Abstract
Aims/hypothesis
After birth, the neonatal islets gradually acquire glucose-responsive insulin secretion, a process that is subjected to maternal imprinting. Although NEFA are major components of breastmilk and insulin secretagogues, their role for functional maturation of neonatal beta cells is still unclear. NEFA are the endogenous ligands of fatty acid receptor 1 (FFA1, encoded by Ffar1 in mice), a Gq-coupled receptor with stimulatory effect on insulin secretion. This study investigates the role of FFA1 in neonatal beta cell function and in the adaptation of offspring beta cells to parental high-fat feeding.
Methods
Wild-type (WT) and Ffar1−/− mice were fed high-fat (HFD) or chow diet (CD) for 8 weeks before mating, and during gestation and lactation. Blood variables, pancreas weight and insulin content were assessed in 1-, 6-, 11- and 26-day old (P1–P26) offspring. Beta cell mass and proliferation were determined in P1–P26 pancreatic tissue sections. FFA1/Gq dependence of insulin secretion was evaluated in isolated islets and INS-1E cells using pharmacological inhibitors and siRNA strategy. Transcriptome analysis was conducted in isolated islets.
Results
Blood glucose levels were higher in CD-fed Ffar1−/− P6-offspring compared with CD-fed WT P6-offspring. Accordingly, glucose-stimulated insulin secretion (GSIS) and its potentiation by palmitate were impaired in CD Ffar1−/− P6-islets. In CD WT P6-islets, insulin secretion was stimulated four- to fivefold by glucose and five- and sixfold over GSIS by palmitate and exendin-4, respectively. Although parental HFD increased blood glucose in WT P6-offspring, it did not change insulin secretion from WT P6-islets. In contrast, parental HFD abolished glucose responsiveness (i.e. GSIS) in Ffar1−/− P6-islets. Inhibition of Gq by FR900359 or YM-254890 in WT P6-islets mimicked the effect of Ffar1 deletion, i.e. suppression of GSIS and of palmitate-augmented GSIS. The blockage of Gi/o by pertussis toxin (PTX) enhanced (100-fold) GSIS in WT P6-islets and rendered Ffar1−/− P6-islets glucose responsive, suggesting constitutive activation of Gi/o. In WT P6-islets, FR900359 cancelled 90% of PTX-mediated stimulation, while in Ffar1−/− P6-islets it completely abolished PTX-elevated GSIS. The secretory defect of Ffar1−/− P6-islets did not originate from insufficient beta cells, since beta cell mass increased with the offspring’s age irrespective of genotype and diet. In spite of that, in the breastfed offspring (i.e. P1–P11) beta cell proliferation and pancreatic insulin content had a genotype- and diet-driven dynamic. Under CD, the highest proliferation rate was reached by the Ffar1−/− P6 offspring (3.95% vs 1.88% in WT P6), whose islets also showed increased mRNA levels of genes (e.g. Fos, Egr1, Jun) typically high in immature beta cells. Although parental HFD increased beta cell proliferation in both WT (4.48%) and Ffar1−/− (5.19%) P11 offspring, only the WT offspring significantly increased their pancreatic insulin content upon parental HFD (5.18 µg under CD to 16.93 µg under HFD).
Conclusions/interpretation
FFA1 promotes glucose-responsive insulin secretion and functional maturation of newborn islets and is required for adaptive offspring insulin secretion in the face of metabolic challenge, such as parental HFD.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.

Introduction
Obesity, a major risk factor for type 2 diabetes, requires enhanced insulin production and adaptive insulin hypersecretion. The failure of pancreatic beta cells to cope with this high insulin demand leads to a relative insulin deficiency, which is ultimately the cause of chronic hyperglycaemia and overt type 2 diabetes in humans [1,2,3].
Glucose-stimulated insulin secretion (GSIS), the most important functional trait of healthy beta cells, is not fully developed in newborn islets. The neonatal islets are poorly glucose responsive and their insulin secretion is largely driven by amino acids [4,5,6]. Glucose responsiveness develops gradually during early postnatal life and is imperative for adequate beta cell function in adulthood. Therefore, the ability of adult beta cells to meet metabolic demands depends on both the intrauterine and neonatal environment, i.e. maternal metabolism and breastmilk feeding [7,8,9]. The consequences of an obesogenic diet in the mother for the metabolic plasticity of beta cells in the offspring and the progeny’s risk for developing diabetes are only partly known [10,11,12]. In rodents, maternal feeding of a high-fat diet (HFD) during pregnancy and lactation resulted in an increase in body weight and beta cell mass, fasting hyperinsulinaemia, hyperglycaemia and glucose intolerance in the offspring [13,14,15]. Consistent with these findings in animal models, maternal obesity in humans is associated with an increased incidence of type 2 diabetes in the offspring [16].
Although NEFA are major components of breast milk [17] and insulin secretagogues, their role in the functional maturation of neonatal beta cells remains elusive. NEFA are endogenous agonists of free fatty acid receptor 1 (FFA1), a Gq/11 G-protein-coupled receptor (GPCR) that stimulates insulin secretion and is selectively expressed in beta cells [18,19,20]. Both Gq/11- and Gs-coupled receptors stimulate insulin secretion, whereas pertussis toxin (PTX)-sensitive Gi/o-coupled receptors inhibit it. Gs-coupled glucagon-like peptide-1 receptor (GLP1R) and FFA1 potentiate GSIS in a glucose-dependent manner, which is a therapeutic requirement for avoiding hypoglycaemia [21]. While GLP1R emerged as an effective tool for type 2 diabetes treatment, FFA1 has not yet progressed beyond clinical trials [22]. Different GPCRs may share one type of G protein, but there is experimental evidence that one receptor can recruit different G proteins. In adult beta cells, Gq is the major G protein recruited by FFA1, but FFA1 shows some degree of promiscuity for G proteins, e.g. Gs or G12/13, depending on the receptor ligand and cell system [23, 24]. Similarly, the Gs-coupled GLP1R can also couple to Gq and in this way maintain insulin secretion under hyperglycaemic conditions. In beta cells exposed to metabolic stress, Gq-mediated signalling is thus a potent stimulator of GSIS [25]. GPCRs also modulate beta cell survival and proliferation. While Gi/oPCRs reduce beta cell proliferation, GLP1R and FFA1 promote beta cell survival and thereby the maintenance of beta cell mass [26,27,28]. Distinct to adult beta cells, with their extremely low proliferation rate, neonatal beta cells are responsive to proliferative cues, and neonatal proliferation impacts on adult beta cell mass. Although a positive effect of NEFA on neonatal beta cell proliferation has been described, a role of FFA1/Gq in this is unclear [29].
