Abstract
Aims/hypothesis
Although insulin resistance induces compensatory increases in beta cell mass and function to maintain normoglycaemia, it is not clear whether insulin resistance can precipitate beta cell dysfunction and hyperglycaemia without a pre-existing beta cell susceptibility. We therefore examined the beta cell phenotype in the MKR mouse, a model in which expression of a dominant-negative IGF 1 receptor (IGF1R) in skeletal muscle leads to systemic insulin resistance and diabetes.
Materials and methods
Circulating glucose, insulin and glucagon concentrations were measured. Insulin sensitivity, glucose tolerance and insulin release in vivo were assessed by i.p. insulin and glucose tolerance tests. Beta cell function was assessed via insulin secretion from isolated islets and the glucose gradient in the perfused pancreas. Beta cell morphology was examined via immunohistochemistry. MKR mice were fed a high-fat diet containing sucrose (HFSD) to test metabolic capacity and beta cell function.
Results
Insulin-resistant MKR mice developed hyperglycaemia and a loss of insulin responsiveness in vivo. Basal insulin secretion from the perfused pancreas was elevated, with no response to glucose. Despite the demand on insulin secretion, MKR mice had increased pancreatic insulin content and beta cell mass mediated through hyperplasia and hypertrophy. The HFSD worsened hyperglycaemia in MKR mice but, despite increased food intake in these mice, failed to induce the obesity observed in wild-type mice.
Conclusions/interpretation
Our studies demonstrate that insulin resistance of sufficient severity can impair glucose-stimulated insulin secretion, thereby undermining beta cell compensation and leading to hyperglycaemia. Moreover, because insulin stores were intact, the secretory defects reflect an early stage of beta cell dysfunction.
Similar content being viewed by others
Introduction
A feedback loop exists between insulin sensitivity and insulin secretion such that changes in sensitivity are accompanied by inverse changes in secretion, resulting in the maintenance of euglycaemia [1]. Insulin resistance exemplifies this relationship because it induces increases in insulin secretion in both rodents and humans, preventing the majority of insulin-resistant individuals from progressing to type 2 diabetes mellitus [2–5]. Rodent models have shown that increases in both beta cell mass and function mediate the compensatory increase in secretion. For example, diabetes does not occur in irs1 −/− mice, despite insulin resistance, due to compensatory increases in beta cell mass and insulin secretion [4, 6]. Similar compensatory changes in beta cell mass and secretion also appear to occur in humans [1, 5, 7, 8].
When beta cell compensation is inadequate for the degree of insulin resistance, progression to diabetes eventually occurs [5, 9]. Although insulin resistance and beta cell dysfunction are present in type 2 diabetes, the nature of the interaction between the two abnormalities is unclear [1, 10, 11]. Since it occurs before the onset of hyperglycaemia, insulin resistance has been proposed to contribute to the development of beta cell dysfunction, and models have therefore been generated to examine the contribution of tissue-specific insulin resistance to the development of diabetes [5, 12–18]. However, mice without muscle-specific insulin receptor (INSR, also known as IR) gene expression are normoglycaemic and develop only mild systemic insulin resistance, probably due in part to compensatory pathways of muscle glucose uptake, including the IGF 1 receptor (IGF1R) pathway. To our knowledge, no muscle-specific Igf1 −/− model exists, and whole-body Igf1r −/− mice die in the immediate post-natal period due to respiratory failure, rendering them unfeasible for long-term studies. The MKR mouse is therefore an excellent model to study the beta cell response to insulin resistance because muscle insulin resistance has been generated through functional inactivation of the INSR and IGF1R pathways [19]. Specifically, a muscle-specific, dominant-negative IGF1R hybridises with the endogenous monomers of the IGF1R and INSR, giving rise to insulin resistance [19]. Insulin resistance also develops in the liver and adipose tissue, and hyperglycaemia occurs at approximately 5–6 weeks of age [19]. Our present study therefore focused on characterising the beta cell phenotype in MKR mice.
