Abstract
Aims/hypothesis
Ageing can lead to reduced insulin sensitivity and loss of pancreatic beta cell function, predisposing individuals to the development of diabetes. The aim of this study was to assess the contribution of microRNAs (miRNAs) to age-associated beta cell dysfunction.
Methods
The global mRNA and miRNA profiles of 3- and 12-month-old rat islets were collected by microarray. The functional impact of age-associated differences in miRNA expression was investigated by mimicking the observed changes in primary beta cells from young animals.
Results
Beta cells from 12-month-old rats retained normal insulin content and secretion, but failed to proliferate in response to mitotic stimuli. The islets of these animals displayed modifications at the level of several miRNAs, including upregulation of miR-34a, miR-124a and miR-383, and downregulation of miR-130b and miR-181a. Computational analysis of the transcriptomic modifications observed in the islets of 12-month-old rats revealed that the differentially expressed genes were enriched for miR-34a and miR-181a targets. Indeed, the induction of miR-34a and reduction of miR-181a in the islets of young animals mimicked the impaired beta cell proliferation observed in old animals. mRNA coding for alpha-type platelet-derived growth factor receptor, which is critical for compensatory beta cell mass expansion, is directly inhibited by miR34a and is likely to be at least partly responsible for the effects of this miRNA.
Conclusions/interpretation
Changes in the level of specific miRNAs that occur during ageing affect the proliferative capacity of beta cells. This might reduce their ability to expand under conditions of increased insulin demand, favouring the development of type 2 diabetes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Insulin secretion from pancreatic beta cells plays a central role in blood glucose homeostasis and metabolism control. Reduced sensitivity of insulin target tissues and a consequent rise in insulin demand is normally compensated by expansion of beta cells and an increase in their secretory activities. Failure in this compensatory process results in the release of insufficient insulin to cover the organism’s needs and the development of type 2 diabetes [1]. Ageing is a risk factor for several metabolic diseases, including type 2 diabetes. Indeed, ageing can affect both insulin secretion and insulin action, and predisposes to glucose intolerance and diabetes [2]. Moreover, ageing is associated with impaired proliferation and increased sensitivity of beta cells to apoptosis [3], reducing their capacity to cope with an insulin-resistance state. At present, the mechanisms underlying these phenomena are not fully understood, but changes in the expression of genes coding for key proteins have been reported to be involved in the age-associated decline of beta cell function.
MicroRNAs (miRNAs) are short non-coding RNAs that bind to the 3′ untranslated region (UTR) of target mRNAs, causing translational repression and/or messenger degradation [4]. During the past decade, several studies have demonstrated the involvement of miRNAs in the regulation of beta cell function and survival [5–7]. Although the role of miRNAs in the regulation of beta cell activities has been investigated in various conditions, including pregnancy and obesity [8–11], the potential contribution of these molecules to age-associated beta cell impairment has not, as yet, been explored.
In this study, we compared the islet miRNA profiles of 3- and 12-month-old rats. Several differentially expressed miRNAs were identified and their roles in beta cell secretion, proliferation and survival upon chronic exposure to proapoptotic conditions were systematically investigated.
Methods
Chemicals
Prolactin, IL-1β, exendin-4 and platelet-derived growth factor (PDGF)-AA were obtained from Sigma-Aldrich (St Louis, MO, USA). TNF-α was obtained from Enzo Life Sciences (Lausanne, Switzerland) and IFNγ was obtained from R&D Systems (Minneapolis, MN, USA).
Animals
Wistar rats were from Janvier Laboratories (Le Genest St Ile, France). All procedures were approved by the Swiss Veterinary Office and were in accordance with National Institutes of Health guidelines.
Islet isolation and cell culture
Islets were isolated by collagenase digestion [12] and cultured in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) [9]. Dissociated islet cells were obtained by trypsin digestion [8]. Human islets were received from the Cell Isolation and Transplantation Center (University of Geneva, Geneva, Switzerland). Dissociated human islet cells were cultured in CMRL medium (Invitrogen) [9]. The rat insulin-secreting cell line INS832/13 was cultured as previously described [8].
Profiling of miRNA and mRNA
RNA was isolated using the miRNeasy Kit (Qiagen, Hombrechtikon, Switzerland). MiRNA expression profiling was carried out using Agilent Technologies (Santa Clara, CA, USA) miRNA Microarrays [9]. Profiling of mRNAs was carried out by Arraystar (Rockville, MD, USA).