The aim of the present study is to understand the role of FFA1 and Gq for proliferation and functional maturation, and specifically, the gain of glucose responsiveness of neonatal beta cells in the context of an obesogenic parental diet.
Methods
All animal experiments were approved by the local authorities (Approval No. M10-18G and M20/21M of the Regional Council Tübingen, Germany). Animal care and handling was conducted in compliance with the German law for the protection of animals used for scientific purposes and in accordance with Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines.
Mouse model and feeding
C57BL/6-RipCre-ROSAmT/mG (B6.Cg-Tg(Ins2-cre)25Mgn/J-B6.129(Cg)-Gt(ROSA)26Sortm4(ACTB-tdTomato,-EGFP)Luo/J) WT and Ffar1−/− animals were generated as described in the electronic supplementary material (ESM) Methods. The islets of these mice display an insulin secretion similar to that of WT and Ffar1−/− C57BL/6 mice, respectively (ESM Fig. 1c–e). WT and Ffar1−/− animals (4 weeks old) randomly assigned to chow (CD) or high-fat diet (HFD) were fed accordingly 8 weeks before mating (Fig. 1a). WT and Ffar1−/− breeding pairs were kept on the respective diet throughout gestation and lactation. Offspring animals (21 days old) were weaned on CD. Male and female offspring were examined at postnatal days 1, 6, 11 and 26 (P1–P26).
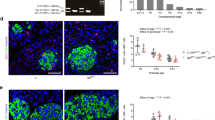
Effects of Ffar1 deletion and parental HFD feeding on blood variables and insulin secretion in P6 and P11 offspring. (a) Experimental design. (b, c) Concentrations of blood glucose in (b) P6- and (c) P11-offspring. Results are expressed as mean ± SEM for n=9–21 animals. (d–g) Insulin secretion from islets of (d, f) P6- and (e, g) P11-offspring isolated and incubated with test substances as described under Methods. Insulin secretion is expressed as mean ± SEM for the indicated number of replicates from n=3 mice per genotype and diet (independent experiments); (d, e) 11–12 replicates from n=3 independent experiments. Black lines and dots represent WT, red lines and dots Ffar1−/− mice; white- and grey-filled bars/dots represent parental CD and HFD, respectively. *p<0.05, **p<0.005, ***p<0.001 between groups as indicated. (d, e) *p<0.05 vs secretion at respective 2.8 mmol/l glucose (glucose effect); ‡p<0.05 vs Ffar1−/− genotype on the same diet and the same glucose concentration (genotype effect); §p<0.05 vs respective genotype on parental CD at the same glucose concentration (diet effect); (f, g) ¶¶¶p<0.001 vs respective 12 mmol/l glucose. d, days
Measurement of blood variables and Oil Red O staining
Blood variables and liver Oil Red O staining were assessed as described in the ESM Methods.
Offspring beta cell mass, beta cell proliferation and pancreatic insulin content
P1–P26 offspring pancreases (male and female animals) were processed as described in the ESM Methods. Pancreatic area (red fluorescence) and insulin-positive area (green fluorescence) were determined using Fiji (version 1.48d). Beta cell mass (mg) was calculated by multiplying relative beta cell area (GFP-positive/Tomato-positive) by pancreas weight (mg). Beta cell proliferation was expressed as per cent of Ki67+/GFP+ of GFP+ cells. For insulin content, P1–P26 offspring pancreases were weighed and minced in ice-cold acid–ethanol (1.5% HCl in 75% ethanol; [vol./vol.]) to extract total insulin. Insulin was measured by ELISA.
Islet isolation and insulin secretion
Islets were isolated from P6/P11 offspring and adult mice (males and females) and cultured as described in the ESM Methods. INS-1E cells (mycoplasma-free, kindly provided by C.B. Wollheim, University of Geneva) and isolated islets were preincubated for 1 h in KRB containing 2.8 mmol/l glucose, then incubated for 1 h in KRB containing 2.8 or 12 mmol/l glucose, supplemented with test substances as indicated in the ESM Methods. Secreted insulin and insulin content after extraction in acid–ethanol were measured by ELISA.
INS-1E cell culture, siRNA treatment, western blotting and immunostaining
INS-1E cells were cultured and treated as described in detail in the ESM Methods.
RNA isolation, quantitative RT-PCR and bulk RNAseq
Isolated islets, INS-1E cells, islet cell suspensions and FACS-sorted islet cells (see ESM Methods) were lysed and cellular RNA was isolated according to the instructions provided by the Nucleospin RNA isolation kit (Macherey-Nagel, Düren, Germany). Cellular RNA (0.1 µg) was transcribed into cDNA using the Transcriptor first strand cDNA synthesis kit (#04897030001, Roche Diagnostics, Indianapolis, USA). Semi-quantitative RT-PCR was performed with the LightCycler 480 system (Roche Diagnostics) using mouse/rat specific primers (Invitrogen; Thermo Fisher Scientific, Waltham, USA) as listed in ESM Table 1. Rps13 was used as housekeeping gene. Aliquots of RNA isolated from CD WT and Ffar1 knockout (KO) P6-offspring islets were subjected to RNAseq analysis as previously described [30].
Cytosolic [Ca2+] measurement
Adult WT islets were kept in culture medium during 1 h of loading with FURA-2 AM (2 µmol/l), then transferred to KRB–Henseleit solution (KHB; ESM Methods) supplemented with 0.5% DMSO or FR900359 (1 µmol/l). The islets were then placed in KHB with 2.8 mmol/l glucose for 5 min baseline recording; thereafter, the glucose concentration was raised to 12 mmol/l and the measurement continued for 30 min. KCl (30 mmol/l) was used as positive control. When pre-treatment with Gq inhibitor was omitted, FR900359 was added during recording at high glucose.
Statistical analysis
Data are expressed as mean ± SEM with the number (n) of replicates and independent experiments as indicated. RNAseq dataset was processed with the R package DESeq2 v1.22.1. All other results were subjected to statistical ANOVA analysis using GraphPad Prism (version 9.1.2). Differences were considered statistically significant at p<0.05 and reported as *p<0.05, **p<0.005 and ***p<0.001. Mice and isolated islets were randomly distributed for analysis. Quantification of histological staining was performed by a trained scientist, who was masked to the experimental condition.