Materials and methods
Animals
Mice had free access to water and food (diet no. 8664; Harlan Tekland, Madison, WI, USA), and were maintained on a 12-h light/dark cycle. For the high-fat-sucrose diet (HFSD) study, the diet was composed of 58% kcal fat and 12.6% kcal sucrose for 18 weeks beginning at 5 weeks of age (diet no. D12331; Research Diets, New Brunswick, NJ, USA). Food intake was measured from 10 to 16 weeks on the HFSD. MKR mice were genotyped by PCR analysis of tail DNA [20]. All studies were performed on male mice using wild-type (WT) male FVB/NJ mice (Charles River, Wilmington, MA, USA) as controls. Animal care procedures were in accordance with the standards of the Canadian Council on Animal Care and approved by the Animal Care and Use Committee at the University of Toronto.
Immunoblot analysis
Tissues were lysed manually in buffer [21]. Proteins were resolved by 10% SDS-PAGE, transferred to polyvinylidene difluoride membranes and probed for the beta-subunit of the IGF1R or INSR (sc-713 and sc-711 respectively; Santa Cruz Biotechnology, Santa Cruz, CA, USA). Protein loading was normalised to beta-actin (Sigma Canada, Oakville, ON, Canada). The chemiluminescence (Amersham Pharmacia Biotech, Buckinghamshire, UK) signals were quantified using Scion Image 1.63 (Scion Corporation, Frederick, MD, USA).
Analytical procedures, glucose and insulin tolerance tests
Blood glucose was measured between 8:00 and 11:00 hours on tail vein blood using a glucometer (Bayer, Toronto, ON, Canada). Insulin and glucagon were measured by RIA of the plasma component of blood collected from the saphenous vein [22]. Intraperitoneal insulin (1 U/kg) and glucose (1.5 g/kg) tolerance tests were performed after 3- and 16-h fasts, respectively, and blood glucose was measured at the times indicated. For insulin release during glucose tolerance testing, the plasma component of blood collected from the tail vein at the times indicated was assayed by insulin ELISA (Crystal Chem, Chicago, IL, USA).
Islet secretion studies
Approximately 2.5–3 ml of RPMI 1640 medium containing types V and XI collagenase (1.5 and 0.7 mg/ml, respectively; Sigma, St Louis, MO, USA), 5.6 mmol/l glucose, 100 U/ml penicillin, and 100 μg/ml streptomycin were perfused through the bile duct. The pancreas was then isolated and incubated at 37°C for 8–10 min with gentle shaking. Islets were hand-picked and cultured overnight prior to the secretion studies. Insulin secretion was performed on groups of ten islets using KRB solution containing the specified glucose concentrations. The islets were subjected to acid–ethanol extraction for measurement of DNA and insulin content using RIA [22, 23].
Pancreas perfusion
We used a modified version of a pancreas perfusion protocol that has been previously described [20, 22, 24]. After 20 min of stabilising perfusion with 1.4 mmol/l glucose, the pancreas was perfused with a linear gradient from 1.4 mmol/l glucose to 20 mmol/l (over 50 min) or 30 mmol/l glucose (over 30 min). The pancreas was subsequently perfused with 20 mmol/l l-arginine and 40 mmol/l glucose combined for 10 min. The perfusate was assayed by RIA [22].
Pancreatic insulin content and islet morphology
Pancreatic insulin content was determined as previously described [25]. Mice were injected with 5-bromo-2′-deoxyuridine (BrdU; 100 mg/kg) 5–6 h before being killed. Tissue processing, and immunostaining for insulin and BrdU have been previously described [20, 24]. The individual beta cell cross-sectional area was calculated by dividing insulin-positive areas by the number of nuclei within each area. Beta cell density was calculated as the average number of nuclei within each insulin-positive area. Each pancreas was considered as a single n value. For the HFSD study, measurements were performed at ×10 magnification on islets isolated for the secretion studies.
Statistical analysis
The data are expressed as means±SEM. Significance was determined by Student’s t-test or one-way ANOVA with Tukey–Kramer or Dunn’s multiple comparisons post test where appropriate. A value of p<0.05 was considered significant.
Results
Dominant-negative IGF1R expression
Immunoblot analysis of the IGF1R revealed increased expression in MKR quadriceps muscle, demonstrating expression of the dominant-negative IGF1R (Fig. 1a). The IGF1R antibody used detects both dominant-negative and endogenous forms, and transgene detection consequently relies on detecting IGF1R overexpression. Expression was also increased in the heart, which was not surprising, given that the muscle creatinine kinase promoter is active in this tissue, and expression in MKR heart tissue has been previously reported [19]. Due to the possibility of receptor downregulation, either as a consequence or mediator of insulin resistance, hepatic and islet IGF1R levels were examined, as was INSR production in quadriceps, islet, liver and heart tissue. No differences were found (Fig. 1a,b).
