doi.org/10.1016/j.jhazmat.2020.122270
Li S, Kang Z, Liu W et al (2021) Reduction behavior and direct reduction kinetics of red mud-biomass composite pellets. J Sustain Metall 7(1):126–135. https://doi.org/10.1007/s40831-020-00326-y
Lu JF, Tsai CJ (2014) Hydrothermal phase transformation of hematite to magnetite. Nanoscale Res Lett 9(1):230. https://doi.org/10.1186/1556-276X-9-230
Pasechnik LA, Skachkov VM, Bogdanova EA et al (2020) A promising process for transformation of hematite to magnetite with simultaneous dissolution of alumina from red mud in alkaline me dium. Hydrometallurgy 196:105438. https://doi.org/10.1016/S0956-053X(00)00107-0
Reid S, Tam J, Yang MF et al (2017) Technospheric mining of rare earth elements from bauxite residue (Red Mud): process optimization, kinetic investigation, and microwave pretreatment. Sci Rep 7:15252. https://doi.org/10.1038/s41598-017-15457-8
Samal S (2021) Utilization of red mud as a source for metal ions—a review. Materials 14(9):2211. https://doi.org/10.3390/ma14092211
Samouhos M, Taxiarchou M, Pilatos G et al (2017) Controlled reduction of red mud by H2 followed by magnetic separation. Miner Eng 105:36–43. https://doi.org/10.1016/j.mineng.2017.01.004
Tanvar H, Mishra B (2021) Hydrometallurgical recycling of red mud to produce materials for inustrial applications: alkali separation, iron leaching and extraction. Metall Mater Trans B 52(5):3543–3557. https://doi.org/10.1007/s11663-021-02285-5
Valeev D, Zinoveev D, Kondratiev A et al (2020) Reductive smelting of neutralized red mud for iron recovery and produced pig iron for heat-resistant castings. Metals 10(1):32. https://doi.org/10.3390/met10010032
Viswanathiah MN, Tareen JAK, Krishnamurthy KV (1980) Low temperature hydrothermal synthesis of magnetite. J Cryst Growth 49(1):189–192. https://doi.org/10.1016/0022-0248(80)90081-0
Wang S, Jin H, Deng Y et al (2020) Comprehensive utilization status of red mud in China: a critical review. J Clean Prod 289(11):125136. https://doi.org/10.1016/j.jclepro.2020.125136
Watanabe Y, Takemura S, Kashiwaya Y et al (1996) Reduction of haematite to magnetite induced by hydrogen ion implantation. J Phys D 29:8–13
Xue SG, Li M, Jiang J et al (2019) Phosphogypsum stabilization of bauxite residue: conversion of its alkaline characteristics. J Environ Sci 77:1–10. https://doi.org/10.1016/j.jes.2018.05.016
Xue SG, Liu Z, Fan JR et al (2022) Insights into variations on dissolved organic matter of bauxite residue during soil-formation processes following 2-year column simulation. Environ Pollut 292(A):118326. https://doi.org/10.1016/j.envpol.2021.118326
Zhang YY, Wei L, Qi YH et al (2016) Recovery of iron and calcium aluminate slag from high-ferrous bauxite by high-temperature reduction and smelting process. Int J Miner Metall Mater 23(8):10. https://doi.org/10.1007/s12613-016-1303-3
Zhong L, Zhang YF, Zhang Y (2009) Extraction of alumina and sodium oxide from red mud by a mild hydro-chemical process. J Hazard Mater 172(2–3):1629–1634. https://doi.org/10.1016/j.jhazmat.2009.08.036
Zhu H, Yang D, Zhu L (2007) Hydrothermal growth and characterization of magnetite (Fe3O4) thin films. Surf Coat Technol 201(12):5870–5874. https://doi.org/10.1016/j.surfcoat.2006.10.037
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant Nos. 52104353).
Funding
Funding was provided by National Natural Science Foundation of China (Grant Number 52104353).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, G., Wang, Y., Qi, T. et al. Cleaning Disposal of High-Iron Bauxite Residue Using Hydrothermal Hydrogen Reduction. Bull Environ Contam Toxicol 109, 163–168 (2022). https://doi.org/10.1007/s00128-022-03516-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-022-03516-4