Results
Parental HFD feeding worsens glucose responsiveness of insulin secretion in Ffar1 −/− offspring
HFD caused significant weight gain in F0 males (Fig. 1a and ESM Fig. 2a,b). Neither HFD nor Ffar1 deletion altered fed blood variables in F0 mice (ESM Fig. 2c–j). However, HFD induced liver steatosis in WT, and to a lesser extent in Ffar1−/− animals, as previously described (ESM Fig. 2k, [19]).
The role of FFA1 in offspring glucose homeostasis was investigated by comparing WT and Ffar1−/− offspring (F1) at P1–P26 (Fig. 1a and ESM Fig. 3). Parental HFD increased blood glucose in WT but not in Ffar1−/− P6- and P11-offspring, respectively (Fig. 1b,c). Of note, plasma glucose of Ffar1−/− P6-offspring was higher than in WT P6-offspring already under parental CD. This difference was transient, as blood glucose levels of P1-, P11- and P26-offspring were similar regardless of genotype (Fig. 1c and ESM Fig. 3a,b). HFD increased leptin, but had not effect on serum adiponectin in Ffar1−/− P6 mice (ESM Fig. 3c,d). Neither parental diet nor Ffar1 deletion altered offspring body and pancreatic weights (ESM Fig. 3e,f). These results suggest that parental HFD and FFA1 deficiency, the latter independent of parental metabolic pressure, transiently affect neonatal in vivo glucose homeostasis.
Elevated blood glucose level in the offspring could be due to impaired beta cell function. Therefore, we examined insulin secretion in P6- and P11-offspring islets (Fig. 1d–g). At P6, glucose (12 and 20 mmol/l) increased (four- to fivefold) insulin secretion of WT islets irrespective of parental diet (Fig. 1d). On the contrary, insulin secretion of Ffar1−/− islets was affected by parental feeding. Thus, when exposed to parental HFD, Ffar1−/− islets were glucose unresponsive, while under parental CD their insulin secretion was significantly stimulated by 20 mmol/l glucose only (Fig. 1d). Of note, at 12 mmol/l glucose, Ffar1−/− islets secreted significantly less insulin than WT islets, irrespective of parental diet (Fig. 1d). Ffar1−/− P6 islets displayed lower insulin content per islet than WT islets, irrespective of parental diet (ESM Fig. 3g). At P11, the negative effect of parental HFD on GSIS of Ffar1−/− islets persisted at 20 mmol/l glucose. However, GSIS of Ffar1−/− islets under parental CD and of WT islets under parental CD and HFD, as well as their insulin content, were similar (Fig. 1e and ESM Fig. 3h).
Endorsing the role of FFA1 in neonatal insulin secretion, palmitate increased GSIS in CD/HFD WT P6- (5.5- and 4.5-fold, respectively) and P11-islets (3.7- and 5.6-fold, respectively), but failed to augment GSIS of Ffar1−/− offspring islets irrespective of parental diet (Fig. 1f,g). Unlike palmitate, exendin-4 stimulated GSIS to a similar extent in WT and Ffar1−/− islets under parental CD at both P6 (five- and sixfold over GSIS, respectively) and P11 (three- and fourfold over GSIS, respectively). Parental HFD did not significantly change the exendin-4-induced effects (Fig. 1f,g). Forskolin, which bypasses GLP1R, augmented GSIS massively, independent of parental diet and genotype (Fig. 1f,g). These results indicate that FFA1 signalling promotes neonatal GSIS and the offspring’s ability to adapt insulin secretion to metabolic challenge, such as high concentration of glucose and NEFA.
Ffar1 KO offspring islets have increased proliferation rate
Since lower GSIS in Ffar1−/− P6-islets might result from disturbed islet maturation, we performed comparative transcriptome analysis (RNAseq) of CD WT and Ffar1 KO P6-islets. We found 24 up- and eight downregulated genes (+1<Log2 fold change [FC]<−1) in Ffar1 KO islets (Fig. 2a, ESM Table 2). With the exception of lower Sst mRNA level, beta cell-specific genes and G protein mRNAs were largely unaltered in Ffar1 KO P6-islets (ESM Table 3 and ESM Fig. 4a).
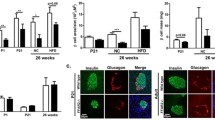
Ffar1 deletion and parental HFD increased expression of proliferation markers in P6-islets. (a) Heat map showing differentially expressed genes in CD Ffar1−/− P6-islets (Log2FC>1 over WT; adjusted p value <0.05); arrowhead denotes genes typically high in immature beta cells (b–d) Semi-quantitative analysis of cellular mRNA levels of P6/P7- and adult offspring islets of WT (black outlines and dots) and Ffar1−/− (red outlines and dots) progenitors fed CD (white-filled bars) and HFD (grey-filled bars). Results are given as mean ± SEM for n=3–10 independent islet preparations. *p<0.05, ***p<0.001 between groups as indicated. (e) Percentage of proliferative, Ki67-positive beta cells (GFP+) and (f) total beta cell mass (mg) assessed as described under Methods. Pancreatic sections (n=4–8 for each animal, of n=3–5 animals/group were examined). (g) Total pancreatic insulin content (µg/pancreas) during postnatal development. Results are expressed as mean ± SEM for n=3–7 pancreases (independent experiments) for each group. (e–g) WT (black line and dots) and Ffar1−/− (red line and dots) offspring animals from CD- (white-filled dots) and HFD- (black-filled dots) fed progenitors; *p<0.05 vs respective P1, † vs respective P6 and ¶ vs respective P11 of the same genotype and diet (time effect); §p<0.05 vs respective genotype on parental CD at P11 (diet effect); ‡p<0.05 vs WT genotype on the same diet at P6 (genotype effect)
Top upregulated and highly expressed in Ffar1−/− P6-islets were the early-response genes Fos, Egr1 and Jun, known to promote proliferation (Fig. 2a). Furthermore, the Ffar1−/− islets had increased levels of Hes1, Myc and Sox9 mRNA, genes typically high in immature beta cells ([31], ESM Fig. 4b). Parental HFD increased the level of Fos and Jun mRNA in WT P6-islets but had an opposite effect on these genes in Ffar1−/− P6-islets (Fig. 2b,c). In addition, Hspa1a, Hspa1b (unfolded protein response) as well as Socs3 and Dusp1 (cytokine signalling) were upregulated in Ffar1−/− P6-islets (ESM Fig. 4c–f). Parental HFD increased these mRNAs in WT P6-islets to levels comparable to those of Ffar1−/− P6-islets. In adult islets, the mRNA levels of the abovementioned genes were low and, similarly to Ffar1, not altered by diet or genotype (Fig. 2b–d and ESM Fig 4c–f). Gene ontology (GO) overrepresentation analysis identified enrichment for terms related to extracellular matrix (ECM) organisation, cytoskeleton, focal adhesion and endoplasmic reticulum (ER) lumen in Ffar1−/− P6-islets (ESM Fig. 4h).