Glucose homeostasis in MKR mice
Overt hyperglycaemia occurred at 6 weeks in MKR mice, although a slight elevation was observed at 3 weeks (Fig. 2a). The degree of hyperglycaemia declined at 8 weeks but was still significantly greater than in WT mice up to 11 weeks, a difference that remained apparent at 31 weeks of age (Fig. 2a). Hyperinsulinaemia was present as early as 3 weeks, and increased progressively with age (Fig. 2b). Fed glucagon levels were also significantly elevated at 8 weeks (Fig. 2c). We confirmed the presence of insulin resistance that was suggested by hyperinsulinaemia in MKR mice by conducting insulin tolerance tests, during which glucose levels failed to decrease in response to exogenous insulin (Fig. 3a,b). To gauge beta cell function, glucose tolerance tests were also performed. IGT was already present at 3 weeks in MKR mice, and worsened by 12 weeks (Fig. 3c,d). Glucose tolerance also deteriorated with age in WT mice (p<0.05 for WT 3 vs 12 weeks at all time points except 90 min; Fig. 3c,d). Despite IGT, the acute-phase response to glucose was still intact in 3-week-old mice (Fig. 3e). In contrast, after the onset of hyperglycaemia at 12 weeks, the acute-phase response was lost in MKR mice, demonstrating the presence of beta cell dysfunction (Fig. 3f).
Glucose homeostasis in MKR and wild-type (WT) mice. Intraperitoneal insulin tolerance tests (1 U/kg insulin) in a 3-week- and b 12-week-old mice (n=6). c, d Intraperitoneal glucose tolerance tests (1.5 g/kg glucose) in 3-week- and 13-week-old mice, respectively; n=5–6 except at 120 min for (c) where n=3. e, f Insulin release during intraperitoneal glucose tolerance tests in 3-week- and 12-week-old mice, respectively; n=8–14. Trianges WT, squares MKR. *p<0.05, **p<0.01, ***p<0.001 vs other group
Insulin secretion from the perfused pancreas
To explore the possibility that altered beta cell glucose sensitivity underlies the insulin secretory dysfunction in MKR mice, pancreata from 12- to 14-week-old mice were perfused with a linear glucose gradient ranging from 1.4 to 20 mmol/l at a rate of ∼0.37 mmol l−1 min−1 (Fig. 4a). Insulin secretion in MKR mice was elevated at low glucose concentrations and did not increase with the glucose concentration, demonstrating a lack of response (Fig. 4a). In contrast, insulin secretion gradually increased in WT mice, and due to this increase, no significant difference in insulin levels was observed between MKR and WT at higher glucose concentrations (Fig. 4a). Increasing the perfusion rate to ∼0.95 mmol l−1 min−1 and the maximum concentration to 30 mmol/l failed to elicit a response from MKR pancreata (Fig. 4b).
Insulin secretion from perfused pancreata of MKR and wild-type (WT) mice. Insulin secretion from 12-week-old mice perfused in situ at a 0.37 mmol l−1 min−1 from 1.4 to 20 mmol/l glucose (n=9–10) and b 0.95 mmol l−1 min−1 from 1.4 to 30 mmol/l glucose (n=7). At the end of the gradient (ESM Fig. 1), the pancreas was stimulated with glucose (40 mmol/l) and arginine (20 mmol/l). Triangles WT, squares MKR
Characterisation of isolated islets
Similar to the perfused pancreas, basal insulin secretion at 2.8 mmol/l was higher in isolated MKR islets than in WT islets (0.80±0.16 vs 0.27±0.08 ng/ml, respectively, p<0.05, n=5). Although a dose-dependent increase in insulin secretion was observed in WT islets, the response of MKR islets was significantly reduced at stimulatory glucose concentrations (Fig. 5a). The reduction in glucose-stimulated insulin secretion (GSIS) was not due to decreased islet insulin content, which was significantly greater in MKR mice (Fig. 5b). When islet insulin content was corrected for cell number by normalisation to DNA, islet insulin content was comparable to WT islets because DNA content was significantly higher in MKR islets (Fig. 5c,d).
