Abstract
Key message
The genetic basis of soybean root system architecture (RSA) and the genetic relationship between shoot and RSA were revealed by integrating data from recombinant inbred population grafting and QTL mapping.
Abstract
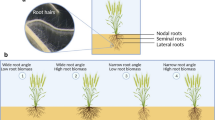
Variations in root system architecture (RSA) affect the functions of roots and thus play vital roles in plant adaptations and agricultural productivity. The aim of this study was to unravel the genetic relationship between RSA traits and shoot-related traits in soybean. This study characterized RSA variability at seedling stage in a recombinant inbred population, derived from a cross between cultivated soybean C08 and wild soybean W05, and performed high-resolution quantitative trait locus (QTL) mapping. In total, 34 and 41 QTLs were detected for RSA-related and shoot-related traits, respectively, constituting eight QTL clusters. Significant QTL correspondence was found between shoot biomass and RSA-related traits, consistent with significant correlations between these phenotypes. RSA-related QTLs also overlapped with selection regions in the genome, suggesting the cultivar RSA could be a partial consequence of domestication. Using reciprocal grafting, we confirmed that shoot-derived signals affected root development and the effects were controlled by multiple loci. Meanwhile, RSA-related QTLs were found to co-localize with four soybean flowering-time loci. Consistent with the phenotypes of the parental lines of our RI population, diminishing the function of flowering controlling E1 family through RNA interference (RNAi) led to reduced root growth. This implies that the flowering time-related genes within the RSA-related QTLs are actually contributing to RSA. To conclude, this study identified the QTLs that determine RSA through controlling root growth indirectly via regulating shoot functions, and discovered superior alleles from wild soybean that could be used to improve the root structure in existing soybean cultivars.






Similar content being viewed by others
Data availability
All data generated or analyzed in this study are included in this published article and its supplementary information files.
References
Abdel-Haleem H, Lee GJ, Boerma RH (2011) Identification of QTL for increased fibrous roots in soybean. Theor Appl Genet 122:935–946. https://doi.org/10.1007/s00122-010-1500-9
Bhat JA, Ali S, Salgotra RK et al (2016) Genomic selection in the era of next generation sequencing for complex traits in plant breeding. Front Genet 7:1–11. https://doi.org/10.3389/fgene.2016.00221
Broman KW, Wu H, Sen Ś, Churchill GA (2003) R/qtl: QTL mapping in experimental crosses. Bioinformatics 19:889–890. https://doi.org/10.1093/bioinformatics/btg112
Cao X, Wu T, Sun S et al (2019) Evaluation by grafting technique of changes in the contribution of root-to-shoot development and biomass production in soybean (glycine max) cultivars released from 1929 to 2006 in China. Crop Pasture Sci 70:585–594. https://doi.org/10.1071/CP19052
Chen YL, Dunbabin VM, Diggle AJ et al (2011) Development of a novel semi-hydroponic phenotyping system for studying root architecture. Funct Plant Biol 38:355–363. https://doi.org/10.1071/FP10241
Chen Y, Zhou T, Siddique KHM (2020) Method for characterization of root traits in chickpea germplasm for legume genomics and breeding (Chapter 14). Springer, New York, NY
Chen H, Kumawat G, Yan Y et al (2021a) Mapping and validation of a major QTL for primary root length of soybean seedlings grown in hydroponic conditions. BMC Genomics 22:1–9. https://doi.org/10.1186/s12864-021-07445-0
Chen Q, Li W, Tan L, Tian F (2021b) Harnessing knowledge from maize and rice domestication for new crop breeding. Mol Plant 14:9–26. https://doi.org/10.1016/j.molp.2020.12.006
