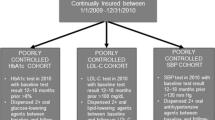
Abstract
Aims/hypothesis
Clinical trial participation should theoretically reduce barriers to care by ensuring medication and healthcare access. We aimed to evaluate disparities in achieving diabetes treatment targets by race/ethnicity and educational attainment within the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial (ClinicalTrials.gov NCT00000620).
Methods
The ACCORD trial included three interventions of varying participant burden: glycaemic (high burden), blood pressure (medium burden) and triglyceride-lowering (low burden). We examined adjusted odds ratios (aORs) for achievement of glycaemic targets, blood pressure targets and a ≥25% reduction in triglyceride levels (a proxy for adherence to fenofibrate therapy) in the first year, and for hypoglycaemia requiring medical assistance at any time, by treatment arm, race/ethnicity and educational attainment using multivariable models adjusted for demographics and clinical characteristics. We explored whether disparities in glycaemic goal achievement were mediated by hypoglycaemia, medication use, change in BMI or number of study visits attended.
Results
Compared with White participants, participants who identified as Black, Hispanic and Other race/ethnicity were less likely to achieve glycaemic targets (aOR [95% CI]) 0.63 [0.55,0.71], 0.73 [0.61, 0.88], 0.82 [0.71, 0.96], respectively); Black participants but not Hispanic and Other race/ethnicity participants were less likely to achieve blood pressure targets (aOR [95% CI] 0.77 [0.65, 0.90], 1.01 [0.78, 1.32], 1.01 [0.81, 1.26], respectively); and Black, Hispanic and Other race/ethnicity participants were equally or more likely to achieve triglyceride reduction (aOR [95% CI] 1.77 [1.38, 2.28], 1.34 [0.98, 1.84], 1.43 [1.10, 1.85], respectively). Differences in goal achievement by educational attainment were generally not significant after adjusting for baseline characteristics. Rates of hypoglycaemia requiring medical assistance were highest among Black individuals and those with lower educational attainment. Associations between race/ethnicity and glycaemic control were partially mediated by differences in insulin dosing and oral medication use.
Conclusions/interpretation
Racially/ethnically minoritised participants in the ACCORD trial were less likely to achieve high-burden (glycaemic) treatment goals but were generally similarly likely to achieve goals of less intensive interventions. Differences in glycaemic treatment goal achievement were partially mediated by differences in medication use but not mediated by hypoglycaemia, change in BMI or study visit attendance.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.

Introduction
The outpatient management of diabetes has centred on achievement of the ‘ABCs’—glycaemic (HbA1c), blood pressure and cholesterol targets—to reduce the risk of microvascular and macrovascular complications. Disparities are widely documented in usual care for type 2 diabetes, with higher rates of diabetes incidence and complications and lower rates of treatment target achievement and novel therapy use among racial/ethnically minoritised and socioeconomically disadvantaged individuals [1,2,3,4,5,6,7,8,9,10,11].
Although disparities in clinical trial enrolment are well known [12,13,14,15], in theory, some disparities should be mitigated within trials in which medications are provided free of charge to all participants, with equivalent access to healthcare personnel dedicated to ensuring that all participants receive optimal care and adhere to trial protocols. It is not known whether differences in the many other contextual factors that contribute to health outcomes, including socioeconomic status, environmental injustice and exposure to systemic and interpersonal racism, lead to persistent disparities in outcomes for the condition under study even within a trial. Thus, evaluating disparities within a clinical trial provides an experimental model in which access to healthcare and treatment for the condition under study are more equal than in usual care, reducing access-related disparities in outcomes, thereby allowing for evaluation of the effects of other social determinants of health in relative isolation.
In addition, clinical trials can be helpful to probe the limits of clinical care. Participants enrolled in trials are often younger, healthier and possibly more health conscious than the overall population [16,17,18,19], and the care they receive within a trial is generally similar or superior to care delivered outside the trial setting, with frequent check-ins, proactive follow-up associated with higher rates of medication adherence, the use of guideline-directed recommendations and relatively easy access to clinicians [20,21,22]. Therefore, the outcomes achieved within some clinical trials may be viewed as a treatment ceiling, that is, the outcomes achievable under the best and most supportive circumstances.
The Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial (ClinicalTrials.gov NCT00000620) may be used as a model to approximate optimal clinical care. ACCORD was a National Heart, Lung, and Blood Institute (NHLBI)-sponsored, large multicentre trial that enrolled participants from 2001 to 2005 and assessed the effect of intensive vs standard glycaemic, blood pressure and triglyceride targets on cardiovascular disease outcomes in adults with type 2 diabetes. The trial design was factorial: all participants were assigned to either intensive (HbA1c <42.1 mmol/mol [6.0%]) or standard (HbA1c 53.0–62.8 mmol/mol [7.0–7.9%]) glycaemic goals, slightly less than half were assigned to intensive (systolic blood pressure [SBP] <120mmHg) or standard (SBP <140mmHg) blood pressure targets, and the remainder were assigned to use of fenofibrate or placebo in addition to background statin therapy. Evidence-based treatment algorithms and medications were provided through the trial, and study investigators were encouraged to use any clinically available treatment to achieve trial targets. Because ACCORD enrolled a diverse population with type 2 diabetes, provided very frequent interactions with clinical research teams (3–12 visits annually) and compared treatment targets rather than treatment methodologies, it provides an ideal model in which to study (1) disparities and (2) the limits of achievable glycaemic and blood pressure control.