Isolated islet parameters in 12-week-old MKR and wild-type (WT) mice. a Glucose-stimulated insulin secretion from isolated islets (n=3 independent experiments with ≥5 mice per genotype). b Islet insulin content (n=5 animals). c Insulin content normalised to DNA (n=5 animals). d Islet DNA content (n=5 animals). **p<0.01, ***p<0.001 vs WT
Pancreatic insulin content and islet morphology
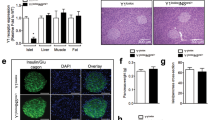
Consistent with the increase in islet insulin content (Fig. 5b), pancreatic insulin content increased with age in MKR mice (Fig. 6c), demonstrating that insulin stores were preserved despite the metabolic abnormalities in these mice. Beta cell mass was significantly increased in MKR mice, and beta cell proliferation, beta cell density and beta cell size were enhanced, consistent with the increase in islet DNA content (Figs. 5d, 6a,d–g). These findings demonstrated the presence of beta cell hypertrophy and hyperplasia in MKR mice. An increase in the proportion of large islets and a corresponding decrease in the number of smaller islets were also observed (Fig. 6h). Glucagon staining appeared normal, with the staining restricted to the islet periphery (Fig. 6b).
Pancreatic insulin content and islet morphology. Immunostaining for a insulin (×10 magnification) and b glucagon (×4 magnification) in pancreatic sections from wild-type (WT) and MKR mice. Black-stained nuclei are positive for BrdU (arrows). c Pancreatic insulin content (n=5–6). d–h Beta cell mass, beta cell proliferation, beta cell density, individual beta cell size, and islet mass distribution at 8–10 weeks, respectively (n=7–9 animals). *p<0.05, **p<0.01, ***p<0.001 vs WT; ## p<0.01 vs MKR at 3 weeks
Effects of a high-fat-sucrose diet
To examine the role of glucolipotoxicity on insulin resistance and beta cell dysfunction, WT and MKR mice were placed on an HFSD for 18 weeks. This study situation more accurately reflects human type 2 diabetes mellitus, in which genetics and environment contribute to disease pathogenesis. Surprisingly, MKR mice on the HFSD (MKR+HFSD) did not gain weight compared to control MKR mice, whereas WT+HFSD mice demonstrated significant weight gain and considerable visceral adiposity (Fig. 7a; Electronic Supplementary Material [ESM] Fig. 1). Food intake was significantly greater in these mice than in WT+HFSD, demonstrating that the absence of obesity in MKR+HFSD was not the result of decreased food intake (Table 1). Normoglycaemia was maintained in WT+HFSD mice, in which the diet did not alter circulating insulin levels (Fig. 7b,c), although there was a trend towards an increase at 18 weeks on the diet. However, the HFSD worsened insulin sensitivity in WT+HFSD mice such that at 18 weeks there was no difference between WT and MKR mice on the HFSD, although a difference had initially been observed (Fig. 8a,b). The HFSD did not worsen insulin resistance in MKR mice (Fig. 8a,b), and correspondingly, no change in insulin levels was observed (Fig. 7c). However, the severity of hyperglycaemia increased and the GSIS was reduced in MKR+HFSD mice (Figs. 7b, 8e). The HFSD had no significant effect on either glucose tolerance (Fig. 8d) or islet area (Fig. 8f).