Chen Y, Wang Z, Ye H et al (2022a) Root physiology and morphology of soybean in relation to stress tolerance, 1st edn. Elsevier
Chen Z, Sun J, Li D et al (2022b) Plasticity of root anatomy during domestication of a maize-teosinte derived population. J Exp Bot 73:139–153. https://doi.org/10.1093/jxb/erab406
Danecek P, Auton A, Abecasis G et al (2011) The variant call format and VCFtools. Bioinformatics 27:2156–2158. https://doi.org/10.1093/bioinformatics/btr330
Den HG, Van Isterdael G, Beeckman T, De Smet I (2010) The roots of a new green revolution. Trends Plant Sci 15:600–607. https://doi.org/10.1016/j.tplants.2010.08.009
Dong L, Fang C, Cheng Q et al (2021) Genetic basis and adaptation trajectory of soybean from its temperate origin to tropics. Nat Commun 12:1–11. https://doi.org/10.1038/s41467-021-25800-3
Dong L, Cheng Q, Fang C et al (2022) Parallel selection of distinct Tof5 alleles drove the adaptation of cultivated and wild soybean to high latitudes. Mol Plant 15:308–321. https://doi.org/10.1016/j.molp.2021.10.004
Feltus FA, Hart GE, Schertz KF et al (2006) Alignment of genetic maps and QTLs between inter- and intra-specific sorghum populations. Theor Appl Genet 112:1295–1305. https://doi.org/10.1007/s00122-006-0232-3
Huang C, Chen Q, Xu G et al (2016) Identification and fine mapping of quantitative trait loci for the number of vascular bundle in maize stem. J Integr Plant Biol 58:81–90. https://doi.org/10.1111/jipb.12358
Kong L, Lu S, Wang Y et al (2018) Quantitative trait locus mapping of flowering time and maturity in soybean using next-generation sequencing-based analysis. Front Plant Sci 9:1–20. https://doi.org/10.3389/fpls.2018.00995
Kou K, Yang H, Li H et al (2022) A functionally divergent SOC1 homolog improves soybean yield and latitudinal adaptation. Curr Biol. https://doi.org/10.1016/j.cub.2022.02.046
Lam HM, Xu X, Liu X et al (2010) Resequencing of 31 wild and cultivated soybean genomes identifies patterns of genetic diversity and selection. Nat Genet 42:1053–1059. https://doi.org/10.1038/ng.715
Liang H, Yu Y, Yang H et al (2014) Inheritance and QTL mapping of related root traits in soybean at the seedling stage. Theor Appl Genet 127:2127–2137. https://doi.org/10.1007/s00122-014-2366-z
Lim CW, Lee YW, Hwang CH (2011) Soybean nodule-enhanced CLE peptides in roots act as signals in gmnark-mediated nodulation suppression. Plant Cell Physiol 52:1613–1627. https://doi.org/10.1093/pcp/pcr091
Liu B, Kanazawa A, Matsumura H et al (2008) Genetic redundancy in soybean photoresponses associated with duplication of the phytochrome a gene. Genetics 180:995–1007. https://doi.org/10.1534/genetics.108.092742
Liu Y, Du H, Li P et al (2020) Pan-genome of wild and cultivated soybeans. Cell 182:162-176.e13. https://doi.org/10.1016/j.cell.2020.05.023
Liu S, Begum N, An T et al (2021) Characterization of root system architecture traits in diverse soybean genotypes using a semi-hydroponic system. Plants. https://doi.org/10.3390/plants10122781
LIU L feng, GAO L, ZHANG L xin, et al (2022) Co-silencing E1 and its homologs in an extremely late-maturing soybean cultivar confers super-early maturity and adaptation to high-latitude short-season regions. J Integr Agric 21:326–335. https://doi.org/10.1016/S2095-3119(20)63391-3
Lu S, Zhao X, Hu Y et al (2017) Natural variation at the soybean j locus improves adaptation to the tropics and enhances yield. Nat Genet 49:773–779. https://doi.org/10.1038/ng.3819
Lu S, Dong L, Fang C et al (2020) Stepwise selection on homeologous PRR genes controlling flowering and maturity during soybean domestication. Nat Genet 52:428–436. https://doi.org/10.1038/s41588-020-0604-7
