Abstract
Key message
Two QTLs with major effects on rolled leaf trait were consistently detected on chromosomes 1A (QRl.hwwg-1AS) and 5A (QRl.hwwg-5AL) in the field experiments.
Abstract
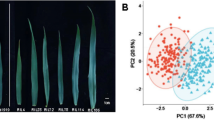
Rolled leaf (RL) is a morphological strategy to protect plants from dehydration under stressed field conditions. Identification of quantitative trait loci (QTLs) underlining RL is essential to breed drought-tolerant wheat cultivars. A mapping population of 154 recombinant inbred lines was developed from the cross between JagMut1095, a mutant of Jagger, and Jagger to identify quantitative trait loci (QTLs) for the RL trait. A linkage map of 3106 cM was constructed with 1003 unique SNPs from 21 wheat chromosomes. Two consistent QTLs were identified for RL on chromosomes 1A (QRl.hwwg-1AS) and 5A (QRl.hwwg-5AL) in all field experiments. QRl.hwwg-1AS explained 24–56% of the phenotypic variation and QRl.hwwg-5AL explained up to 20% of the phenotypic variation. The combined percent phenotypic variation associated with the two QTLs was up to 61%. Analyses of phenotypic and genotypic data of recombinants generated from heterogeneous inbred families of JagMut1095 × Jagger delimited QRl.hwwg-1AS to a 6.04 Mb physical interval. This work lays solid foundation for further fine mapping and map-based cloning of QRl.hwwg-1AS.



Similar content being viewed by others
Data availability
All the data are included in the manuscript and the supplementary information.
References
Bai G, Kolb FL, Shaner G, Domier LL (1999) Amplified fragment length polymorphism markers linked to a major quantitative trait locus controlling scab resistance in wheat. Phytopathology 89:343–348. https://doi.org/10.1094/PHYTO.1999.89.4.343
Baker NR, Rosenqvist E (2004) Applications of chlorophyll fluorescence can improve crop production strategies: an examination of future possibilities. J Exp Bot 55:1607–1621. https://doi.org/10.1093/jxb/erh196
Blum A (2005) Drought resistance, water-use efficiency, and yield potential—Are they compatible, dissonant, or mutually exclusive? Aust J Agric Res 56:1159–1168. https://doi.org/10.1071/AR05069
Bogard M, Hourcade D, Piquemal B et al (2021) Marker-based crop model-assisted ideotype design to improve avoidance of abiotic stress in bread wheat. J Exp Bot 72:1085–1103. https://doi.org/10.1093/jxb/eraa477
Bradbury PJ, Zhang Z, Kroon DE et al (2007) TASSEL: software for association mapping of complex traits in diverse samples. Bioinformatics 23:2633–2635. https://doi.org/10.1093/bioinformatics/btm308
Cao HX, Zhang ZB, Xu P et al (2007) Mutual physiological genetic mechanism of plant high water use efficiency and nutrition use efficiency. Colloids Surf B 57:1–7. https://doi.org/10.1016/j.colsurfb.2006.11.036
Clarke JM (1986) Effect of leaf rolling on leaf water loss in Trilicam spp. J Plant Sci 66:8–13
Corlett JE, Jones HG, Massacci A et al (1994) Water deficit, leaf rolling and susceptibility to photoinhibition in field grown sorghum. Physiol Plant 92:423–430. https://doi.org/10.1111/J.1399-3054.1994.tb08831.x
Doerge RW, Churchill GA (1996) Permutation tests for multiple loci affecting a quantitative character. Genetics 142:285–294. https://doi.org/10.1093/genetics/142.1.285
Fang L, Zhao F, Cong Y et al (2012) Rolling-leaf14 is a 2OG-Fe (II) oxygenase family protein that modulates rice leaf rolling by affecting secondary cell wall formation in leaves. Plant Biotech J 10:524–532. https://doi.org/10.1111/J.1467-7652.2012.00679.x
Gao L, Yang G, Li Y et al (2019) Fine mapping and candidate gene analysis of a QTL associated with leaf rolling index on chromosome 4 of maize (Zea mays L.). Theor Appl Genet 132:3047–3062. https://doi.org/10.1007/S00122-019-03405-1
Gouache D, le Bris X, Bogard M et al (2012) Evaluating agronomic adaptation options to increasing heat stress under climate change during wheat grain filling in France. Eur J Agron 39:62–70. https://doi.org/10.1016/J.EJA.2012.01.009
Gupta PK, Balyan HS, Gahlaut V (2017) QTL analysis for drought tolerance in wheat: present status and future possibilities. Agronomy 7(1):5. https://doi.org/10.3390/agronomy7010005