In vivo parameters of MKR and wild-type (WT) mice on a high-fat-sucrose diet (HFSD). Mice were placed on the diet at 5 weeks of age. Open triangles WT, filled triangles WT+HFSD, open squares MKR, filled squares MKR+HFSD. a Body weight (n=7–20). Significant differences were seen within the HFSD and control groups, except between the control groups at 18 weeks on the diet. b Blood glucose (n=7–20). Significant differences were seen between the HFSD groups, except at 11 weeks on the diet. c Plasma insulin for the HFSD (n=6–15). # p<0.05, ## p<0.01, ### p<0.001 within genotypes; **p<0.01, ***p<0.001 vs WT on the same diet
Glucose homeostasis in MKR and wild-type (WT) mice on a high-fat-sucrose diet (HFSD). Mice were placed on the diet at 5 weeks of age. Intraperitoneal insulin tolerance tests after a 4 weeks (n=7) and b 18 weeks (n=4–5) on the HFSD. c, d Glucose tolerance after 4 weeks (n=8–11) and 18 weeks (n=5–13), respectively on the HFSD. e Insulin secretion from isolated islets in response to 11 mmol/l glucose (n=3 independent experiments with three animals per group) and f islet area (n=24–28 islets) after 18 weeks on the HFSD. Open triangles WT, filled triangles WT+HFSD, open squares MKR, filled squares MKR+HFSD. aa p<0.01, aaa p<0.001 between control groups; b p<0.05, bb p<0.01, bbb p<0.001 between HFSD groups; c p<0.05 vs. WT
Discussion
The MKR mouse is a model of insulin resistance and type 2 diabetes, in which the primary defect is muscle insulin resistance [19]. This led to systemic insulin resistance which was unaffected by the HFSD (Figs. 3a,b and 8a,b). The absence of a further reduction in insulin sensitivity was probably due to the resistance of MKR mice to HFSD-induced obesity (Fig. 7a), potentially a consequence of resistance to the weight-gain-inducing effects of insulin, due to the insulin resistance already present. The lack of obesity was not due to decreased food intake, which was increased in HFSD-fed MKR mice (Table 1). An increase in caloric intake in association with decreased body weight was also observed in MKR mice on a normal diet (Table 1), consistent with previous findings [26]. The precise mechanisms responsible for these findings are unclear, although factors including increased metabolic and physical activity may contribute [20]. The lower body weight in MKR mice is due to a reduction in lean body mass, and changes in adipose tissue mass, ranging from a slight increase to non-significant change, have been reported [26, 27].
In association with hyperglycaemia, a worsening of IGT and defects in insulin secretion were observed in MKR mice. Specifically, insulin secretion was elevated at basal glucose concentrations (Fig. 4), the acute insulin response to glucose was absent (Fig. 3f), a progressive increase in the concentration of glucose failed to elicit a response from the perfused pancreas (Fig. 4), and the isolated islet response to glucose was noticeably blunted (Fig. 5a). The absence of a response from the perfused pancreas suggested a loss of glucose sensitivity. Although a square wave of hyperglycaemia stimulated a blunted, monophasic response in previous pancreas perfusion studies in MKR mice [20, 24], such an abrupt rise in glucose does not occur physiologically, and the glucose gradient therefore better represents in vivo glucose dynamics. A further decrease in the response to glucose was observed in MKR+HFSD islets, and could be responsible for the exacerbation of hyperglycaemia in HFSD-fed MKR mice (Figs. 7b, 8e). A trend towards a reduction in GSIS was also found in WT+HFSD islets (Fig. 8e). These findings not only clearly demonstrate the presence of beta cell dysfunction in MKR mice, they also reveal that insulin resistance of sufficient severity, originating in muscle, can precipitate beta cell dysfunction-a novel finding given that other models of muscle-specific insulin resistance have normal or only slightly impaired glucose homeostasis [14, 15, 28]. The GSIS abnormalities in MKR mice were not due to alterations in the ability to secrete insulin, given that the perfused pancreas response to arginine was increased, which demonstrates that the secretory capacity is greater in MKR mice [20, 24]. Hyperglucagonaemia was also observed after the onset of hyperglycaemia (Fig. 2c), consistent with the findings of chronic hyperglucagonaemia in type 2 diabetes [29]. This suggested that dysregulated glucagon secretion may also contribute to the onset of hyperglycaemia in MKR mice.
Beta cell dysfunction occurred in MKR mice despite compensatory increases in insulin content and beta cell mass mediated by hyperplasia and hypertrophy (Figs. 3e,f, 4, 5, 6). These findings are consistent with the compensatory increases in beta cell mass observed in states of insulin resistance [3, 17], and the lack of change in MKR+HFSD islet mass may well be due to the absence of a further increase in insulin resistance (Fig. 8b,f). Moreover, the elevation in pancreatic insulin content was substantially increased at 31 weeks as were circulating insulin levels (Figs. 2b, 6c), demonstrating the persistence of beta cell compensation with age and indicating that the onset of hyperglycaemia in MKR mice is not due to a low potential of the FVB/NJ strain for beta cell compensation. Similarly, although transfer of the Lepr db mutation to the FVB/NJ background results in hyperglycaemia, these mice nonetheless maintain insulin secretion and exhibit an enormous increase in beta cell number [30].