Measurement of miRNA and mRNA expression
MiRNA expression was assessed using the miRCURY LNA Universal RT miRNA PCR kit (Exiqon, Vedbaek, Denmark). Measurements of mRNA levels were performed by quantitative real-time PCR (qPCR; Bio-Rad, Reinach, Switzerland) with custom-designed primers (Microsynth, Balgach, Switzerland) (see Electronic Supplementary Material [ESM] Methods). MiRNA expression was normalised to the level of U6 or miR-7, while mRNA expression was normalised to 18S. The level of selected miRNAs in cadaveric human islets from 11 normoglycaemic donors, and their association with age, was assessed in our previously published global miRNA profiling dataset [13].
Transfection and modulation of miRNA levels
INS832/13 cells and primary rat or human dissociated islet cells were transfected with Lipofectamine 2000 (Invitrogen), either with RNA oligonucleotide duplexes (Eurogentec, Seraing, Belgium) corresponding to the sequence of the miRNAs (overexpression) or with single-stranded miScript miRNA Inhibitors (Qiagen) that specifically inhibit endogenous miRNAs [9]. A custom-designed small interfering RNA duplex directed against green fluorescent protein (Eurogentec) and a miScript reference miRNA (Qiagen) were used as respective controls.
Insulin secretion
The insulin content of and insulin secretion by dissociated rat islet cells were measured by ELISA [14].
Cell death assessment
Rat and human islet cells were incubated with 1 μg/ml Hoechst 33342 (Invitrogen) for 1 min. At least 103 cells/condition were analysed under fluorescence microscopy (AxioCam MRc 5, Zeiss, Feldbach, Switzerland) to score the fraction of cells displaying pycnotic nuclei.
Proliferation assay
Cells were cultured on poly-l-lysine-coated glass coverslips. They were fixed with cold methanol, permeabilised with 0.5% saponin (Sigma-Aldrich) and exposed for 1 h to antibodies against Ki67 (1:400) (Abcam, Cambridge, UK) and insulin (1:10,000) (Millipore, Zug, Switzerland). Coverslips were then incubated for 1 h with anti-rabbit Alexa-Fluor-488 and anti-mouse Alexa-Fluor-555 antibodies (Invitrogen). Images were collected on an AxioVision fluorescence microscope.
Luciferase assay
A luciferase reporter construct was generated by inserting 212 nucleotides of the 3′ UTR sequence of rat Pdgfra surrounding the putative binding site of miR-34a between the XhoI and EcoRI sites of psiCHECK-1 (ESM Methods). Luciferase activity was measured using a dual-luciferase reporter assay (Promega, Madison, WI, USA). Firefly luciferase activity was normalised for transfection efficiency with the SV40-driven Renilla activity generated by pGL3-Basic (Promega).
Western blotting
Cells were lysed in Laemmli buffer. Lysates were resolved by SDS-PAGE, transferred to a PVDF membrane and detected using antibodies against PDGF receptor α (PDGFRα) (catalogue no. 3174; Cell Signaling Technology, Danvers, MA, USA) and α-tubulin (Fluka Chemie, Buchs, Switzerland) [15]. After 1 h incubation at room temperature with horseradish peroxidase-conjugated secondary antibodies (Fluka Chemie), membranes were revealed by chemiluminescence (Immobilon, Millipore) using the ChemiDoc XRS+ System (Bio-Rad Laboratories).
miRNA target enrichment analysis
For each differentially expressed miRNA, we estimated the median number of miRNA recognition elements (Mobs) predicted, using TargetScan (version 6.2) [16], in the 3′ UTR (rn6, downloaded from UCSC [17] on 10 April 2015) of n up- or downregulated genes. To obtain an empirical p value associated with each Mobs, we independently estimated 1,000 times the median TargetScan predicted density of miRNA recognition elements for N regions of matching length randomly sampled from the 3′ UTRs of rat islet mRNAs that were not differentially expressed.
Statistical analysis
Statistical differences were tested using Student’s t test or for multiple comparisons, with ANOVA followed by a post hoc Dunnett test, with a discriminating p value of 0.05 (SAS statistical package, Cary, NC, USA).
Results
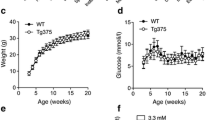
As observed by others [18, 19], 12-month-old male Wistar rats displayed an increase in body weight but no difference in blood glucose levels compared with 3-month-old animals (ESM Fig. 1). In agreement with these observations, insulin content and glucose-stimulated insulin secretion from islets isolated from older animals were comparable with those of younger animals (Fig. 1a, b) and the sensitivity of islet cells to apoptosis was unchanged (Fig. 1c). However, in contrast to beta cells from young animals, those isolated from 12-month-old rats displayed no proliferative response to exendin-4, PDGF or prolactin (Fig. 1d–f).