Here, we report a stratified analysis of ACCORD in which we tested for differences in rates of arm-specific goal achievement by race/ethnicity and by educational attainment. We hypothesised that disparities would persist even within the clinical trial setting and would be greatest for highly intensive interventions (e.g. the glycaemic interventions) and least for minimally intensive interventions (e.g. use of fenofibrate or placebo). Additionally, we explored the inability of many participants to achieve treatment goals even within a clinical trial and the factors that may prevent goal achievement.
Methods
Data source and participants
The ACCORD trial recruited 10,251 adults with uncontrolled type 2 diabetes who were either between the ages of 40 and 79 years with prevalent CVD or between the ages of 55 and 79 years with risk factors for CVD; who did not have a BMI > 45 kg/m2, advanced kidney disease or other serious illness or who had not experienced any recent serious hypoglycaemic events; and who resided in either the USA or Canada. The recruited study population was largely representative of the older adult US population with diabetes at the time of recruitment, with the exceptions of over-representation of men (due to recruitment from VA sites), under-representation of individuals without a high school degree and over-representation of individuals with a college degree [23]. These participants were then randomly assigned to intensive or standard glycaemic treatment goals; 4733 participants were also assigned to the blood pressure trial and 5518 were assigned to the lipid trial. For this analysis, we used deidentified datasets obtained from the NHLBI Biologic Specimen and Data Repository Information Coordinating Center (BioLINCC). The study was deemed exempt from review by the Mass General Brigham Institutional Review Board (protocol no. 2019P003795).
The treatment protocols used in the ACCORD trial have been described previously [24,25,26,27]. In brief, individuals in the intensive glycaemic control arm initiated at least two classes of medications, with dose intensification or addition of a new medication class monthly, aiming for HbA1c <42.1 mmol/mol (6.0%), with de-escalation only in the setting of adverse effects. After reaching the goal, visits decreased to once every 2 months with interim phone calls between visits. Participants in the intensive glycaemic arm were instructed to monitor their blood glucose levels twice daily if at goal and four times daily if not at goal, with self-titration protocols employed to allow variable mealtime dosing and overall dose adjustment every 4 days. Individuals in the standard glycaemic control arm received physician-led treatment escalation or de-escalation to maintain HbA1c at approximately 53.0–62.8 mmol/mol (7.0–7.9%), with visits every 2–4 months. Participants in this arm were instructed to monitor their blood glucose levels approximately once per day, with self-titration used only to prevent hypoglycaemia. Participants in the blood pressure trial were seen every 2–4 months, depending on treatment arm, and received medication titration to achieve blood pressure goals, de-escalating only in the setting of adverse effects. Participants in the lipid trial received either fenofibrate or placebo, added to a background of universal statin use. Medications for glycaemic, blood pressure and lipid management and glucose monitoring supplies were provided by the trial, and study teams actively managed these conditions throughout the course of the trial at no cost to the participants.
In this analysis, we included all ACCORD participants for whom data on both race/ethnicity and educational attainment were available (n=10,244). All participants were included in the analyses of glycaemic goal achievement, participants in the blood pressure trial were considered in the analyses of SBP goal achievement, and only those randomly assigned to receive fenofibrate were included in the analyses of the lipid trial (as there was no proxy measure for medication adherence in those randomly assigned to the placebo group in the lipid trial).
Exposures, outcomes and covariates
The primary exposures in this analysis were (1) race/ethnicity, based on self-report and categorised as White, Black, Hispanic or Other in trial documentation, and (2) highest level of educational attainment, categorised as less than high school, high school diploma, some college education or a college degree. ‘Other’ race or ethnicity included individuals who self-reported as American Indian/Alaska Native, First Nation (Aboriginal Canadian), Asian, Native Hawaiian/Other, Pacific Islander, French Canadian or Other. These categories were combined prior to inclusion of the dataset in the NHLBI BioLINCC data repository.
The primary outcomes of this analysis were achievement of the respective goals of each intervention 12 months following trial entry. The 12 month time point was chosen to allow time for titration of glycaemic and blood pressure medications, because both the standard and the intensive arms had reached glycaemic plateaus at this time point, and because the retention rate at this time was high, minimising bias related to differential follow-up by race/ethnicity [25]. For the glycaemic intervention, achievement was considered having an HbA1c level ≤42.1 mmol/mol (6.0%) in the intensive arm or ≤62.8 mmol/mol (7.9%) in the standard arm. For the blood pressure intervention, achievement was considered having an SBP <120 mmHg in the intensive arm or <140 mmHg in the standard arm. For the lipid intervention, achievement was considered as having a ≥25% reduction in triglyceride levels, a proxy measure of fenofibrate adherence based on anticipated changes in lipid profile with fenofibrate use [28,29,30]. Absolute changes in HbA1c, SBP and triglyceride levels were examined as secondary outcomes. Finally, a safety measure, time to first hypoglycaemic event requiring medical assistance, was analysed as a secondary outcome.