Manavalan LP, Prince SJ, Musket TA et al (2015) Identification of novel QTL governing root architectural traits in an interspecific soybean population. PLoS ONE 10:1–18. https://doi.org/10.1371/journal.pone.0120490
Muñoz N, Qi X, Li MW et al (2016) Improvement in nitrogen fixation capacity could be part of the domestication process in soybean. Heredity (edinb) 117:84–93. https://doi.org/10.1038/hdy.2016.27
Paez-Garcia A, Motes CM, Scheible WR et al (2015) Root traits and phenotyping strategies for plant improvement. Plants 4:334–355. https://doi.org/10.3390/plants4020334
Péret B, Desnos T, Jost R et al (2014) Root architecture responses: in search of phosphate. Plant Physiol 166:1713–1723. https://doi.org/10.1104/pp.114.244541
Prince SJ, Song L, Qiu D et al (2015) Genetic variants in root architecture-related genes in a glycine soja accession, a potential resource to improve cultivated soybean. BMC Genomics 16:1–20. https://doi.org/10.1186/s12864-015-1334-6
Prince SJ, Vuong TD, Wu X et al (2020) Mapping quantitative trait loci for soybean seedling shoot and root architecture traits in an inter-specific genetic population. Front Plant Sci 11:1–13. https://doi.org/10.3389/fpls.2020.01284
Puig J, Pauluzzi G, Guiderdoni E, Gantet P (2012) Regulation of shoot and root development through mutual signaling. Mol Plant 5:974–983. https://doi.org/10.1093/mp/sss047
Qi X, Li MW, Xie M et al (2014) Identification of a novel salt tolerance gene in wild soybean by whole-genome sequencing. Nat Commun 5:1–11. https://doi.org/10.1038/ncomms5340
Salim M, Chen Y, Ye H et al (2022) Screening of soybean genotypes based on root morphology and shoot traits using the semi-hydroponic phenotyping platform and rhizobox technique. Agronomy. https://doi.org/10.3390/agronomy12010056
Sedivy EJ, Wu F, Hanzawa Y (2017) Soybean domestication: the origin, genetic architecture and molecular bases. New Phytol 214:539–553. https://doi.org/10.1111/nph.14418
Slovak R, Ogura T, Satbhai SB et al (2016) Genetic control of root growth: from genes to networks. Ann Bot 117:9–24. https://doi.org/10.1093/aob/mcv160
Takeshima R, Hayashi T, Zhu J et al (2016) A soybean quantitative trait locus that promotes flowering under long days is identified as FT5a, a FLOWERING LOCUS T ortholog. J Exp Bot 67:5247–5258. https://doi.org/10.1093/jxb/erw283
Tanaka N, Uno H, Okuda S et al (2017) SRPP, a cell wall protein is involved in development and protection of seeds and root hairs in arabidopsis thaliana. Plant Cell Physiol 58:760–769. https://doi.org/10.1093/pcp/pcx008
Tracy SR, Nagel KA, Postma JA et al (2020) Crop improvement from phenotyping roots: highlights reveal expanding opportunities. Trends Plant Sci 25:105–118. https://doi.org/10.1016/j.tplants.2019.10.015
Voss-Fels KP, Robinson H, Mudge SR et al (2018) VERNALIZATION1 modulates root system architecture in wheat and barley. Mol Plant 11:226–229. https://doi.org/10.1016/j.molp.2017.10.005
Waidmann S, Sarkel E, Kleine-Vehn J (2020) Same same, but different: growth responses of primary and lateral roots. J Exp Bot 71:2397–2411. https://doi.org/10.1093/JXB/ERAA027
Waines JG, Ehdaie B (2007) Domestication and crop physiology: roots of green-revolution wheat. Ann Bot 100:991–998. https://doi.org/10.1093/aob/mcm180
Wang F, Nan H, Chen L et al (2019) A new dominant locus, E11, controls early flowering time and maturity in soybean. Mol Breed. https://doi.org/10.1007/s11032-019-0978-3
Wang L, Sun S, Wu T et al (2020) Natural variation and CRISPR/Cas9-mediated mutation in GmPRR37 affect photoperiodic flowering and contribute to regional adaptation of soybean. Plant Biotechnol J 18:1869–1881. https://doi.org/10.1111/pbi.13346
Wang X, Li MW, Wong FL et al (2021) Increased copy number of gibberellin 2-oxidase 8 genes reduced trailing growth and shoot length during soybean domestication. Plant J 107:1739–1755. https://doi.org/10.1111/tpj.15414