Hypertrophy and hyperplasia also mediate beta cell mass expansion in the Zucker Diabetic Fatty (ZDF) rat model of type 2 diabetes, leading to increased GSIS. However, when normalised for the expansion in mass, secretion in the ZDF rat is actually decreased per unit of beta cell mass, demonstrating beta cell dysfunction [2]. Moreover, this highlights the possibility that hypertrophy and an increased rate of proliferation may paradoxically contribute to beta cell dysfunction. For instance, a rapid rate of proliferation induced by beta-cell-specific overexpression of c-Myc, a transcription factor essential for cell cycle progression, results in an undifferentiated phenotype and severe hyperglycaemia in the neonatal period [31]. Additionally, beta cell hypertrophy is associated with beta cell dedifferentiation, including increased c-Myc production, in the 90% pancreatectomy model of diabetes [32, 33]. However, phloridzin-mediated normalisation of hyperglycaemia in this model also normalised beta cell hypertrophy and improved the derangements in gene expression, suggesting that chronic hyperglycaemia, as opposed to hypertrophy in itself, was primarily responsible for the dedifferentiation, and mediated its effects through mechanisms both dependent and independent of hypertrophy [32]. Therefore, while it is possible that hypertrophy and proliferation could contribute to beta cell dysfunction in MKR mice, it also seems likely that extrinsic factors may be predominantly responsible.
Consequently, the precise mechanisms underlying beta cell dysfunction in MKR mice are unclear. Given that no known genetic beta cell defect exists in these mice, it can be assumed that insulin resistance initiates a sequence of events, leading to beta cell dysfunction and the onset of hyperglycaemia. The glucolipotoxicity theory proposes that simultaneous elevation of glucose and lipids results in intracellular accumulation of lipids and lipid metabolites, which are ultimately detrimental to beta cell function and survival [34]. Thus, lowering either glucose or lipids should improve beta cell function [35]. Improving lipid metabolism by activation of peroxisome proliferator-activated receptor alpha in MKR mice normalised hyperglycaemia and insulin secretion, suggesting that lipotoxicity or glucolipotoxicity is responsible for the beta cell dysfunction in this model [24]. Alternatively, the involvement of insulin signalling in beta cell function suggests hyperinsulinaemia may contribute to MKR beta cell dysfunction because circulating insulin levels remained elevated and IGT persists in phloridzin-treated MKR mice [9, 26, 36–38].
In conclusion, severe insulin resistance, secondary to defects in muscle insulin/IGF1R signalling, leads to beta cell dysfunction in the MKR model, undermining beta cell compensation and resulting in the development of diabetes. Our studies suggest that functional defects as opposed to reduced beta cell mass may underlie some forms of diabetes. Moreover, they provide insight into the possible role of insulin resistance in the development of beta cell dysfunction.