Key covariates in the glycaemic, blood pressure and lipid analyses included age, self-reported sex, years since diabetes diagnosis, baseline laboratory or vital measurements (HbA1c, SBP or triglycerides, respectively), baseline medication use (insulin use, number of blood pressure medications and statin use, respectively) and pre-existing cardiovascular disease.
Statistical analysis
Baseline characteristics are reported as means and SDs for continuous measurements and as number and proportion for categorical measurements. Differences between self-reported race/ethnicity and educational attainment groups were assessed using ANOVA for continuous variables and χ2 tests for categorical variables.
Because of high rates of missing outcome data at 12 months (14.5% for HbA1c, 7.4% for SBP and 6.3% for triglycerides) and differential missingness by race/ethnicity and by educational attainment (electronic supplementary material [ESM] Table 1), treatment goal achievement was evaluated following several different assumptions about missing data. In the primary analysis, a simple imputation approach was used: if HbA1c (or SBP) measurement at 12 months was missing, the mean of the 8 and 16 month measurements was imputed as the 12 month value. As triglycerides were only checked annually, if the value was missing at 12 months, the 24 month value was used as the 12 month value. Individuals whose data could not be imputed (<6% for each outcome) were excluded.
We assessed unadjusted differences in rates of treatment goal achievement by treatment arm and by (1) race/ethnicity and (2) educational attainment using χ2 tests, and changes in HbA1c, SBP and triglycerides using ANOVA. We next examined the association between exposures and binary treatment goal achievement in multivariable logistic regression models, initially in (1) ‘base models’ adjusted for age, sex and treatment arm, and then in (2) ‘full models’ additionally adjusted for diabetes duration, outcome-specific disease control (baseline HbA1c, SBP or triglyceride levels, respectively), outcome-specific medication use (baseline use of insulin, number of blood pressure medications and use of statins, respectively) and pre-existing cardiovascular disease, (3) full models additionally adjusted for BMI, (4) interaction models including all covariates adjusted for in full models, as well as interactions between exposure (race/ethnicity or educational attainment, respectively) and trial arm and (5) a single combined model including both race/ethnicity and educational attainment and all covariates included in the full models. Finally, we examined whether the following variables mediated the association between race/ethnicity and glycaemic goal achievement using structural equation modelling (ESM Fig. 1) [31]: (1) number of documented hypoglycaemic events in the first year, (2) change in total daily dose of insulin in the first year, (3) total number of non-insulin diabetes medications used at the end of the first year, (4) change in BMI during the first year and (5) number of visits attended during the first year.
Five sensitivity analyses, using different methods to account for missing outcome data, were performed to examine the association of the exposures with the outcomes under different assumptions. These sensitivity analyses included outcome definitions based on (1) goal achievement at any time in the first year; (2) inverse probability weighting to adjust for factors potentially related to missingness [32]; and simple imputations testing the assumption that data were missing not at random, including (3) the primary analysis imputation with the assumption that all whose data could not be imputed did not meet goals and (4 and 5) ‘worst case’ and ‘best case’ scenarios assuming that all individuals with missing data either did not or did meet treatment goals, respectively (see ESM Methods for details) [33].
Finally, we examined the association between exposures and time to a first hypoglycaemic event requiring medical assistance using Cox proportional hazards models adjusted for age, sex, treatment arm, diabetes duration, baseline insulin use, baseline HbA1c and pre-existing cardiovascular disease.
All analyses were performed using R version 4.0.2 (R Core Team; Vienna, Austria).
Results
Baseline characteristics
The baseline characteristics of ACCORD participants have been previously reported [25]. In brief, of the 10,244 participants included in this analysis, the mean age was 62.8 years, 39% were female, 62.4% reported White race/ethnicity (vs 19.0%, 7.2% and 11.4% reporting Black, Hispanic and Other, respectively) and 26.0% had a college degree (vs 14.8%, 26.4% and 32.8% having less than a high school diploma, a high school diploma or some college education, respectively). There were differences in baseline characteristics, including in demographics, health conditions, diabetes history, medication use and disease control (HbA1c, SBP and triglyceride levels), across strata of race/ethnicity and educational attainment (Table 1, ESM Table 2). Rates of missing data for key outcomes were generally lowest among White participants and those with a college degree (ESM Table 1).
Achievement of glycaemic, blood pressure and lipid goals by race/ethnicity
Within the first year following random assignment, 52% of White participants compared with 39%, 43% and 48% of Black, Hispanic and Other race or ethnicity participants, respectively, achieved the HbA1c goal set by their randomisation arm (Fig. 1a, ESM Table 3), although absolute decreases in HbA1c, SBP and triglycerides were similar between groups (ESM Table 4). ORs of goal achievement between groups were similar across sensitivity analyses examining different methods to account for missing data (ESM Fig. 2a). Similar racial/ethnic disparities existed in both the intensive and standard treatment arms (ESM Table 5, ESM Fig. 3a).