The top ranking upregulated gene in Ffar1−/− P6-islets was Prodh2, encoding the low abundant hydroxyproline dehydrogenase, a mitochondrial enzyme that catabolises 4-hydroxy-proline. Prodh2 upregulation was diet- and age-independent (ESM Fig. 4a,g and Fig. 2a). In accordance with the increased expression of Prodh2, and unlike in WT P6-islets, 4-hydroxy-proline, but also l-proline, improved GSIS of CD Ffar1−/− P6-islets (ESM Fig. 4i,j). Thus, in Ffar1−/− P6-islets PRODH2 supports amino acid-responsive insulin secretion, a functional trait of immature beta cells.
Considering the upregulation of proliferative genes in Ffar1−/− P6-islets, we quantified beta cell proliferation in P1–P26 offspring pancreases (Fig. 2e and ESM Fig. 5). The percentage of Ki67-positive beta cells increased during the breastmilk feeding period, (P1–P11) and declined in weaned P26-offspring, regardless of parental diet and genotype. Notably, under CD conditions, the highest proliferation rate was achieved by Ffar1−/− P6-offspring (3.95±0.69%), which had a significantly higher percentage of Ki67-positive beta cells than WT P6-offspring (1.88±0.56%) (i.e. genotype effect). Under parental HFD conditions, the peak of proliferation shifted toward P11 in both WT and Ffar1−/− offspring, and their proliferation rates were significantly higher (4.48±0.35% and 5.19±0.69% in WT and Ffar1−/−, respectively) than those of corresponding P11 under CD (2.38±0.46% and 2.80±0.42% in WT and Ffar1−/−, respectively; i.e. diet effect) (Fig. 2e). In parallel, beta cell mass increased gradually from P1 to P26 (Fig. 2f). The overall increase was not altered by diet, yet its quality was slightly different between genotypes. Thus, in WT offspring beta cell mass reached the highest level at P26, whereas in Ffar1−/− offspring it stopped increasing further after P11 (Fig. 2f). In parallel to the gain in beta cell mass, pancreatic insulin content steadily increased from P1 to P26. However, as long as the offspring were breastfed, solely the WT progeny were able to adjust their insulin content to parental HFD, as the insulin content of the WT P11-offspring was significantly higher under HFD (16.93±1.75 µg) than under CD (5.18±1.14 µg). Furthermore, WT P11-offspring had a higher insulin content under parental HFD than Ffar1−/− P11-offspring (9.78±0.35 µg) (Fig. 2g).
These results suggest that FFA1 deficiency promotes an immature phenotype in neonatal beta cells along with increased beta cell proliferation, and it impairs the offspring’s ability to adapt insulin content to maternal HFD during breastfeeding.
GSIS is regulated by G proteins in mouse islets
Since we found no altered expression of genes promoting glucose responsiveness and insulin secretion in Ffar1−/− P6-islets, we asked whether impaired neonatal GSIS might emerge as a consequence of disturbed signalling downstream of FFA1. The major FFA1 signalling involved in stimulation of GSIS relies on Gq-coupled phospholipase C and generation of inositol 1,4,5-trisphosphate (IP3) and DAG (diacylglycerol), which then increase cytosolic [Ca2+] [32]. To assess whether Gq promotes GSIS in WT P6-islets, we used FR900359, a specific Gq inhibitor purified from Ardisia crenata leaves as described earlier [33]. In WT P6-islets, FR900359 inhibited palmitate- and TUG-469-amplified insulin secretion (Fig. 3a). Of note, FR900359 also abolished GSIS and its inhibitory effect persisted in the presence of tolbutamide- and exendin-4 (Fig. 3a,b).
Effects of Gq inhibitors and of Gi/o inactivation on GSIS in P6-islets. (a–c, e) WT and (d, f) Ffar1−/− P6-islets were isolated and incubated with test substances as described under Methods. Insulin secretion is expressed as mean ± SEM for the given number of replicates (3–4/experiment) from n=3–4 independent experiments. *p<0.05, **p<0.005, ***p<0.001 between groups as indicated. Ex-4, exendin-4; FR900, FR900359; PTX, pertussis toxin; Tolbut, tolbutamide; YM254, YM-254890
As inhibition of GSIS upon Ffar1 deletion or Gq inhibition may result from derepressed Gi/o-mediated inhibitory pathways, we used PTX to inactivate the Gi/o proteins [34]. PTX increased 100-fold GSIS of WT P6-islets without affecting basal secretion (Fig. 3c). FR900359 reduced the PTX effect by 90%, suggesting that GSIS largely depends on Gq when Gi/o signalling is off. Of note, the simultaneous inhibition of Gq and Gi/o (FR900359 + PTX) unmasked a 14-fold stimulation of insulin secretion by glucose in WT P6-islets (Fig. 3c). PTX-mediated inactivation of Gi/o rendered Ffar1−/− P6-islets glucose responsive (tenfold stimulation), an effect cancelled by FR900359 (Fig. 3d). To confirm the role of Gq for neonatal GSIS, a second Gq inhibitor (YM-254890) was tested. YM-254890 recapitulated the effects of FR900359 on glucose and palmitate-stimulated insulin secretion in WT P6-islets (Fig. 3e). Interestingly, both FR900359 and YM-254890 reduced forskolin-stimulated insulin secretion of Ffar1−/− P6-islets by 40% (Fig. 3f). These findings attest to a key role of Gq for glucose responsiveness and insulin secretion of neonatal islets.
FFA1 can also recruit G12/13, a G protein able to activate RhoA/Rho-associated protein kinase (ROCK) and trigger actin network remodelling. Since ROCK inhibition sustains functional maturation of iPS-derived beta cells [35], we examined the effect of RhoA/ROCK inhibitors in P6-islets. RhoA inhibition with exoenzyme C3 slightly improved glucose responsiveness in Ffar1 KO P6-islets (Fig. 4a). In WT P6-islets, RhoA inhibitor neither improved GSIS nor rescued this after FR900359 treatment (Fig. 4b). Similarly, the ROCK inhibitor H1152 improved glucose responsiveness in Ffar1−/− P6-islets (Fig. 4c) but had no effect in WT P6-islets (Fig. 4d), although the cortical actin web of WT beta cells became punctured upon H1152 treatment (Fig. 4e).