Insulin secretion, apoptosis and proliferation in islets of aged rats. (a) Insulin secretion of rat islet cells in response to 2 mmol/l (black bars) or 20 mmol/l (white bars) glucose. Results are represented as fold change compared with 2 mmol/l glucose in 3-month-old rats. (b) Insulin content was normalised to total protein content. (c) Apoptosis was measured in the islet cells of young (white bars) and aged (black bars) animals treated with IL-1β (10 ng/ml), IFNγ (300 ng/ml) or with a cytokine (Cyt.) mix (0.1 ng/ml IL-1β, 10 ng/ml TNF-α and 30 ng/ml IFNγ). *p < 0.05 vs untreated rats of the same age. (d–f) Beta cell proliferation in rat islet cells of 3- and 12-month-old animals treated with exendin-4 (Ex-4; 100 nmol/l) (d), PDGF-AA (50 ng/ml) (e) or prolactin (PRL; 500 ng/ml) (f) for 5 days was assessed by staining with anti-Ki67 and anti-insulin antibodies. Results are means ± SD of at least three independent experiments. *p < 0.05 by ANOVA and Dunnett’s post hoc test. Ctrl, control; mo, month
To determine whether these phenotypic traits were linked to transcriptomic differences, we used microarray analysis to compare the global mRNA profiles of the islets isolated from young and old rats. We detected 632 upregulated and 397 downregulated genes (fold change >2.0; nominal p < 0.05, n = 3) in the islets of 12-month-old rats (ESM Tables 1, 2; microarray data are deposited in Gene Expression Omnibus under the accession code GSE72466). These changes are unlikely to reflect differences in islet composition, since the islet β-cell content is not modified in 12-month-old animals [20]. Among the upregulated mRNAs, pathway analysis revealed an enrichment of genes involved in insulin secretion, mitogen-activated protein kinase signalling and MODY (ESM Table 3). Changes in the expression of selected genes belonging to these pathways were verified by qPCR (ESM Fig. 2). In particular, we confirmed the induction of genes involved in insulin secretion (such as the calcium-channel subunits Cacna1c and Cacna1d) and MODY (Slc2a2 and Neurod1), and encoding the transcription factor p53 (Tp53).
MiRNAs are major regulators of gene expression and are involved in senescence and other age-associated phenomena [21–23]. To study the potential contribution of these RNAs to age-associated beta cell dysfunction, we analysed by microarray modifications in the islet miRNA profile of 12-month-old animals. Out of 307 detected miRNAs, 69 displayed changes in their expression level (nominal p < 0.05) (ESM Table 4; GSE72466). Changes in the level of all miRNAs displaying an adjusted p < 0.05 were verified by qPCR. We indeed confirmed the upregulation of miR-124a and miR-383 and the downregulation of miR-181a and miR-130b observed by microarray (Fig. 2a–d). The modifications in the levels of these miRNAs persisted and were in some cases exacerbated in the islets of 23-month-old animals (Fig. 2a–d). Interestingly, miR-124a plays a role in beta cell development [24] and insulin secretion [25], while miR-383 is differentially expressed in db/db mice and in mice fed a high-fat diet, two models of type 2 diabetes [9]. We were unable to detect reproducible changes in the levels of miR-29b, miR-129-1* (currently annotated miR-129-1-3p), miR-484 and miR-488 (ESM Fig. 3). Thus, these miRNAs were not further investigated.
MiR-34a increases in different tissues during ageing [26, 27] and plays a role in islet cell survival and insulin secretion [28]. Although they did not reach statistical significance, the microarray data suggested a possible increase of miR-34a, miR-34b and miR-34c in the islets of old animals (ESM Table 4). Indeed, analysis of miR-34a expression by qPCR confirmed an upregulation of this miRNA in the islets of both 12- and 23-month-old rats (Fig. 2e). Islet miR-34a expression was also positively correlated with age in human islets from normoglycaemic donors of different ages (r 2 = 0.58, p = 0.007) (Fig. 3a). In contrast, we did not detect significant (p > 0.05) correlations between the levels of the other differentially expressed miRNAs and the age of islet donors (data not shown). In this case, the effect of ageing observed in congenic animals may have been masked by genetic variations in the human donors or by other confounding factors such as sex or BMI [29].