In adjusted analyses, self-reported Black, Hispanic and Other race/ethnicity was associated with lower odds of glycaemic goal achievement (OR [95% CI] 0.63 [0.55, 0.71], 0.73 [0.61, 0.88], 0.82 [0.71, 0.96], respectively, compared with self-reported White race/ethnicity; Table 2, Fig. 2, ESM Table 6). A weak negative interaction was seen between arm and Black race/ethnicity, suggesting even lower odds of goal achievement in Black participants in the intensive glycaemic arm; no other evidence of arm-by-race/ethnicity interactions were observed (ESM Table 7). Adjusting for change in BMI or for educational attainment did not significantly alter the association between race/ethnicity and goal achievement (ESM Tables 8, 9).
Regarding SBP goals, at 12 months, 69% of White participants compared with 61%, 69% and 70% of Black, Hispanic and Other race/ethnicity participants, respectively, achieved their arm-specific blood pressure goals (Fig. 1a, ESM Table 3). ORs for goal achievement between groups were similar across sensitivity analyses (ESM Fig. 2b), and similar disparities existed in both the intensive and standard treatment arms (ESM Fig. 3b). In adjusted analyses, only Black race/ethnicity was associated with lower rates of goal achievement (OR [95% CI] 0.77 [0.65, 0.90]; Table 2, Fig. 2, ESM Table 6), with similar results in analyses adjusted for change in BMI or educational attainment (ESM Table 8, 9).
Finally, regarding lowering of triglyceride levels at 12 months, rates of ≥25% reduction in triglyceride levels were 46% among White participants compared with 50%, 50% and 54% among Black, Hispanic and Other race/ethnicity participants, respectively (Fig. 1a, ESM Table 3). In adjusted analyses, rates of achieving this level of triglyceride reduction were higher among Black (OR [95% CI] 1.77 [1.38, 2.28]) and Other (OR [95% CI] 1.43 [1.10, 1.85]) race/ethnicity individuals than among White individuals (Table 2, Fig. 2, ESM Table 6), with similar results in analyses adjusted for change in BMI or educational attainment (ESM Tables 8, 9).
We next tested the degree to which disparities in glycaemic control were mediated by hypoglycaemia, insulin dosing, non-insulin diabetes medication use, change in BMI and number of trial visits attended (see Methods). Differences in insulin dosing and non-insulin diabetes medication use mediated 9.9% and 6.9% of this association among Black participants, 10.9% and 0% of this association among Hispanic participants, and 22.0% and 13.1% of this association among Other race/ethnicity participants, respectively. Each of the other factors mediated <4% of the total effect of Black, Hispanic or Other race/ethnicity on glycaemic goal achievement, suggesting that this association was not significantly mediated by these factors (ESM Tables 10, 11).
Achievement of glycaemic, blood pressure and lipid goals by educational attainment
Within the first year following randomisation, 51% of college graduates compared with 45%, 48% and 47% of those with less than a high school diploma, a high school diploma or some college education, respectively, achieved the HbA1c goal set by their randomisation arm (Fig. 1b, ESM Table 3), with no significant differences in fully adjusted analyses (Table 3, ESM Table 12).
Regarding SBP goals, at 12 months, 71% of college graduates compared with 66%, 65% and 66% of those with less than a high school diploma, a high school diploma or some college education, respectively, achieved their arm-specific blood pressure goals (Fig. 1b, ESM Table 3). In adjusted analyses, having a high school diploma (OR [95% CI] 0.77 [0.64, 0.92]) or some college education (OR [95% CI] 0.78 [0.66, 0.93]) was associated with lower rates of goal achievement (Table 3, ESM Table 12).
Finally, regarding lowering of triglyceride levels at 12 months, rates of ≥25% reduction in triglycerides were 48% among college graduates compared with 44%, 48% and 50% those with less than a high school diploma, a high school diploma or some college education, respectively (Fig. 1b, ESM Table 3), with no significant differences in fully adjusted analyses (Table 3, ESM Table 12).
For all outcomes, no consistent arm-by-educational attainment interactions were observed (ESM Table 13), and no difference was seen in analyses adjusted for change in BMI (ESM Table 14).
Hypoglycaemia requiring medical assistance by race/ethnicity and educational attainment
Absolute rates of hypoglycaemia requiring medical assistance were highest among Black participants (12.3%) compared with Hispanic (7.7%), Other race/ethnicity (5.1%) and White (7.4%) participants (Fig. 3a, ESM Table 3, ESM Fig. 4). In adjusted time-to-event analyses, Black race/ethnicity (HR [95% CI] 1.61 [1.37, 1.89]) was associated with a higher hazard of hypoglycaemia requiring medical assistance than White race/ethnicity (ESM Table 15).
With regard to educational attainment, absolute rates of hypoglycaemia requiring medical assistance were highest among those with less than a high school education (11.2%), followed by those with a high school diploma (9.4%), some college education (7.3%) and a college degree (6.1%; Fig. 3b, ESM Table 3). In adjusted analyses, having less than a high school diploma (HR [95% CI] 1.57 [1.26, 1.96]) or a high school diploma (HR [95% CI] 1.41 [1.15, 1.72]) was associated with a higher hazard of hypoglycaemia requiring medical assistance than having a college degree (ESM Table 15).