Effects of RhoA and ROCK inhibition on GSIS in WT and Ffar1−/− P6-islets. (a, c) Ffar1−/− and (b, d) WT P6-islets were isolated and incubated with test substances as indicated and described under Methods. Results are expressed as mean ± SEM for the given number of replicates (3–4/experiment) from n=3–7 independent experiments. **p<0.005, ***p<0.001 between groups as indicated. (e) WT P6-islet cells were isolated and cultured as described under Methods. H1152 (1 µmol/l) was added for 1 h in cell culture. The cells were stained for actin (phalloidin, red) and insulin (anti-insulin Ab, magenta). Scale bar, 40 µm. Inh, inhibition
Together, these results indicate that GSIS in neonatal mouse islets largely depends on the activity of stimulatory Gq and inhibitory Gi/o.
Downregulation of Gq expression in INS-1E cells mimics the effect of Gq inhibitors
To further confirm the need for Gq for GSIS, we downregulated Gq expression in INS-1E cells. Following Gnaq siRNA treatment, mRNA and protein levels of Gq were reduced by 80% (Fig. 5a–c), and glucose- and palmitate-stimulated insulin secretion were significantly lower than in INS-1E cells transfected with control siRNA (Fig. 5d). Moreover, the GLP1R antagonist exendin-(9-39) lowered GSIS in control cells but not in INS-1E cells with downregulated Gq (Fig. 5d). Thus, GSIS of INS-1E cells depends partly on active Gq.
GSIS of INS-1E cells depends on Gq. (a–c) INS-1E cells were incubated, transfected and cellular RNA and proteins extracted as described under Methods. (a) Relative mRNA levels of Gnaq assessed by RT-PCR and (b, c) protein level of Gq in untransfected cells (INS-1) and upon treatment with non-targeting (siCon) and anti-Gnaq siRNA. Results are expressed as mean ± SEM of n=3 independent experiments. (d, e) Insulin secretion of (d) INS-1E cells upon treatment with non-targeting (white bars) and Gnaq (green bars) siRNA and (e) INS-1E cells treated as indicated. Results are expressed as mean ± SEM for the given number of replicates (3–4/experiment) from n=3–6 independent experiments; *p<0.05, **p<0.005, ***p<0.001 between groups as indicated. (f) Representative confocal images of INS-1E cells treated as described under Methods and immunostained for Gq (green). Nuclei were stained with DAPI (blue). Scale bar, 50 µm. Ex-9, exendin-(9-39); FR900, FR900359; Palm, palmitate
GSIS of INS-1E cells was also inhibited by FR900359 (Fig. 5e). Since FR900359 acts as a guanine nucleotide dissociation inhibitor for Gq [33] we next assessed via immunostaining whether FR900359 interferes with the subcellular distribution of Gq. In INS-1E cells exposed to glucose (2.8 or 12 mmol/l) ± palmitate, the Gq-associated fluorescence displayed a plasma membrane-like distribution (Fig. 5f, upper panels). In cells treated with FR900359 we detected mainly cytosolic residing Gq-associated fluorescence (Fig. 5f, lower panels). Hence, FR900359 interferes with the subcellular distribution of Gq and in this way it may also hinder GPCRs from optimal recruitment of Gq.
GSIS and cytosolic Ca2+ of adult islets are sensitive to Gq inhibition
To assess whether dependency of GSIS on Gq is restricted to neonatal islets, we performed GSIS in FR900359-treated adult WT islets. Similar to the neonates, FR900359 inhibited GSIS, exendin-4- and palmitate-amplified insulin secretion in adult islets (Fig. 6a,b). In view of the pronounced effect of Gq inhibitors on GSIS, we tested whether FR900359 interferes with cytosolic [Ca2+] in adult WT islets. Although FR900359 reduced the amplitude of [Ca2+] in response to 12 mmol/l glucose (17.3% of AUC glucose), it did not interfere with the initial changes to [Ca2+] induced by glucose (Fig. 6c,d). Acute addition of FR900359 triggered fast reduction of [Ca2+] without affecting the KCl-induced rise in [Ca2+] (8.3% of AUC glucose; Fig. 6e,f). These rather small effects of FR900359 on cytosolic [Ca2+] may not explain its profound effect on GSIS. In accordance with its lack of effect on KCl-induced [Ca2+] increase, FR900359 did not inhibit KCl-stimulated insulin secretion in WT adult islets (Fig. 6g). These observations suggest that Gq action might involve inhibition of a hyperpolarising channel.
Effects of Gq inhibitor FR900359 on GSIS and cytosolic [Ca2+] in adult islets. (a, b, g) Adult WT islets were isolated and incubated as described under Methods. Insulin secretion is expressed as mean ± SEM for the given number of replicates (3–4/experiment) from n=3–5 independent experiments. (c–f) Cytosolic Ca2+ was assessed with FURA-2 in adult mouse islets cultured and treated as described in the Methods. The islets were (c, d) preincubated for 1 h with FR900359 before the recording was started or (e, f) acutely treated with FR900359 during FURA-2 recording. (c, e) Representative traces of cytosolic [Ca2+] measurements. (d, f) AUC of cytosolic [Ca2+] expressed as mean ± SEM for n=4–8 independent experiments. (g) Insulin secretion from adult WT islets isolated and incubated with test substances as described under Methods. Insulin secretion is expressed as mean ± SEM for the indicated number of replicates (3–4/experiment) from n=4 independent experiments. Ex-4, exendin-4; FR/FR900, FR900359; HG, high glucose (12 mmol/l); LG, low glucose (2.8 mmol/l); Palm, palmitate
Discussion
In the present study, we investigated the role of FFA1 on offspring islet function during neonatal life using a well characterised Ffar1−/− mouse model [19]. We show here that deletion of FFA1, as well as inhibition of its signalling partner Gq, impairs GSIS of P6-islets. Moreover, the ability of the offspring islets to sustain insulin secretion when experiencing metabolic challenge (i.e. parental high-fat feeding) depends on FFA1. As such, FFA1 deletion rendered P6- and P11-islets under parental HFD glucose unresponsive, whereas in WT P6- and P11-islets neither GSIS nor exendin-4 or palmitate-stimulated insulin secretion were affected by HFD. These observations suggest that FFA1 and Gq promote neonatal insulin secretion, especially during early life when functional maturation of beta cells is subjected to parental imprinting.