Age-associated changes in miR-34a expression in human islets and in other rat organs. RNA was isolated from the islets of human donors of various ages (a) and from the brain (b) and liver (c) of rats aged 3, 12 and 23 months. (a) The miR-34a level was positively correlated with the age of human islet donors. miRNA expression was measured using TaqMan Low Density Array plates (Life Technologies, Zug, Switzerland) and a global mean normalisation strategy to control for RNA input. Relative miR-34a levels were calculated using the mean ΔCt of all 11 samples and log2 transformed for normality. miR-34a expression in brain (b) and liver (c) of rats. The data are expressed as fold change vs the level of expression in 3-month-old rats. (b, c) Data represent means ± SD *p < 0.05 vs 3-month-old rats by ANOVA and Dunnett’s post hoc test. Mo, month
We then verified whether the observed modifications in miRNA expression are specific for islet cells. Most of the selected miRNAs displayed expression profiles in insulin target tissues (liver, skeletal muscle and adipose tissue) and brain that differed from those of islets (ESM Table 5). In contrast, and in line with the literature [26, 27], miR-34a was upregulated in the liver and brain of older rats relative to 3-month-old rats (Fig. 3b, c).
To investigate the impact of the selected miRNAs on beta cell activities, islet cells of 3-month-old animals were transfected with miRNA mimics or anti-miRs to reproduce the changes in their expression that occur during ageing (ESM Fig. 4). Modifications in the levels of these miRNAs did not affect insulin content or insulin secretion (Fig. 4). Since ageing is characterised by increased susceptibility of beta cells to apoptotic stimuli [3], we investigated the impact of these miRNAs on beta cell survival. We found that overexpression of miR-34a triggers apoptosis of rat (Fig. 5a) and human islet cells (Fig. 5b). Downregulation of miR-130b and overexpression of miR-383 did not affect apoptosis of rat (Fig. 5c, e) or human (Fig. 5d, f) islet cells, whereas they exerted a protective effect when the cells were treated with cytokines. Downregulation of miR-181a or overexpression of miR-124a modified neither the basal apoptotic rate nor survival in the presence of cytokines (ESM Fig. 5).
Impact of changes in miRNA expression on insulin secretion and content. Dissociated rat islet cells were transfected with oligonucleotides leading to overexpression (a–c) or downregulation (d, e) of the indicated miRNAs. Insulin secretion (a–e) and insulin content (f, g) in response to 2 mmol/l (black bars) or 20 mmol/l (white bars) glucose was measured 48 h post transfection. Insulin release is expressed as fold change vs 2 mmol/l glucose in the control (ctrl) condition. Data represent means ± SD
Impact of changes in miRNA expression on apoptosis. Dispersed rat (a, c, e) and human (b, d, f) islet cells were transfected with oligonucleotides leading to overexpression (a, b, e, f) or downregulation (c, d) of the indicated miRNAs. Apoptosis was assessed by scoring the cells displaying pycnotic nuclei. Incubation of cells transfected with control (ctrl) oligonucleotides during 24 h with a mix of proinflammatory cytokines (10 ng/ml TNF-α, 0.1 ng/ml IL-1β and 30 ng/ml IFNγ; cyt) was used as a positive control for apoptosis. Results are the means ± SD of three or four independent experiments. *p < 0.05 (vs control for a, b) by ANOVA and Dunnett’s post hoc test
We next assessed the impact of the selected miRNAs on beta cell proliferation. We observed that overexpression of miR-34a (Fig. 6a, b) or downregulation of miR-181a (Fig. 6c, d) did not affect basal beta cell proliferation, but did inhibit proliferation stimulated by exendin-4 or PDGF-AA. In contrast, beta cell proliferation was unaffected by overexpression of miR-383 and miR-124a or downregulation of miR-130b (Fig. 6e–g).
Impact of changes in miRNA expression on beta cell proliferation. Rat islet cells were transfected with oligonucleotides leading to overexpression (a, b, e, f) or downregulation (c, d, g) of the indicated miRNAs and treated with exendin-4 (Ex-4; 100 nmol/l) or PDGF-AA (50 ng/ml) (black bars). Beta cell proliferation was assessed 48 h later by anti-Ki67 and anti-insulin staining. The results represent the means ± SD of three to six independent experiments. *p < 0.05 by ANOVA and Dunnett’s post hoc test. Ctrl, control
To unravel the mechanisms underlying the effects of miR-34a and miR-181a on age-associated beta cell dysfunction, we searched for potential targets within genes expressed in the islets of aged animals. Analysis of the protein-coding genes differentially expressed in islets of 12-month-old rats revealed significant enrichment for potential targets of miR-181a in the upregulated genes (Fig. 7b, ESM Table 6) and depletion of predicted targets in the downregulated genes (Fig. 7a, ESM Table 7). Moreover, the upregulated genes were significantly depleted from miR-34a targets (Fig. 7d, ESM Table 6) and, although the values did not reach statistical significance (p < 0.071), the downregulated genes tended to be enriched in the predicted targets of this miRNA (Fig. 7c, ESM Table 7). These observations suggest that age-associated changes in miR-181a and miR-34a levels contribute to the gene expression differences observed in the islets of aged rats.