Discussion
In this secondary analysis of the ACCORD trial, we observed disparities in achievement of diabetes treatment targets and incidence of hypoglycaemia requiring medical assistance by race/ethnicity and by educational attainment. In particular, we found that, compared with White participants, Black, Hispanic and Other race/ethnicity participants were less likely to achieve glycaemic targets; Black participants but not Hispanic and Other race/ethnicity participants were less likely to achieve blood pressure targets; and Black, Hispanic and Other race/ethnicity participants were equally or more likely to achieve a reduction in triglyceride levels, after adjustment for likely confounders. These findings have important implications for the generalisability of clinical trial results, especially in clinical trials with a high participant burden or difficult-to-implement interventions, and for understanding the complex causes of health disparities, which persist even in highly supportive clinical settings. Additionally, the low overall rates of goal achievement despite excellent access to clinicians and medications speak to the limits of clinical management even in a resource-abundant setting.
Variable degrees of racial/ethnic and socioeconomic disparities in treatment effects and treatment goal achievement have been described in other type 2 diabetes clinical trials, primarily in trials of intensive lifestyle interventions, including the Diabetes Prevention Program [34], Look AHEAD [35] and REAL HEALTH-Diabetes [36]. As a factorial trial testing three different hypotheses, ACCORD provides a unique opportunity to examine the effect of race/ethnicity and socioeconomic status under different intervention conditions, all approximating clinical care yet with varying degrees of participant burden, within the same population and care model. We found that racial/ethnic disparities were largest for the high-burden glycaemic intervention (requiring self-monitoring, frequent medication escalation and complex self-titration regimens) but small or even non-existent for the less intensive blood pressure intervention (requiring monitoring by a clinician and occasional medication titration) and the relatively low-burden lipid intervention (requiring adherence to a single medication, fenofibrate).
One possible explanation for lower rates of glycaemic goal achievement among Black participants is that mean HbA1c levels may be 4.3 mmol/mol (0.4%) higher for the same mean glucose levels among Black or African American individuals [37,38,39,40,41,42]. Black participants did have modestly higher HbA1c levels at baseline in the ACCORD study. HbA1c levels that modestly overestimate mean glucose values, a phenomenon that was not recognised at the time the ACCORD trial was conducted, may have contributed to the higher rates of hypoglycaemia among Black participants despite lower rates of goal achievement. However, disparities in goal achievement persisted in multivariable models adjusted for baseline HbA1c, and lower rates of goal achievement were also seen among Hispanic and Other race/ethnicity participants, so this biological association is unlikely to fully explain the key findings of our analysis and additional aetiologies should be considered.
Importantly, ACCORD was a trial not of a specific treatment but of treatment target achievement, employing any clinically available medications or treatment methodologies as necessary to achieve treatment targets and stopping only in the face of adverse safety outcomes. This use of a wide range of treatments replicates the approach taken in usual care. Unlike usual care, however, in ACCORD, treatment and medications were provided free of charge, as was intensive support from and access to specialist care, supplemented by proactive outreach that is often employed to retain participants in trials. Moreover, by protocol design, researchers and participants pursued treatment goals more aggressively than in clinical care, where lack of treatment intensification is often attributed to clinical inertia. Thus, disparities seen within this study reflect not a lack of access to medications, healthcare or support but rather a difference either in implementation of trial protocols or in factors that limited implementation of the protocol (e.g. hypoglycaemia, differential follow-up, baseline differences in health literacy or education regarding diabetes self-management).
This clinical trial model also informs our understanding of the impact, or lack thereof, of other commonly cited causes of health disparities, including differences in healthcare access, behaviours such as adherence, and socioeconomic status and other social determinants of health, on the differences observed in this analysis. First, as all participants were enrolled in a trial with similar goals and treatment protocols, factors such as healthcare and medication access are likely to have had a lower impact on the observed disparities compared with usual care. Although up to 22% of the difference in glycaemic goal achievement appeared to be mediated by differences in insulin or non-insulin medication use, the majority of this difference was not explained by any mediating factors tested. Differences in participant behaviours are plausible; however, the similar number of visits attended, lack of mediation by number of visits attended and higher likelihood of triglyceride reduction with fenofibrate among Black, Hispanic and Other race/ethnicity participants suggest that protocol adherence, at least, was comparable if not superior within these groups compared with White participants. Finally, social determinants of health that are associated with race/ethnicity, including socioeconomic status, environmental differences and exposure to racism, may have contributed to differences in the ability to meet treatment targets. These include baseline differences in health literacy and diabetes-related education, as well as the absence of specifically culturally tailored education within ACCORD, which could not be assessed in this analysis. Importantly, adjusting for educational attainment, a proxy measure of socioeconomic status, or race/ethnicity-by-educational attainment interactions did not alter the primary findings (ESM Tables 7, 9), suggesting that socioeconomic status alone is unlikely to be the primary contributor to the observed racial/ethnic disparities. Race and ethnicity are social constructs that nonetheless correlate with myriad unmeasurable adverse circumstances, such as barriers related to the environment and exposure to racism and racist practices; these associated factors remain possible explanations. Finally, although study teams were following a protocol, the intensification of therapy even within the trial may have differed by race and ethnicity, either because of unconscious bias or for appropriate reasons that are correlated with race and ethnicity, as supported by the modest degree of mediation of the association between race/ethnicity and glycaemic goal achievement by insulin dose and non-insulin medication use.