Hsiao TC, O’Toole JC, Yambao EB et al (1984) Influence of osmotic adjustment on leaf rolling and tissue death in rice (Oryza sativa L.). Plant Physiol 75:338–341. https://doi.org/10.1104/PP.75.2.338
Hu J, Zhu L, Zeng D et al (2010) Identification and characterization of NARROW AND ROLLED LEAF 1, a novel gene regulating leaf morphology and plant architecture in rice. Plant Mol Biol 73:283–292. https://doi.org/10.1007/S11103-010-9614-7
International Wheat Genomic Sequencing Consortium (IWGSC) (2018) Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 361:eaar7191. https://doi.org/10.1126/science.aar7191
Jordan K, Wang S, Lun Y et al (2015) A haplotype map of allohexaploid wheat reveals distinct patterns of selection on homoeologous genomes. Genome Biol 16(1):1–18. https://doi.org/10.1186/S13059-015-0606-4
Juarez MT, Kui JS, Thomas J et al (2004) microRNA-mediated repression of rolled leaf1 specifies maize leaf polarity. Nature 428:84–88. https://doi.org/10.1038/nature02363
Kadioglu A, Terzi R (2007) A dehydration avoidance mechanism: leaf rolling. Bot Rev 73:290–302. https://doi.org/10.1663/0006-8101(2007)73[290:adamlr]2.0.CO;2
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12:357–360
Kosambi DD (1943) The estimation of map distances from recombination values. Ann Eug 12:172–175. https://doi.org/10.1111/j.1469-1809.1943.tb02321.x
Li H, Handsaker B, Wysoker A et al (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25:2078–2079. https://doi.org/10.1093/Bioinformatics/btp352
Liu X, Li M, Liu K et al (2016) Semi-rolled leaf2 modulates rice leaf rolling by regulating abaxial side cell differentiation. J Exp Bot 67:2139–2150. https://doi.org/10.1093/jxb/erw029
McKenna A, Hanna M, Banks E et al (2010) The genome analysis toolkit: a mapreduce framework for analyzing next-generation DNA sequencing data. Genome Res 20:1297–1303. https://doi.org/10.1101/gr.107524.110
Meng L, Li H, Zhang L et al (2015) QTL IciMapping: integrated software for genetic linkage map construction and quantitative trait locus mapping in biparental populations. Crop J 3:269–283. https://doi.org/10.1016/j.cj.2015.01.001
Myşków B, Góralska M, Lenarczyk N et al (2018) Putative candidate genes responsible for leaf rolling in rye (Secale cereale L.). BMC Genet 19:1–11. https://doi.org/10.1186/S12863-018-0665-0/
Obsa BT, Eglinton J, Coventry S et al (2016) Genetic analysis of developmental and adaptive traits in three doubled haploid populations of barley (Hordeum vulgare L.). Theor Appl Genet 129:1139–1151. https://doi.org/10.1007/S00122-016-2689-z
Oppenheimer HR (1960). Adaptation to drought: xerophytism. Plant-water relationships in arid and semi-arid conditions: reviews of research. The United Nations Educulionul, Scicntijc and Cultural Orgunizulion, Printed by C. J. Bucher, Lucerne, Swilzerland
Patel RK, Jain M (2012) NGS QC Toolkit: a toolkit for quality control of next generation sequencing data. PLoS ONE 7:e30619. https://doi.org/10.1371/journal.0030619
Peleg Z, Fahima T, Krugman T et al (2009) Genomic dissection of drought resistance in durum wheat × wild emmer wheat recombinant inbred line population. Plant Cell Environ 32:758–779. https://doi.org/10.1111/J.1365-3040.2009.01956.x
Poland JA, Rife TW (2012) Genotyping-by-sequencing for plant breeding and genetics. Plant Genome 5:92. https://doi.org/10.3835/plantgenome2012.05.0005
Price AH, Young EM, Tomos AD (1997) Quantitative trait loci associated with stomatal conductance, leaf rolling and heading date mapped in upland rice (Oryza sativa). New Phytol 137:83–91. https://doi.org/10.1046/J.1469-8137.1997.00818.x
Qiang Z, Tianqing Z, Long H et al (2016) Joint mapping and allele mining of the rolled leaf trait in rice (Oryza sativa L.). PLoS ONE 11:e0158246. https://doi.org/10.1371/JOURNAL.PONE.0158246
Rawat N, Joshi A, Pumphrey M et al (2019) A tilling resource for hard red winter wheat variety Jagger. Crop Sci 59:1666–1671. https://doi.org/10.2135/CROPSCI2019.01.0011
Saglam A, Kadioglu A, Demiralay M et al (2014) Leaf rolling reduces photosynthetic loss in maize under severe drought. ACTA Bot Croat 73:315–323. https://doi.org/10.2478/botcro-2014-0012
Saneoka H, Ogata S, Agata W (1996) Cultivar differences in dry matter production and leaf water relations in water-stressed maize (Zea mays L.). Jpn J Grassl Sci 41(4):294–301