Number of miR-34a and miR-181a recognition elements in the 3′ UTR of differentially expressed mRNAs in islets of 12-month-old rats. The median number of recognition elements (Mobs) for miR-181a (a, b) and miR-34a (c, d) predicted in the 3′ UTR of all down- (a, c) and upregulated (b, d) mRNAs in ageing (arrows) was compared with a null distribution of the median number of predicted recognition elements obtained for 1,000 randomly sampled sets of 3′ UTRs from mRNAs expressed in rat islets. p is the associated empirical p value and corresponds to the proportion of values in the null distribution that are either smaller or larger (left and right side of the distribution, respectively) than Mobs. (a) p = 0.025; (b) p = 0.023; (c) p = 0.071; (d) p = 0
PDGFRα, a receptor whose decrease contributes to impaired age-associated beta cell proliferation [30], is a member of the beta cell ‘disallowed gene’ family [31]. Western blot analysis revealed that the levels of this receptor are reduced in the islets of 12-month-old rats (Fig. 8a). PDGFRα is a predicted target of miR-34a (www.targetscan.org) and is regulated by this miRNA in cancer cells [32]. Although Pdgfra expression is upregulated in islet beta cells after DICER deletion and global miRNA suppression [33], its targeting by miR-34a in beta cells has not been explored. To experimentally verify PDGFRα as a target for miR-34a in these cells, INS832/13 cells were transfected with a luciferase construct containing the 3′ UTR of Pdgfra mRNA. Overexpression of miR-34a resulted in reduced luciferase activity, confirming that Pdgfra is indeed a direct target of miR-34a (Fig. 8b). Moreover, western blot analysis of rat islet cells overexpressing miR-34a confirmed that the miRNA reduces the levels of PDGFRα (Fig. 8c, d).
PDGFRα is a direct miR-34a target. (a) The levels of PDGFα in the islets of 3- and 12-month-old rats was determined by western blotting. The dot blot indicates the values of each of the tested rats (*p < 0.05 by Student’s t test). (b) INS832/13 cells were co-transfected with miR-34a mimic and with a reporter construct containing the 3′ UTR of PDGFRα. Luciferase activity was measured 2 days later. The results are expressed in relative light units (RLU) and are means ± SD (n = 3). *p < 0.05 vs control (Ctrl) by Student’s t test. (c, d) Dissociated rat islet cells were transfected with a control oligonucleotide or with the miR-34a mimic. Two days later, the levels of PDGFRα were analysed by western blotting. The figure shows a representative blot (c) and band quantification (d) from four independent experiments. Data are means ± SD. *p < 0.05 vs control
Discussion
Ageing is a risk factor for the development of type 2 diabetes. In fact, it is associated with a progressive decrease in insulin sensitivity and a decline in beta cell function [2]. The mechanisms underlying these effects are not fully understood, but are likely to involve changes in gene expression. Here, we investigated the transcriptomic modifications occurring in islets of 12-month-old rats. In agreement with other studies [34], we did not observe alterations in insulin secretion or apoptosis in the islets of these animals, suggesting that beta cells preserve the capacity to accomplish their basic tasks. However, beta cells from 12-month-old rats were unable to respond to mitogens. The loss of beta cell proliferation is probably not a significant handicap under normal conditions, but might constitute a major obstacle to the achievement of blood glucose homeostasis under conditions of insulin resistance requiring compensatory beta cell mass expansion.
Our data indicate that the expression of several protein-coding genes and miRNAs is already modified in the islets of 12-month-old rats. Some of these transcriptomic changes are likely to have a positive impact on beta cell function. For instance, we detected the upregulation of genes involved in insulin release, such as those coding for calcium-channel subunits [35–37] and the glucose transporter GLUT-2 (Slc2a2) [38]. These data suggest the existence of compensatory mechanisms operating to maintain efficient insulin secretion in 12-month-old animals.
In addition to differences in protein-coding genes, the islets of 12-month-old rats also displayed modifications in the miRNA profile. When changes in the levels of selected miRNAs were reproduced in islet cells of young animals, none of them affected insulin biosynthesis or secretion. In cell lines, overexpression of miR-124a and miR-34a has previously been reported to inhibit insulin secretion [25, 28, 39] but this observation was not confirmed in another study, although glucose-induced Ca2+ fluxes were affected by miR-124a overexpression [24]. These discrepant findings might be due to differences in the level of overexpression achieved after cell transfection. Moreover, because of transcriptomic differences, these miRNAs might have distinct impacts on insulin secretion in primary beta cells and tumoral beta cell lines. These miRNAs reduce the expression of key components of the machinery of insulin exocytosis [25, 28, 39]. Thus, although overexpression in beta cells of young animals did not significantly impair insulin release, we cannot exclude the possibility that the presence of higher levels of miR-34a or miR-124a would exacerbate the secretory decline of ageing beta cells.