Finally, despite the intensive interventions and flexible, physician-led treatment strategies (targeting a level of glycaemic control but not following a specific treatment regimen) that were employed in this trial, a significant proportion of participants remained unable to achieve treatment targets, particularly glycaemic targets. In this setting, with participants committed to trial participation, very frequent clinician visits and medications provided at no charge, we may expect that many potential barriers to optimal treatment outcomes are overcome. However, the proportions of individuals unable to meet glycaemic targets were 72–85% and 25–36% in the intensive and standard arms, respectively, and the proportions unable to meet blood pressure targets were 37–45% and 24–33% in the intensive and standard arms, respectively, depending on race/ethnicity (ESM Table 5), similar to at-target rates reported in recent studies of the general population [8,9,10]. Recognition of these limits of clinical care within the ACCORD trial can inform clinical goals in real-world settings in which fewer resources are available.
This analysis has numerous strengths including the large sample size from a frequently assessed population exposed to clinically representative therapies and follow-up. However, it must be interpreted in light of its study design. As a post hoc, observational analysis of a trial that was not designed to assess disparities, described associations are exploratory. Analyses were not adjusted for multiple testing; with 48 subgroup–outcome comparisons, two to three statistically significant results would be expected on the basis of chance alone [43]. There were moderate levels of missing data related to the primary outcome, leading to use of imputation of the primary outcome that may have introduced bias; however, multiple sensitivity analyses were performed using different assumptions for the missing data, including the possibility that data are missing not at random, and results were unchanged in these analyses.
In conclusion, we found that ACCORD participants who identified as Black, Hispanic or Other race/ethnicity were less likely than White participants to achieve the glycaemic goals of the trial (and experienced higher rates of hypoglycaemia); Black participants were less likely than White participants to achieve blood pressures goals; but Black, Hispanic and Other race/ethnicity participants were equally or more likely to achieve a significant reduction in triglyceride levels compared with White participants. Disparities persisted after adjusting for baseline characteristics, disease duration and control, and educational attainment and were not mediated by side effects or adherence. This suggests that disparities exist even within clinical trials but that these disparities may depend on the intensity, or degree of participant burden, of the intervention studied. The barriers to achieving high-intensity treatment goals may be greater for some populations, even in clinical trials and especially in real-world settings.
Abbreviations
- ACCORD:
-
Action to Control Cardiovascular Risk in Diabetes
- BioLINCC:
-
Biologic Specimen and Data Repository Information Coordinating Center
- NHLBI:
-
National Heart, Lung, and Blood Institute
- SBP:
-
Systolic blood pressure
References
Centers for Disease Control and Prevention (2022) By the numbers: diabetes in America. Available from https://www.cdc.gov/diabetes/health-equity/diabetes-by-the-numbers.html. Accessed 6 Nov 2022
Heidemann DL, Joseph NA, Kuchipudi A, Perkins DW, Drake S (2016) Racial and economic disparities in diabetes in a large primary care patient population. Ethn Dis 26(1):85–90. https://doi.org/10.18865/ed.26.1.85
Rawshani A, Svensson AM, Zethelius B, Eliasson B, Rosengren A, Gudbjörnsdottir S (2016) Association between socioeconomic status and mortality, cardiovascular disease, and cancer in patients with type 2 diabetes. JAMA Intern Med 176(8):1146–1154. https://doi.org/10.1001/jamainternmed.2016.2940
Perna L, Thien-Seitz U, Ladwig KH, Meisinger C, Mielck A (2010) Socio-economic differences in life expectancy among persons with diabetes mellitus or myocardial infarction: results from the German MONICA/KORA study. BMC Public Health 10(1):135. https://doi.org/10.1186/1471-2458-10-135
Cromer SJ, Lauffenburger JC, Levin R, Patorno E (2023) Deficits and disparities in early uptake of glucagon-like peptide 1 receptor agonists and SGLT2i among Medicare-insured adults following a new diagnosis of cardiovascular disease or heart failure. Diabetes Care 46(1):65–74. https://doi.org/10.2337/dc22-0383
Eberly LA, Yang L, Eneanya ND et al (2021) Association of race/ethnicity, gender, and socioeconomic status with sodium-glucose cotransporter 2 inhibitor use among patients with diabetes in the US. JAMA Network Open 4(4):e216139–e216139. https://doi.org/10.1001/jamanetworkopen.2021.6139