Watanabe S, Harada K, Abe J (2011) Genetic and molecular bases of photoperiod responses of flowering in soybean. Breed Sci 61:531–543. https://doi.org/10.1270/jsbbs.61.531
Williams B, Kantartzi SK, Meksem K et al (2012) Genetic analysis of root and shoot traits in the ‘Essex’ By ‘Forrest’ recombinant inbred Line (RIL) population of soybean [Glycine max (L.) Merr.]. J Plant Genome Sci 1:1–9. https://doi.org/10.5147/jpgs.2012.0051
Xia Z, Watanabe S, Yamada T et al (2012) Positional cloning and characterization reveal the molecular basis for soybean maturity locus E1 that regulates photoperiodic flowering. Proc Natl Acad Sci U S A. https://doi.org/10.1073/pnas.1117982109
Xie M, Chung CYL, Li MW et al (2019) A reference-grade wild soybean genome. Nat Commun 10:1–12. https://doi.org/10.1038/s41467-019-09142-9
Xu G, Wang X, Huang C et al (2017) Complex genetic architecture underlies maize tassel domestication. New Phytol 214:852–864. https://doi.org/10.1111/nph.14400
Zhang Z, Zhang X, Lin Z et al (2018) The genetic architecture of nodal root number in maize. Plant J 93:1032–1044. https://doi.org/10.1111/tpj.13828
Zhao Y, Jiang C, hui, Rehman RMA, et al (2019) Genetic analysis of roots and shoots in rice seedling by association mapping. Genes and Genomics 41:95–105. https://doi.org/10.1007/s13258-018-0741-x
Zhao Y, Yin Z, Wang X et al (2021) Genetic basis and network underlying synergistic roots and shoots biomass accumulation revealed by genome-wide association studies in rice. Sci Rep 11:1–15. https://doi.org/10.1038/s41598-021-93170-3
Zhou R, Chen HF, Wang XZ et al (2011) Analysis of QTLs for root traits at seedling stage in soybean. Acta Agron Sin 37:1151–1158. https://doi.org/10.1016/S1875-2780(11)60032-1
Acknowledgements
This work was supported by grants from the Hong Kong Research Grants Council Area of Excellence Scheme (AoE/M-403/16) and the Lo Kwee-Seong Biomedical Research Fund to H-ML, the Hong Kong Scholars Program (XJ2020019) to CH, and UWA Research Collaboration Awards (2022/GR000814) to YC and H-ML. We would like to thank Ms. Jee Yan Chu for copy-editing this manuscript. Any opinions, findings, conclusions, or recommendations expressed in this publication do not reflect the views of the Government of the Hong Kong Special Administrative Region or the Innovation and Technology Commission.
Funding
This work was supported by Hong Kong Research Grants Council Area of Excellence Scheme (AoE/M‐403/16) and Lo Kwee-Seong Biomedical Research Fund awarded to H-ML, and UWA Research Collaboration Awards (2022/GR000814) to YC and H-ML. CH was supported by the Hong Kong Scholars Program (XJ2020019).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. H-ML coordinated and supervised the project. F-LW and CW prepared RI lines materials. ZW, SL and YC designed and set up the semi-hydroponic platform for root phenotyping and phenotypic data collection. ZW, CH, F-LW, C-KM, C-CS, AL and W-SY performed grafting assays and phenotypic data collection. ZW, QW and C-KM performed phenotypic data analyses. ZW, M-WL, YN, CH, ZX, MH and XW performed QTL mapping and locus analyses. YN calculated FST values. LL, WH and TH generated the transgenic soybean lines. ZW and M-WL wrote the first draft of the manuscript. CH, W-SY, YN, ZX, MH and AL commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Volker Hahn.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Z., Huang, C., Niu, Y. et al. QTL analyses of soybean root system architecture revealed genetic relationships with shoot-related traits. Theor Appl Genet 135, 4507–4522 (2022). https://doi.org/10.1007/s00122-022-04235-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-022-04235-4