Several of the miRNAs displaying expression changes in ageing animals affect the survival of beta cells. Indeed, downregulation of miR-130b or induction of miR-383 improved the survival of beta cells under proapoptotic conditions. Similar protective effects have already been reported for other miRNAs under insulin-resistance conditions [8, 9]. The changes in the levels of these miRNAs might be sufficient to compensate for the proapoptotic effect elicited by the concomitant upregulation of miR-34a. This suggests that a balance between the action of proapoptotic and antiapoptotic miRNAs prevents a net loss of beta cells in 12-month-old rats.
The most striking defect observed in beta cells of 12-month-old rats was the loss of proliferation in response to mitotic stimuli. Interestingly, overexpression of miR-34a or blockade of miR-181a was sufficient to reproduce this phenotypic trait in the islets of younger animals. The mechanism through which the downregulation of miR-181a contributes to the age-associated impairment of beta cell proliferation remains to be established. However, analysis of differentially expressed genes in the islets of 12-month-old rats revealed an enrichment of miR-181a targets in the upregulated mRNAs and a reduction in those that were downregulated. Thus, attenuation of the repressive activity of this miRNA appears to contribute to transcriptomic modifications occurring in the islets of ageing animals. The islet gene-expression profile of ageing animals also appears to be influenced by the induction of miR-34a. Indeed, the 3′ UTR of the upregulated genes was depleted from potential recognition sequences for this miRNA, and the putative miR-34a targets tended to be more frequent in genes downregulated in ageing. Some of the targets of miR-34a might be directly involved in the proliferative defect. Indeed, we demonstrated that the mRNA coding for PDGFRα is directly targeted by miR-34a. PDGFR signalling plays a critical role in postnatal beta cell mass expansion and in beta cell regeneration [30]. Thus, the translational repression of Pdgfra exerted by the induction of miR-34a is likely to contribute to the loss of proliferative capacity observed during ageing.
The increase in the level of miR-34a in the islets of ageing rats is probably linked to the activation of p53 signalling. Indeed, we have previously reported that, in insulin-secreting cells, this transcription factor binds to the promoter of miR-34a and triggers expression of the miRNA [40]. The p53 pathway is induced by a variety of stresses and plays a pivotal role in cellular senescence and metabolic homeostasis [41, 42]. p53 and miR-34a are linked in a positive-feedback loop to sirtuin-1 (SIRT1). Indeed, p53 induces the expression of miR-34a and the miRNA targets and represses SIRT1, preventing SIRT1-mediated deacetylation of p53 and, in turn, promoting the activity of the transcription factor [43]. Consistent with this model, we found that increases in p53 and miR-34a in the islets of ageing animals were indeed associated with reduced levels of SIRT1.
In addition to being upregulated in islets, the level of miR-34a was also elevated in the liver and brain of old rats, pointing to a general role for this miRNA in the ageing process. Moreover, the expression of this miRNA has been found to be abnormally elevated in islets and insulin target tissues of obese animals [40, 44, 45], contributing to beta cell failure and insulin resistance. Being at the crossroad between ageing and metabolic imbalance, these findings point to miR-34a induction as an important risk factor for the development of type 2 diabetes. This will particularly hold true for individuals expressing single nucleotide polymorphisms within the precursor of miR-34a that result in increased expression of this miRNA [46].
In this study, we identified a group of islet miRNAs that display expression changes during ageing and are likely to contribute to the progressive failure of beta cells to compensate for insulin resistance. A better understanding of the role and the mode of action of these miRNAs in beta cells might open the way to the development of new strategies for the prevention and/or treatment of type 2 diabetes.