In functionally mature islets, glucose effectiveness on insulin secretion is modulated by GPCRs, that is, stimulatory Gs-coupled (GLP1R, gastric inhibitory polypeptide receptor [GIPR], glucagon receptor [GCGR]) and Gq/11-coupled (FFA1, cholinergic receptor muscarinic 3 [CHRM3]) receptors, and inhibitory Gi/o-coupled receptors (adrenoceptor alpha 2A [ADRA2A], somatostatin receptor [SSTR]) [36]. Distinct from the Gs-coupled receptors therapeutically targeted to treat insulin deficiency in type 2 diabetes, and in spite of the considerable evidence for the positive role of FFA1 for insulin secretion, the therapeutic potential of FFA1 remains controversial [19, 21, 22, 26, 37].
We provide new evidence that FFA1 and its downstream signalling partner Gq improve glucose responsiveness, and describe a Gq–Gi/o interplay. Thus, neonatal GSIS was restricted by Gi/o, as PTX-induced inactivation of Gi/o markedly increased neonatal GSIS. On the other hand, PTX-mediated increase of GSIS largely depended on Gq, as FR900359 curtailed 90% of PTX-stimulated GSIS. Nevertheless, FR900359 had no impact on KCl-augmented insulin secretion, suggesting that Gq likely acts via inhibition of a hyperpolarising K+-channel. Such inhibition of Gi/o-activated K+-channels (GIRK1/4) by Gq-coupled muscarinic receptor has been already described [38]. Previously, Sassmann et al described diminished GSIS of mouse islets with beta cell-specific deletion of Gq/11, as a consequence of impaired closure of KATP-channels [39]. In agreement, we show that Gq inhibition (FR900359 and YM254890) abrogate GSIS. Although FR900359 lowered the plateau phase of glucose-induced cytosolic [Ca2+] increase, it did not affect the initial reduction and the subsequent rise of [Ca2+] triggered by glucose, implying: (1) no toxic effect on islet cell metabolism; and (2) a possible activation of hyperpolarising K+-channels distinct from KATP-channels. In line with this, FR900359 also inhibited tolbutamide-stimulated insulin secretion.
Beside a putative Gq-mediated inhibition of hyperpolarising K+-channels, a role of Gq in Ca2+ mobilisation and Ca2+-dependent exocytosis [40] could explain the loss of GSIS upon Gq inhibition. Indeed, DAG generated by Gq-activated phospholipase C can interact with the SNAP receptor (SNARE) protein Munc13-1 and prime the secretory vesicles for exocytosis in chromaffin cells [41]. The sensitivity of exendin-4- and forskolin-augmented GSIS toward Gq inhibition is in disagreement with some previous observations [39, 42] but in accordance with a recent work indicating that GLP-1R/Gq signalling rescues insulin secretion of islets exposed to metabolic stress [25]. Whether such a Gs-to-Gq shift explains this discrepancy needs further experimental work.
FR900359 abrogated GSIS and siRNA-mediated downregulation of Gnaq impaired GSIS in INS-1E cells, suggesting a direct action of Gq in beta cells. However, we cannot exclude an action of Gq inhibitor in alpha cells, as Gq supports secretion of glucagon, which augments insulin secretion via paracrine activation of GLP1R [43].
Neonatal beta cells are proliferative, a capacity gradually lost with the acquisition of functional competence [44]. Our RNAseq results, in accordance with previous observations [31, 45], revealed elevated expression of several markers of beta cell immaturity and dedifferentiation (Sox9, Hes1, Myc) in Ffar1−/− P6-offspring islets, a further indication that FFA1 promotes functional maturation. In fact, 15 of the 24 top-upregulated (Log2FC>1) genes in Ffar1−/− P6-islets are highly expressed in juvenile islets and downregulated in adult islets, as previously shown (GEO: GSE87375 in [31, 46]). Several lines of evidence indicate that maternal overnutrition impairs offspring beta cell function and increases offspring risk for type 2 diabetes [10, 11]. Similarly, a loss-of-function mutation of FFA1, which is more common in severely obese individuals, is associated with impaired cytosolic [Ca2+] and reduced insulin secretion, indicating the importance of FFA1 for adaptive insulin secretion under conditions of lipid overload [37]. Both Ffar1 deletion and parental HFD modulated offspring beta cell proliferation, but with different consequences. Thus, Ffar1 deletion, in spite of promoting proliferation of P6-islets, increased neither functional beta cell mass nor pancreatic insulin content, very likely because proliferation was driven by the immature phenotype (i.e. Myc-dependent) [44]. Among the P6-offspring insulin content was the lowest in the HFD KO group, i.e. the one with the glucose-unresponsive islets. Although HFD extended the proliferative time window toward P11 in a genotype-independent manner, only WT P11-offspring increased their pancreatic insulin content, confirming the necessity of FFA1 for offspring adaptation to parental metabolic stress. These FFA1- and parental diet-related effects were limited to the pre-weaning phase, (i.e. exposure to maternal nutrition), as all P26 offspring displayed similar proliferation rates, beta cell mass and insulin content. The lipid-rich neonatal environment may therefore justify the functional relevance of FFA1 during early life, since FFA1 is the main mediator of long-chain fatty acid effects on GSIS [20, 47]. Whether Gq solely transmits the effects of FFA1 in neonatal islets is, to our knowledge, not yet known. Nevertheless, Gi and Gs were reported to regulate neonatal beta cell proliferation [28, 48].
Based on our results, we postulate that FFA1 and Gq promote GSIS in neonatal islets, and that FFA1 is required for offspring adaptation to parental fat overload. In conclusion, therapeutic modulation of FFA1/Gq signalling might protect from the adverse effects of maternal obesity on offspring glucose homeostasis.