Abbreviations
- BrdU:
-
5-bromo-2′-deoxyuridine
- ESM:
-
Electronic Supplementary Material
- GSIS:
-
glucose-stimulated insulin secretion
- HFSD:
-
high-fat sucrose diet
- IGF1R:
-
IGF 1 receptor
- INSR:
-
insulin receptor
- WT:
-
wild-type
- ZDF:
-
Zucker Diabetic Fatty
References
Kahn SE (2003) The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of Type 2 diabetes. Diabetologia 46:3–19
Pick A, Clark J, Kubstrup C et al (1998) Role of apoptosis in failure of beta-cell mass compensation for insulin resistance and beta-cell defects in the male Zucker diabetic fatty rat. Diabetes 47:358–364
Bruning JC, Winnay J, Bonner-Weir S, Taylor SI, Accili D, Kahn CR (1997) Development of a novel polygenic model of NIDDM in mice heterozygous for IR and IRS-1 null alleles. Cell 88:561–572
Kubota N, Tobe K, Terauchi Y et al (2000) Disruption of insulin receptor substrate 2 causes type 2 diabetes because of liver insulin resistance and lack of compensatory beta-cell hyperplasia. Diabetes 49:1880–1889
Weyer C, Bogardus C, Mott DM, Pratley RE (1999) The natural history of insulin secretory dysfunction and insulin resistance in the pathogenesis of type 2 diabetes mellitus. J Clin Invest 104:787–794
Araki E, Lipes MA, Patti ME et al (1994) Alternative pathway of insulin signalling in mice with targeted disruption of the IRS-1 gene. Nature 372:186–190
Butler AE, Janson J, Bonner-Weir S, Ritzel R, Rizza RA, Butler PC (2003) Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes 52:102–110
Kloppel G, Lohr M, Habich K, Oberholzer M, Heitz PU (1985) Islet pathology and the pathogenesis of type 1 and type 2 diabetes mellitus revisited. Surv Synth Pathol Res 4:110–125
Withers DJ, Gutierrez JS, Towery H et al (1998) Disruption of IRS-2 causes type 2 diabetes in mice. Nature 391:900–904
Gerich JE (1999) Is insulin resistance the principal cause of type 2 diabetes? Diabetes Obes Metab 1:257–263
Kahn SE (2001) Clinical review 135: The importance of beta-cell failure in the development and progression of type 2 diabetes. J Clin Endocrinol Metab 86:4047–4058
Lillioja S, Mott DM, Howard BV et al (1988) Impaired glucose tolerance as a disorder of insulin action. Longitudinal and cross-sectional studies in Pima Indians. N Engl J Med 318:1217–1225
Lillioja S, Mott DM, Spraul M et al (1993) Insulin resistance and insulin secretory dysfunction as precursors of non-insulin-dependent diabetes mellitus. Prospective studies of Pima Indians. N Engl J Med 329:1988–1992
Chang PY, Benecke H, Marchand-Brustel Y, Lawitts J, Moller DE (1994) Expression of a dominant-negative mutant human insulin receptor in the muscle of transgenic mice. J Biol Chem 269:16034–16040
Bruning JC, Michael MD, Winnay JN et al (1998) A muscle-specific insulin receptor knockout exhibits features of the metabolic syndrome of NIDDM without altering glucose tolerance. Mol Cell 2:559–569
Bruning JC, Gautam D, Burks DJ et al (2000) Role of brain insulin receptor in control of body weight and reproduction. Science 289:2122–2125
Michael MD, Kulkarni RN, Postic C et al (2000) Loss of insulin signaling in hepatocytes leads to severe insulin resistance and progressive hepatic dysfunction. Mol Cell 6:87–97
Bluher M, Michael MD, Peroni OD et al (2002) Adipose tissue selective insulin receptor knockout protects against obesity and obesity-related glucose intolerance. Dev Cell 3:25–38
Fernandez AM, Kim JK, Yakar S et al (2001) Functional inactivation of the IGF-I and insulin receptors in skeletal muscle causes type 2 diabetes. Genes Dev 15:1926–1934
Heron-Milhavet L, Haluzik M, Yakar S et al (2004) Muscle-specific overexpression of CD36 reverses the insulin resistance and diabetes of MKR mice. Endocrinology 145:4667–4676
Wang X, Li H, De Leo D et al (2004) Gene and protein kinase expression profiling of reactive oxygen species-associated lipotoxicity in the pancreatic beta-cell line MIN6. Diabetes 53:129–140
Joseph JW, Koshkin V, Zhang CY et al (2002) Uncoupling protein 2 knockout mice have enhanced insulin secretory capacity after a high-fat diet. Diabetes 51:3211–3219
Roduit R, Masiello P, Wang SP, Li H, Mitchell GA, Prentki M (2001) A role for hormone-sensitive lipase in glucose-stimulated insulin secretion: a study in hormone-sensitive lipase-deficient mice. Diabetes 50:1970–1975