Abbreviations
- miRNA:
-
MicroRNA
- Mobs :
-
Median number of miRNA recognition elements
- PDGF:
-
Platelet-derived growth factor
- PDGFRα:
-
Platelet-derived growth factor receptor α
- qPCR:
-
Quantitative real-time PCR
- SIRT1:
-
Sirtuin-1
- UTR:
-
Untranslated region
References
Prentki M, Nolan CJ (2006) Islet β cell failure in type 2 diabetes. J Clin Invest 116:1802–1812
Gong Z, Muzumdar RH (2012) Pancreatic function, type 2 diabetes, and metabolism in aging. Int J Endocrinol 2012:320482
Gunasekaran U, Gannon M (2011) Type 2 diabetes and the aging pancreatic beta cell. Aging (Albany NY) 3:565–575
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297
Dumortier O, Hinault C, van Obberghen E (2013) MicroRNAs and metabolism crosstalk in energy homeostasis. Cell Metab 18:312–324
Guay C, Jacovetti C, Nesca V, Motterle A, Tugay K, Regazzi R (2012) Emerging roles of non-coding RNAs in pancreatic β-cell function and dysfunction. Diabetes Obes Metab 14(Suppl 3):12–21
Esguerra JL, Eliasson L (2014) Functional implications of long non-coding RNAs in the pancreatic islets of Langerhans. Front Genet 5:209
Jacovetti C, Abderrahmani A, Parnaud G et al (2012) MicroRNAs contribute to compensatory β cell expansion during pregnancy and obesity. J Clin Invest 122:3541–3551
Nesca V, Guay C, Jacovetti C et al (2013) Identification of particular groups of microRNAs that positively or negatively impact on beta cell function in obese models of type 2 diabetes. Diabetologia 56:2203–2212
Poy MN, Hausser J, Trajkovski M et al (2009) miR-375 maintains normal pancreatic α- and β-cell mass. Proc Natl Acad Sci U S A 106:5813–5818
Tattikota SG, Rathjen T, McAnulty SJ et al (2014) Argonaute2 mediates compensatory expansion of the pancreatic βcell. Cell Metab 19:122–134
Gotoh M, Maki T, Satomi S et al (1987) Reproducible high yield of rat islets by stationary in vitro digestion following pancreatic ductal or portal venous collagenase injection. Transplantation 43:725–730
Locke JM, da Silva XG, Dawe HR, Rutter GA, Harries LW (2014) Increased expression of miR-187 in human islets from individuals with type 2 diabetes is associated with reduced glucose-stimulated insulin secretion. Diabetologia 57:122–128
Jacovetti C, Jimenez V, Ayuso E et al (2015) Contribution of Intronic miR-338-3p and its hosting gene AATK to compensatory β-cell mass expansion. Mol Endocrinol 29:693–702
Allagnat F, Martin D, Condorelli DF, Waeber G, Haefliger JA (2005) Glucose represses connexin36 in insulin-secreting cells. J Cell Sci 118:5335–5344
Garcia DM, Baek D, Shin C, Bell GW, Grimson A, Bartel DP (2011) Weak seed-pairing stability and high target-site abundance decrease the proficiency of lsy-6 and other microRNAs. Nat Struct Mol Biol 18:1139–1146
Karolchik D, Baertsch R, Diekhans M et al (2003) The UCSC genome browser database. Nucleic Acids Res 31:51–54
Wang SY, Halban PA, Rowe JW (1988) Effects of aging on insulin synthesis and secretion. Differential effects on preproinsulin messenger RNA levels, proinsulin biosynthesis, and secretion of newly made and preformed insulin in the rat. J Clin Invest 81:176–184
Matveyenko AV, Veldhuis JD, Butler PC (2008) Adaptations in pulsatile insulin secretion, hepatic insulin clearance, and β-cell mass to age-related insulin resistance in rats. Am J Physiol Endocrinol Metab 295:E832–E841
Sone H, Kagawa Y (2005) Pancreatic beta cell senescence contributes to the pathogenesis of type 2 diabetes in high-fat diet-induced diabetic mice. Diabetologia 48:58–67
Gorospe M, Abdelmohsen K (2011) MicroRegulators come of age in senescence. Trends Genet 27:233–241
Jung HJ, Suh Y (2012) MicroRNA in aging: from discovery to biology. Curr Genomics 13:548–557
Noren Hooten N, Abdelmohsen K, Gorospe M, Ejiogu N, Zonderman AB, Evans MK (2010) MicroRNA expression patterns reveal differential expression of target genes with age. PLoS One 5:e10724
Baroukh N, Ravier MA, Loder MK et al (2007) MicroRNA-124a regulates Foxa2 expression and intracellular signaling in pancreatic β-cell lines. J Biol Chem 282:19575–19588
Lovis P, Gattesco S, Regazzi R (2008) Regulation of the expression of components of the exocytotic machinery of insulin-secreting cells by microRNAs. Biol Chem 389:305–312
Liu N, Landreh M, Cao K et al (2012) The microRNA miR-34 modulates ageing and neurodegeneration in Drosophila. Nature 482:519–523
Li N, Muthusamy S, Liang R, Sarojini H, Wang E (2011) Increased expression of miR-34a and miR-93 in rat liver during aging, and their impact on the expression of Mgst1 and Sirt1. Mech Ageing Dev 132:75–85