Eberly LA, Yang L, Essien UR et al (2021) Racial, ethnic, and socioeconomic inequities in glucagon-like peptide-1 receptor agonist use among patients with diabetes in the US. JAMA Health Forum 2(12):e214182. https://doi.org/10.1001/jamahealthforum.2021.4182
Venkatraman S, Echouffo-Tcheugui JB, Selvin E, Fang M (2022) Trends and disparities in glycemic control and severe hyperglycemia among US Adults with diabetes using insulin, 1988–2020. JAMA Network Open 5(12):e2247656. https://doi.org/10.1001/jamanetworkopen.2022.47656
Cromer SJ, Wexler DJ, Kazemian P (2021) Correlates of analog vs human basal insulin use among individuals with type 2 diabetes: a cross-sectional study. Diabetes Res Clin Pract 175:108825. https://doi.org/10.1016/j.diabres.2021.108825
Kazemian P, Shebl FM, McCann N, Walensky RP, Wexler DJ (2019) Evaluation of the cascade of diabetes care in the United States, 2005–2016. JAMA Intern Med 179(10):1376–1385. https://doi.org/10.1001/jamainternmed.2019.2396
Bundy JD, Mills KT, He H et al (2023) Social determinants of health and premature death among adults in the USA from 1999 to 2018: a national cohort study. Lancet Public Health 8(6):e422-431. https://doi.org/10.1016/S2468-2667(23)00081-6
Akturk HK, Agarwal S, Hoffecker L, Shah VN (2021) Inequity in racial-ethnic representation in randomized controlled trials of diabetes technologies in type 1 diabetes: critical need for new standards. Diabetes Care 44(6):e121-123. https://doi.org/10.2337/dc20-3063
Bowe T, Salabati M, Soares RR et al (2022) Racial, ethnic, and gender disparities in diabetic macular edema clinical trials. Ophthalmol Retina 6(6):531–533. https://doi.org/10.1016/j.oret.2022.01.018
Clemens KK, Woodward M, Neal B, Zinman B (2020) Sex Disparities in cardiovascular outcome trials of populations with diabetes: a systematic review and meta-analysis. Diabetes Care 43(5):1157–1163. https://doi.org/10.2337/dc19-2257
Avgerinos I, Karagiannis T, Liakos A, Tsapas A, Bekiari E (2023) Racial, ethnic and sex disparities among participants in cardiovascular outcomes trials in type 2 diabetes: a systematic review and descriptive analysis. Diabetes Obes Metab 25(2):618–622. https://doi.org/10.1111/dom.14900
Mettlin C, Cummings KM, Walsh D (1985) Risk factor and behavioral correlates of willingness to participate in cancer prevention trials. Nutr Cancer 7(4):189–198. https://doi.org/10.1080/01635588509513855
Ludmir EB, Mainwaring W, Lin TA et al (2019) Factors associated with age disparities among cancer clinical trial participants. JAMA Oncol 5(12):1769–1773. https://doi.org/10.1001/jamaoncol.2019.2055
Unger JM, Hershman DL, Fleury ME, Vaidya R (2019) Association of patient comorbid conditions with cancer clinical trial participation. JAMA Oncol 5(3):326–333. https://doi.org/10.1001/jamaoncol.2018.5953
Bray GA (2006) Baseline characteristics of the randomised cohort from the Look AHEAD (Action for Health in Diabetes) study. Diabetes Vasc Dis Res 3(3):202–215. https://doi.org/10.3132/dvdr.2006.031
van Onzenoort HAW, Menger FE, Neef C et al (2011) Participation in a clinical trial enhances adherence and persistence to treatment. Hypertension 58(4):573–578. https://doi.org/10.1161/HYPERTENSIONAHA.111.171074
Hallstrom A, Friedman L, Denes P et al (2003) Do arrhythmia patients improve survival by participating in randomized clinical trials? Observations from the Cardiac Arrhythmia Suppression Trial (CAST)and the Antiarrhythmics Versus Implantable Defibrillators Trial (AVID). Control Clin Trials 24(3):341–352. https://doi.org/10.1016/S0197-2456(03)00002-3
Braunholtz DA, Edwards SJ, Lilford RJ (2001) Are randomized clinical trials good for us (in the short term)? Evidence for a “trial effect.” J Clin Epidemiol 54(3):217–224. https://doi.org/10.1016/S0895-4356(00)00305-X
Fang M (2018) Trends in the prevalence of diabetes among U.S. Adults: 1999-2016. Am J Prev Med 55(4):497–505. https://doi.org/10.1016/j.amepre.2018.05.018
Gerstein HC, Riddle MC, Kendall DM et al (2007) Glycemia treatment strategies in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial. Am J Cardiol 99(12 Suppl.):S34-43. https://doi.org/10.1016/j.amjcard.2007.03.004
ACCORD Study Group (2008) Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 358(24):2545–2559. https://doi.org/10.1056/NEJMoa0802743
Cushman WC, Grimm RH, Cutler JA et al (2007) Rationale and design for the blood pressure intervention of the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial. Am J Cardiol 99(12A):44i–55i. https://doi.org/10.1016/j.amjcard.2007.03.005
Buse JB (2007) Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial: design and methods. Am J Cardiol 99(12 Suppl.):S21-33. https://doi.org/10.1016/j.amjcard.2007.03.003
Feingold KR (2000) Triglyceride lowering drugs. In: Feingold KR, Anawalt B, Boyce A et al (eds), Endotext. MDText.com, South Dartmouth, MA. Available from http://www.ncbi.nlm.nih.gov/books/NBK425699/. Accessed 6 Dec 2022
Zimetbaum P, Frishman WH, Kahn S (1991) Effects of gemfibrozil and other fibric acid derivatives on blood lipids and lipoproteins. J Clin Pharmacol 31(1):25–37. https://doi.org/10.1002/j.1552-4604.1991.tb01883.x