Kim H, Haluzik M, Asghar Z et al (2003) Peroxisome proliferator-activated receptor-alpha agonist treatment in a transgenic model of type 2 diabetes reverses the lipotoxic state and improves glucose homeostasis. Diabetes 52:1770–1778
Joseph JW, Kalitsky J, St Pierre S, Brubaker PL (2000) Oral delivery of glucagon-like peptide-1 in a modified polymer preparation normalizes basal glycaemia in diabetic db/db mice. Diabetologia 43:1319–1328
Zhao H, Yakar S, Gavrilova O et al (2004) Phloridzin improves hyperglycemia but not hepatic insulin resistance in a transgenic mouse model of type 2 diabetes. Diabetes 53:2901–2909
Toyoshima Y, Gavrilova O, Yakar S et al (2005) Leptin improves insulin resistance and hyperglycemia in a mouse model of type 2 diabetes. Endocrinology 146:4024–4035
Kim JK, Michael MD, Previs SF et al (2000) Redistribution of substrates to adipose tissue promotes obesity in mice with selective insulin resistance in muscle. J Clin Invest 105:1791–1797
Jiang G, Zhang BB (2003) Glucagon and regulation of glucose metabolism. Am J Physiol Endocrinol Metab 284:E671–E678
Chua S Jr, Liu SM, Li Q, Yang L, Thassanapaff VT, Fisher P (2002) Differential beta cell responses to hyperglycaemia and insulin resistance in two novel congenic strains of diabetes (FVB-Lepr (db)) and obese (DBA-Lep (ob)) mice. Diabetologia 45:976–990
Laybutt DR, Weir GC, Kaneto H et al (2002) Overexpression of c-Myc in beta-cells of transgenic mice causes proliferation and apoptosis, downregulation of insulin gene expression, and diabetes. Diabetes 51:1793–1804
Jonas JC, Sharma A, Hasenkamp W et al (1999) Chronic hyperglycemia triggers loss of pancreatic beta cell differentiation in an animal model of diabetes. J Biol Chem 274:14112–14121
Laybutt DR, Glandt M, Xu G et al (2003) Critical reduction in beta-cell mass results in two distinct outcomes over time. Adaptation with impaired glucose tolerance or decompensated diabetes. J Biol Chem 278:2997–3005
Ruderman N, Prentki M (2004) AMP kinase and malonyl-CoA: targets for therapy of the metabolic syndrome. Nat Rev Drug Discov 3:340–351
Prentki M, Joly E, El Assaad W, Roduit R (2002) Malonyl-CoA signaling, lipid partitioning, and glucolipotoxicity: role in beta-cell adaptation and failure in the etiology of diabetes. Diabetes 51(Suppl 3):S405–S413
Kulkarni RN, Bruning JC, Winnay JN, Postic C, Magnuson MA, Kahn CR (1999) Tissue-specific knockout of the insulin receptor in pancreatic beta cells creates an insulin secretory defect similar to that in type 2 diabetes. Cell 96:329–339
Kulkarni RN, Winnay JN, Daniels M et al (1999) Altered function of insulin receptor substrate-1-deficient mouse islets and cultured beta-cell lines. J Clin Invest 104:R69–R75
Leibiger IB, Leibiger B, Berggren PO (2002) Insulin feedback action on pancreatic beta-cell function. FEBS Lett 532 2002:1–6
Acknowledgements
This work was supported by grants from the Canadian Diabetes Association to M. B. Wheeler, the Canadian Institutes of Health Research (CIHR) to M. B. Wheeler and C. B. Chan (MOP 12898), and the American Diabetes Association to D. LeRoith. M. B. Wheeler was supported by a CIHR Investigator Award and D. Yau was supported by a Banting and Best Diabetes Centre-Novo Nordisk Studentship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Asghar and Chan contributed equally to this work.
An erratum to this article can be found at http://dx.doi.org/10.1007/s00125-006-0157-z
Electronic supplementary materials
Fig. S1
Representative images of visceral fat in WT and MKR mice (as indicated) on an HFSD (GIF 81 kb)
Fig. S2
Glucose-stimulated insulin secretion during glucose gradient perfusion of the pancreas in situ in MKR and wild-type (WT) mice. Infusion of 1.4 to 30 mmol/l glucose was at a rate of 0.95 mmol−1 min−1 (n=7). The pancreas was stimulated with glucose (40 mmol/l) and arginine (20 mmol/l) at the end of the gradient. Triangles WT, squares MKR (GIF 49 kb)
Rights and permissions
About this article
Cite this article
Asghar, Z., Yau, D., Chan, F. et al. Insulin resistance causes increased beta-cell mass but defective glucose-stimulated insulin secretion in a murine model of type 2 diabetes. Diabetologia 49, 90–99 (2006). https://doi.org/10.1007/s00125-005-0045-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-005-0045-y