Shiferaw B, Smale M, Braun HJ et al (2013) Crops that feed the world 10. past successes and future challenges to the role played by wheat in global food security. Food Sec 5:291–317. https://doi.org/10.1007/s12571-013-0263-y
Singh BN, Mackill DJ (2008) Genetics of leaf rolling under drought stress. Rice Genet II:159–166. https://doi.org/10.1142/9789812814272_0015
Singh S, Singh TN (2000) Significance of leaf rolling in rice during water stress. Ind J Plant Physiol 5:214–218
Sommer R, Glazirina M, Yuldashev T et al (2013) Impact of climate change on wheat productivity in Central Asia. Agric Ecosyst Environ 178:78–99. https://doi.org/10.1016/j.agee.2013.06.011
Tahmasebi S, Heidari B, Pakniyat H et al (2016) Mapping QTLs associated with agronomic and physiological traits under terminal drought and heat stress conditions in wheat (Triticum aestivum L.). Genome 60:26–45. https://doi.org/10.1139/gen-2016-0017
Timmermans MCP, Schultes NP, Jankovsky JP et al (1998) Leafbladeless1 is required for dorsoventrality of lateral organs in maize. Development 125:2813–2823. https://doi.org/10.1242/dev.125.15.2813
Turgut R, Kadioglu A (1998) The effect of drought, temperature and irradiation on leaf rolling in Ctenanthe setosa. Biol Plant 41:629–633. https://doi.org/10.1023/A:1001817105685
Turner NC, Begg JE (1981) Plant-water relations and adaptation to stress. Plant Soil 58:97–131. https://doi.org/10.1007/bf02180051
Untergasser A, Cutcutache I, Koressaar T et al (2012) Primer3-new capabilities and interfaces. Nucleic Acids Res 40:e115. https://doi.org/10.1093/nar/gks596
Verma A, Niranjana M, Jha SK et al (2020) QTL detection and putative candidate gene prediction for leaf rolling under moisture stress condition in wheat. Sci Rep 10:1–13. https://doi.org/10.1038/s41598-020-75703-4
Wang JK (2009) Inclusive composite interval mapping of quantitative trait genes. Acta Agron Sin 35:239–245. https://doi.org/10.3724/sp.j.1006.2009.0023
Wang S (2007) Windows QTL cartographer 2.5
Xiang JJ, Zhang GH, Qian Q et al (2012) Semi-Rolled Leaf1 encodes a putative glycosylphosphatidylinositol-anchored protein and modulates rice leaf rolling by regulating the formation of bulliform cells. Plant Physiol 159:1488–1500. https://doi.org/10.1104/pp.112.199968
Xu Y, Wang Y, Long Q et al (2014) Overexpression of OsZHD1, a zinc finger homeodomain class homeobox transcription factor, induces abaxially curled and drooping leaf in rice. Planta 239:803–816. https://doi.org/10.1007/S00425-013-2009-7
Xu P, Ali A, Han B et al (2018) Current advances in molecular basis and mechanisms regulating leaf morphology in rice. Front Plant Sci. https://doi.org/10.3389/FPLS.2018.01528
Zou LP, Sun XH, Zhang ZG et al (2011) Leaf rolling controlled by the homeodomain leucine zipper class IV gene Roc5 in rice. Plant Physiol 156:1589–1602. https://doi.org/10.1104/PP.111.176016
Acknowledgements
This is contribution number 23-030-J from the Kansas Agricultural Experiment Station. Author thanks Dr. Michael Pumphrey for providing the Jagger mutant population. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. The USDA is an equal opportunity provider and employer.
Funding
Funding is provided by the US Wheat and Barley Scab Initiative, the Agriculture and Food Research Initiative Competitive Grant 2022-68013-36439 (WheatCAP) from the USDA National Institute of Food and Agriculture, and Chinese Scholarship Council.
Author information
Authors and Affiliations
Contributions
GB designed the research; RB, YX, SAP, and AB performed the research. NL and ZS conducted mutant screening. EA and KJ provided exome-capture data. RB, GZ, AF, and JR performed field phenotyping. RB and GB wrote the manuscript. All the authors provided inputs and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
The authors declare that the experiments comply with the current laws of the country in which they were performed.
Additional information
Communicated by Xianchun Xia.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bian, R., Liu, N., Xu, Y. et al. Quantitative trait loci for rolled leaf in a wheat EMS mutant from Jagger. Theor Appl Genet 136, 52 (2023). https://doi.org/10.1007/s00122-023-04284-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00122-023-04284-3