Rosenson RS (2008) Fenofibrate: treatment of hyperlipidemia and beyond. Expert Rev Cardiovasc Ther 6(10):1319–1330. https://doi.org/10.1586/14779072.6.10.1319
VanderWeele TJ (2016) Mediation analysis: a practitioner’s guide. Annu Rev Public Health 37:17–32. https://doi.org/10.1146/annurev-publhealth-032315-021402
Seaman SR, White IR (2013) Review of inverse probability weighting for dealing with missing data. Stat Methods Med Res 22(3):278–295. https://doi.org/10.1177/0962280210395740
Pedersen AB, Mikkelsen EM, Cronin-Fenton D et al (2017) Missing data and multiple imputation in clinical epidemiological research. Clin Epidemiol 9:157–166. https://doi.org/10.2147/CLEP.S129785
O’Brien MJ, Whitaker RC, Yu D, Ackermann RT (2015) The comparative efficacy of lifestyle intervention and metformin by educational attainment in the Diabetes Prevention Program. Prev Med 77:125–130. https://doi.org/10.1016/j.ypmed.2015.05.017
Cromer SJ, Meigs J, Wexler DJ (2022) Racial/ethnic and socioeconomic disparities in weight outcomes, cardiovascular events, and mortality in the look AHEAD trial. Diabetes Res Clin Pract 192:110095. https://doi.org/10.1016/j.diabres.2022.110095
Berkowitz SA, Chang Y, Porneala B, Cromer SJ, Wexler DJ, Delahanty LM (2020) Does the effect of lifestyle intervention for individuals with diabetes vary by food insecurity status? A preplanned subgroup analysis of the REAL HEALTH randomized clinical trial. BMJ Open Diabetes Res Care 8(1):e001514. https://doi.org/10.1136/bmjdrc-2020-001514
Menke A, Rust KF, Savage PJ, Cowie CC (2014) Hemoglobin A1c, fasting plasma glucose, and 2-hour plasma glucose distributions in U.S. population subgroups: NHANES 2005-2010. Ann Epidemiol 24(2):83–89. https://doi.org/10.1016/j.annepidem.2013.10.008
Kirk JK, D’Agostino RB, Bell RA et al (2006) Disparities in HbA1c levels between African-American and non-Hispanic white adults with diabetes: a meta-analysis. Diabetes Care 29(9):2130–2136. https://doi.org/10.2337/dc05-1973
Selvin E (2016) Are there clinical implications of racial differences in HbA1c? A difference, to be a difference, must make a difference. Diabetes Care 39(8):1462–1467. https://doi.org/10.2337/dc16-0042
Meigs JB, Grant RW, Piccolo R et al (2014) Association of African genetic ancestry with fasting glucose and HbA1c levels in non-diabetic individuals: the Boston Area Community Health (BACH) Prediabetes Study. Diabetologia 57(9):1850–1858. https://doi.org/10.1007/s00125-014-3301-1
Bergenstal RM, Gal RL, Connor CG et al (2017) Racial differences in the relationship of glucose concentrations and hemoglobin A1c levels. Ann Intern Med 167(2):95–102. https://doi.org/10.7326/M16-2596
Hivert MF, Christophi CA, Jablonski KA et al (2019) Genetic ancestry markers and difference in A1c between African American and White in the Diabetes Prevention Program. J Clin Endocrinol Metab 104(2):328–336. https://doi.org/10.1210/jc.2018-01416
Wang R, Lagakos SW, Ware JH, Hunter DJ, Drazen JM (2007) Statistics in medicine–reporting of subgroup analyses in clinical trials. N Engl J Med 357(21):2189–2194. https://doi.org/10.1056/NEJMsr077003
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Acknowledgements
This manuscript was prepared using Action to Control Cardiovascular Risk in Diabetes (ACCORD) data obtained from the NHLBI BioLINCC and does not necessarily reflect the opinions or views of ACCORD or the NHLBI.
Data availability
The data that support the findings of this study are not openly available for reasons of sensitivity but may be accessed from the NHLBI Biologic Specimen and Data Repository Information Coordinating Center (BioLINCC; https://biolincc.nhlbi.nih.gov/) pending institutional review board and repository approval.
Funding
SJC was supported by the National Institutes of Health (grant no. F32DK127545) and the American Diabetes Association (grant no. 7–21-JDFM-005). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Authors’ relationships and activities
SJC reports a close family member employed by a Johnson & Johnson company and has served on advisory boards for Alexion Pharmaceuticals. DJW reports serving on the Data Monitoring Committees for Novo Nordisk. TT declares that there are no relationships or activities that might bias, or be perceived to bias, their work.
Contribution statement
The study was conceived by SJC and DJW. The analysis was performed by SJC and TT. All authors contributed substantially to the design and interpretation of the study, critically reviewed the manuscript and provided final approval for publication. SJC is the guarantor of this work.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cromer, S.J., Thaweethai, T. & Wexler, D.J. Racial/ethnic and socioeconomic disparities in achievement of treatment goals within a clinical trial: a secondary analysis of the ACCORD trial. Diabetologia 66, 2261–2274 (2023). https://doi.org/10.1007/s00125-023-05997-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-023-05997-2