Abbreviations
- CD:
-
Chow diet
- FFA1:
-
Free fatty acid receptor 1
- FC:
-
Fold change
- GLP1R:
-
Glucagon-like peptide-1 receptor
- GPCR:
-
G-protein-coupled receptor
- GSIS:
-
Glucose-stimulated insulin secretion
- HFD:
-
High-fat diet
- KO:
-
Knockout
- P1–P26:
-
Postnatal days 1, 6, 11 and 26
- PTX:
-
Pertussis toxin
- ROCK:
-
Rho-associated protein kinase
- WT:
-
Wild-type
References
Prentki M, Nolan CJ (2006) Islet beta cell failure in type 2 diabetes. J Clin Invest 116:1802–1812. https://doi.org/10.1172/JCI29103
Weir GC, Bonner-Weir S (2004) Five stages of evolving beta-cell dysfunction during progression to diabetes. Diabetes 53(Suppl 3):S16–S21. https://doi.org/10.2337/diabetes.53.suppl_3.S16
Cohrs CM, Panzer JK, Drotar DM et al (2020) Dysfunction of persisting β cells is a key feature of early type 2 diabetes pathogenesis. Cell Rep 31:107469. https://doi.org/10.1016/j.celrep.2020.03.033
Helman A, Cangelosi AL, Davis JC et al (2020) A nutrient-sensing transition at birth triggers glucose-responsive insulin secretion. Cell Metab 31:1004–1016. https://doi.org/10.1016/j.cmet.2020.04.004. (e1005)
Jermendy A, Toschi E, Aye T et al (2011) Rat neonatal beta cells lack the specialised metabolic phenotype of mature beta cells. Diabetologia 54:594–604. https://doi.org/10.1007/s00125-010-2036-x
Henquin JC, Nenquin M (2018) Immaturity of insulin secretion by pancreatic islets isolated from one human neonate. J Diabetes Investig 9:270–273. https://doi.org/10.1111/jdi.12701
O’Dowd JF, Stocker CJ (2013) Endocrine pancreatic development: impact of obesity and diet. Front Physiol 4:170. https://doi.org/10.3389/fphys.2013.00170
Moullé VS, Parnet P (2019) Effects of nutrient intake during pregnancy and lactation on the endocrine pancreas of the offspring. Nutrients 11(11):2708. https://doi.org/10.3390/nu11112708
Stolovich-Rain M, Enk J, Vikesa J et al (2015) Weaning triggers a maturation step of pancreatic β cells. Dev Cell 32:535–545. https://doi.org/10.1016/j.devcel.2015.01.002
Nielsen JH, Haase TN, Jaksch C et al (2014) Impact of fetal and neonatal environment on beta cell function and development of diabetes. Acta Obstet Gynecol Scand 93:1109–1122. https://doi.org/10.1111/aogs.12504
Jacovetti C, Regazzi R (2022) Mechanisms underlying the expansion and functional maturation of β-cells in newborns: impact of the nutritional environment. Int J Mol Sci 23(4):2096. https://doi.org/10.3390/ijms23042096
Elsakr JM, Gannon M (2017) Developmental programming of the pancreatic islet by in utero overnutrition. Trends Dev Biol 10:79–95
Nicholas LM, Nagao M, Kusinski LC, Fernandez-Twinn DS, Eliasson L, Ozanne SE (2020) Exposure to maternal obesity programs sex differences in pancreatic islets of the offspring in mice. Diabetologia 63:324–337. https://doi.org/10.1007/s00125-019-05037-y
Graus-Nunes F, Dalla Corte Frantz E, Lannes WR, da Silva Menezes MC, Mandarim-de-Lacerda CA, Souza-Mello V (2015) Pregestational maternal obesity impairs endocrine pancreas in male F1 and F2 progeny. Nutrition 31:380–387. https://doi.org/10.1016/j.nut.2014.08.002
Bringhenti I, Ornellas F, Mandarim-de-Lacerda CA, Aguila MB (2016) The insulin-signaling pathway of the pancreatic islet is impaired in adult mice offspring of mothers fed a high-fat diet. Nutrition 32:1138–1143. https://doi.org/10.1016/j.nut.2016.03.001
Lahti-Pulkkinen M, Bhattacharya S, Wild SH et al (2019) Consequences of being overweight or obese during pregnancy on diabetes in the offspring: a record linkage study in Aberdeen, Scotland. Diabetologia 62:1412–1419. https://doi.org/10.1007/s00125-019-4891-4
Ballard O, Morrow AL (2013) Human milk composition: nutrients and bioactive factors. Pediatr Clin North Am 60:49–74. https://doi.org/10.1016/j.pcl.2012.10.002
Briscoe CP, Tadayyon M, Andrews JL et al (2003) The orphan G protein-coupled receptor GPR40 is activated by medium and long chain fatty acids. J Biol Chem 278:11303–11311. https://doi.org/10.1074/jbc.M211495200
Steneberg P, Rubins N, Bartoov-Shifman R, Walker MD, Edlund H (2005) The FFA receptor GPR40 links hyperinsulinemia, hepatic steatosis, and impaired glucose homeostasis in mouse. Cell Metab 1:245–258. https://doi.org/10.1016/j.cmet.2005.03.007
Itoh Y, Kawamata Y, Harada M et al (2003) Free fatty acids regulate insulin secretion from pancreatic beta cells through GPR40. Nature 422:173–176. https://doi.org/10.1038/nature01478
Ghislain J, Poitout V (2017) The role and future of FFA1 as a therapeutic target. Handb Exp Pharmacol 236:159–180. https://doi.org/10.1007/164_2016_51
Nauck M (2016) Incretin therapies: highlighting common features and differences in the modes of action of glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors. Diabetes Obes Metab 18:203–216. https://doi.org/10.1111/dom.12591
Hauge M, Vestmar MA, Husted AS et al (2015) GPR40 (FFAR1) - combined Gs and Gq signaling in vitro is associated with robust incretin secretagogue action ex vivo and in vivo. Mol Metab 4:3–14. https://doi.org/10.1016/j.molmet.2014.10.002
Rives ML, Rady B, Swanson N et al (2018) GPR40-mediated Gα12 activation by allosteric full agonists highly efficacious at potentiating glucose-stimulated insulin secretion in human islets. Mol Pharmacol 93:581–591. https://doi.org/10.1124/mol.117.111369
Oduori OS, Murao N, Shimomura K et al (2020) Gs/Gq signaling switch in β cells defines incretin effectiveness in diabetes. J Clin Invest 130:6639–6655. https://doi.org/10.1172/JCI140046
Wagner R, Kaiser G, Gerst F et al (2013) Reevaluation of fatty acid receptor 1 as a drug target for the stimulation of insulin secretion in humans. Diabetes 62:2106–2111. https://doi.org/10.2337/db12-1249
Buteau J (2008) GLP-1 receptor signaling: effects on pancreatic beta-cell proliferation and survival. Diabetes Metab 34(Suppl 2):S73-77. https://doi.org/10.1016/S1262-3636(08)73398-6
Berger M, Scheel DW, Macias H et al (2015) Gαi/o-coupled receptor signaling restricts pancreatic β-cell expansion. Proc Natl Acad Sci USA 112:2888–2893. https://doi.org/10.1073/pnas.1319378112