Roggli E, Britan A, Gattesco S et al (2010) Involvement of microRNAs in the cytotoxic effects exerted by proinflammatory cytokines on pancreatic β-cells. Diabetes 59:978–986
Guay C, Regazzi R (2015) Role of islet microRNAs in diabetes: which model for which question? Diabetologia 58:456–463
Chen H, Gu X, Liu Y et al (2011) PDGF signalling controls age-dependent proliferation in pancreatic β-cells. Nature 478:349–355
Pullen TJ, Rutter GA (2013) When less is more: the forbidden fruits of gene repression in the adult β-cell. Diabetes Obes Metab 15:503–512
Kang HW, Crawford M, Fabbri M et al (2013) A mathematical model for microRNA in lung cancer. PLoS One 8:e53663
Martinez-Sanchez A, Nguyen-Tu MS, Rutter GA (2015) DICER inactivation identifies pancreatic β-cell "disallowed" genes targeted by microRNAs. Mol Endocrinol 29:1067–1079
Takahashi A, Motomura K, Kato T et al (2005) Transgenic mice overexpressing nuclear SREBP-1c in pancreatic β-cells. Diabetes 54:492–499
Dubois M, Vacher P, Roger B et al (2007) Glucotoxicity inhibits late steps of insulin exocytosis. Endocrinology 148:1605–1614
Gao N, White P, Doliba N, Golson ML, Matschinsky FM, Kaestner KH (2007) Foxa2 controls vesicle docking and insulin secretion in mature βcells. Cell Metab 6:267–279
Reinbothe TM, Alkayyali S, Ahlqvist E et al (2013) The human L-type calcium channel Cav1.3 regulates insulin release and polymorphisms in CACNA1D associate with type 2 diabetes. Diabetologia 56:340–349
McCulloch LJ, van de Bunt M, Braun M, Frayn KN, Clark A, Gloyn AL (2011) GLUT2 (SLC2A2) is not the principal glucose transporter in human pancreatic beta cells: implications for understanding genetic association signals at this locus. Mol Genet Metab 104:648–653
Sebastiani G, Po A, Miele E et al (2015) MicroRNA-124a is hyperexpressed in type 2 diabetic human pancreatic islets and negatively regulates insulin secretion. Acta Diabetol 52:523–530
Lovis P, Roggli E, Laybutt DR et al (2008) Alterations in microRNA expression contribute to fatty acid-induced pancreatic β-cell dysfunction. Diabetes 57:2728–2736
Campisi J (2005) Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 120:513–522
Berkers CR, Maddocks OD, Cheung EC, Mor I, Vousden KH (2013) Metabolic regulation by p53 family members. Cell Metab 18:617–633
Yamakuchi M, Lowenstein CJ (2009) MiR-34, SIRT1 and p53: the feedback loop. Cell Cycle 8:712–715
Zhao E, Keller MP, Rabaglia ME et al (2009) Obesity and genetics regulate microRNAs in islets, liver, and adipose of diabetic mice. Mamm Genome 20:476–485
Trajkovski M, Hausser J, Soutschek J et al (2011) MicroRNAs 103 and 107 regulate insulin sensitivity. Nature 474:649–653
Locke JM, Lango Allen H, Harries LW (2014) A rare SNP in pre-miR-34a is associated with increased levels of miR-34a in pancreatic beta cells. Acta Diabetol 51:325–329
Funding
This work was supported by the Swiss National Science Foundation (grant number 310030-146138) (RR) and by the ‘Fondation Francophone pour la Recherche sur le Diabète’ (RR). GAR was supported by a Wellcome Trust Senior Investigator Award (WT098424AIA), an MRC Programme Grant (MR/J0003042/1) and a Royal Society Wolfson Research Merit Award. LWH and JML were supported by a Wellcome Trust project grant (number 089845/Z/09/Z).
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Contribution statement
KT generated the data and wrote the manuscript. CG and FA contributed to the acquisition of data and critically reviewed the manuscript. LWH, JML, ACM and GAR contributed to data acquisition and interpretation, and reviewed the manuscript. RR conceived the experiments, interpreted the data and wrote the manuscript. All authors approved the final version. RR is the guarantor of this work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Methods
(PDF 163 kb)
ESM Fig. 1
(PDF 78 kb)
ESM Fig. 2
(PDF 83 kb)
ESM Fig. 3
(PDF 11 kb)
ESM Fig. 4
(PDF 85 kb)
ESM Fig. 5
(PDF 80 kb)
ESM Table 1
(PDF 2147 kb)
ESM Table 2
(PDF 1457 kb)
ESM Table 3
(PDF 121 kb)
ESM Table 4
(PDF 1047 kb)
ESM Table 5
(PDF 349 kb)
ESM Table 6
(PDF 303 kb)
ESM Table 7
(PDF 237 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Tugay, K., Guay, C., Marques, A.C. et al. Role of microRNAs in the age-associated decline of pancreatic beta cell function in rat islets. Diabetologia 59, 161–169 (2016). https://doi.org/10.1007/s00125-015-3783-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-015-3783-5