Brelje TC, Bhagroo NV, Stout LE, Sorenson RL (2008) Beneficial effects of lipids and prolactin on insulin secretion and beta-cell proliferation: a role for lipids in the adaptation of islets to pregnancy. J Endocrinol 197:265–276. https://doi.org/10.1677/JOE-07-0657
Gerst F, Kemter E, Lorza-Gil E et al (2021) The hepatokine fetuin-A disrupts functional maturation of pancreatic beta cells. Diabetologia 64:1358–1374. https://doi.org/10.1007/s00125-021-05435-1
Qiu WL, Zhang YW, Feng Y, Li LC, Yang L, Xu CR (2017) Deciphering pancreatic islet β cell and α cell maturation pathways and characteristic features at the single-cell level. Cell Metab 25:1194–1205. https://doi.org/10.1016/j.cmet.2017.04.003. (e1194)
Fujiwara K, Maekawa F, Yada T (2005) Oleic acid interacts with GPR40 to induce Ca2+ signaling in rat islet beta-cells: mediation by PLC and L-type Ca2+ channel and link to insulin release. Am J Physiol Endocrinol Metab 289:E670–E677. https://doi.org/10.1152/ajpendo.00035.2005
Schrage R, Schmitz AL, Gaffal E et al (2015) The experimental power of FR900359 to study Gq-regulated biological processes. Nat Commun 6:10156. https://doi.org/10.1038/ncomms10156
Peterhoff M, Sieg A, Brede M, Chao CM, Hein L, Ullrich S (2003) Inhibition of insulin secretion via distinct signaling pathways in alpha2-adrenoceptor knockout mice. Eur J Endocrinol 149:343–350. https://doi.org/10.1530/eje.0.1490343
Ghazizadeh Z, Kao DI, Amin S et al (2017) ROCKII inhibition promotes the maturation of human pancreatic beta-like cells. Nat Comm 8:298. https://doi.org/10.1038/s41467-017-00129-y
Ahrén B (2009) Islet G protein-coupled receptors as potential targets for treatment of type 2 diabetes. Nat Rev Drug Discov 8:369–385. https://doi.org/10.1038/nrd2782
Vettor R, Granzotto M, De Stefani D et al (2008) Loss-of-function mutation of the GPR40 gene associates with abnormal stimulated insulin secretion by acting on intracellular calcium mobilization. J Clin Endocrinol Metab 93:3541–3550. https://doi.org/10.1210/jc.2007-2680
Hill JJ, Peralta EG (2001) Inhibition of a Gi-activated potassium channel (GIRK1/4) by the Gq-coupled m1 muscarinic acetylcholine receptor. J Biol Chem 276:5505–5510. https://doi.org/10.1074/jbc.M008213200
Sassmann A, Gier B, Gröne HJ, Drews G, Offermanns S, Wettschureck N (2010) The Gq/G11-mediated signaling pathway is critical for autocrine potentiation of insulin secretion in mice. J Clin Invest 120:2184–2193. https://doi.org/10.1172/JCI41541
Pfeil EM, Brands J, Merten N et al (2020) Heterotrimeric G protein subunit Gαq is a master switch for Gβγ-mediated calcium mobilization by Gi-coupled GPCRs. Mol Cell 80:940–954. https://doi.org/10.1016/j.molcel.2020.10.027. (e946)
Bauer CS, Woolley RJ, Teschemacher AG, Seward EP (2007) Potentiation of exocytosis by phospholipase C-coupled G-protein-coupled receptors requires the priming protein Munc13-1. J Neurosci 27:212–219. https://doi.org/10.1523/JNEUROSCI.4201-06.2007
Lorza-Gil E, Kaiser G, Rexen Ulven E et al (2020) FFA2-, but not FFA3-agonists inhibit GSIS of human pseudoislets: a comparative study with mouse islets and rat INS-1E cells. Sci Rep 10:16497. https://doi.org/10.1038/s41598-020-73467-5
Svendsen B, Larsen O, Gabe MBN et al (2018) Insulin secretion depends on intra-islet glucagon signaling. Cell Rep 25:1127–1134. https://doi.org/10.1016/j.celrep.2018.10.018. (e1122)
Puri S, Roy N, Russ HA et al (2018) Replication confers beta cell immaturity. Nat Commun 9:485. https://doi.org/10.1038/s41467-018-02939-0
Diedisheim M, Oshima M, Albagli O et al (2018) Modeling human pancreatic beta cell dedifferentiation. Mol Metab 10:74–86. https://doi.org/10.1016/j.molmet.2018.02.002
Arda HE, Li L, Tsai J et al (2016) Age-dependent pancreatic gene regulation reveals mechanisms governing human beta cell function. Cell Metab 23:909–920. https://doi.org/10.1016/j.cmet.2016.04.002
Hauke S, Keutler K, Phapale P, Yushchenko DA, Schultz C (2018) Endogenous fatty acids are essential signaling factors of pancreatic beta-cells and insulin secretion. Diabetes 67:1986–1998. https://doi.org/10.2337/db17-1215
Serra-Navarro B, Fernandez-Ruiz R, García-Alamán A et al (2021) Gsα-dependent signaling is required for postnatal establishment of a functional β-cell mass. Mol Metab 53:101264. https://doi.org/10.1016/j.molmet.2021.101264
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Acknowledgements
Some of the data were presented as an abstract at the 58th EASD Annual Meeting in 2022. Items from https://bioicons.com were used for the graphical abstract.
Data availability
The complete gene list of RNAseq data is available on request from the authors.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was supported by a grant (01GI0925) from the Federal Ministry of Education and Research (BMBF) to the German Center for Diabetes Research (DZD e.V.). ERU is funded by the Lundbeck Foundation (grant R307-2018-2950). EK and GMK gratefully acknowledge support of this work by the DFG-funded Research Unit FOR2372 with the grants 290847012/FOR2372 (to EK), as well as 290827466/FOR2372 (to GMK).
Authors’ relationships and activities
The authors declare that there are no relationships or activities that might bias, or be perceived to bias, their work.
Contribution statement
SU, FG and ELG designed the experiments and drafted the manuscript. FG, ELG, SH, GK, CC and MDAH performed data acquisition and analysis. GK, CC, MDAH, SH, ERU, TU, LPR, GMK, EK, ALB and HUH contributed to interpretation of data and critically revised the manuscript. All authors approved the final version to be published. FG and ELG are the guarantors of this work, have full access to all data and assume responsibility for the integrity of the data and the accuracy of the data analysis.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lorza-Gil, E., Kaiser, G., Carlein, C. et al. Glucose-stimulated insulin secretion depends on FFA1 and Gq in neonatal mouse islets. Diabetologia 66, 1501–1515 (2023). https://doi.org/10.1007/s00125-023-05932-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-023-05932-5