Abstract
Key message
New translocation lines with T6V#4S·6AL in the Ph1 and ph1b backgrounds were developed with improved inheritance of powdery mildew resistance.
Abstract
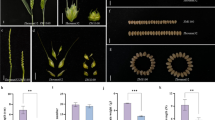
The wheat-Dasypyrum villosum T6V#4S·6DL translocation line Pm97033, which exhibits strong powdery mildew (PM) resistance, was developed many years ago, but has limited application in wheat breeding. One of the major reasons for this is that the translocation chromosome has low transmission rate, which makes it difficult to obtain ideal genotype through recombination with other elite agronomic traits in a limited segregating population. Further modifications are thus needed to make better use of this genetic resource. In this study, Pm97033 and the T6V#2S·6AL translocation line NY-W were hybridized with the CS ph1b mutant, and two F1 hybrids were hybridized with each other. Then, plants homozygous for the ph1b deletion carrying the alien chromosome arm(s) 6V#2S and 6V#4S were identified from the segregating populations using molecular markers. New T6V#4S·6AL and T6V#2-6V#4S·6AL translocations were identified by molecular markers and confirmed by genomic in situ hybridization (GISH). Individuals that were heterozygous or homozygous for the translocation chromosome in Ph1 and ph1b backgrounds were obtained. The ratio of PM resistance vs. susceptibility in the self-pollinated heterozygous plants was 3:1, and the phenotype was completely consistent with the KASP genotyping. Thus, the new translocation chromosomes had higher transmission rate than the original T6V#4S·6DL, and so can be effectively applied in breeding programs.






Similar content being viewed by others
Availability of data and materials
There are supplementary data in this manuscript and all data are included in the main text. The materials used in this study will be available upon request.
References
An D, Zheng Q, Luo Q, Ma P, Zhang H, Li L, Han F, Xu H, Xu Y, Zhang X, Zhou Y (2015) Molecular cytogenetic identification of a new wheat-rye 6R chromosome disomic addition line with powdery mildew resistance. PLoS ONE 10:e0134534. https://doi.org/10.1371/journal.pone.0134534
An D, Ma P, Zheng Q, Fu S, Li L, Han F, Han G, Wang J, Xu Y, Jin Y, Luo Q, Zhang X (2019) Development and molecular cytogenetic identificationof a new wheat-rye 4R chromosome disomic addition line with resistances to powdery mildew, stripe rust and sharp eyespot. Theor Appl Genet 132:257–272. https://doi.org/10.1007/s00122-018-3214-3
Ayala-Navarrete LI, Mechanicos AA, Gibson JM, Singh D, Bariana HS, Fletcher J, Shorter S, Larkin PJ (2013) The Pontin series of recombinant alien translocations in bread wheat: Single translocations integrating combinations of Bdv2, Lr19 and Sr25 disease-resistance genes from Thinopyrum intermedium and Th ponticum. Theor Appl Genet 126:2467–2475. https://doi.org/10.1007/s00122-013-2147-0
Bie T, Zhao R, Zhu S, Chen S, Cen B, Zhang B, Gao D, Jiang Z, Chen T, Wang L, Wu R, He H (2015) Development and characterization of marker MBH1 simultaneously tagging genes Pm21 and PmV conferring resistance to powdery mildew in wheat. Mol Breed 35:189. https://doi.org/10.1007/s11032-015-0385-3
Cao A, Xing L, Wang X, Yang X, Wang W, Sun Y, Qian C, Ni J, Chen Y, Liu D, Wang X, Chen P (2011) Serine/threonine kinase gene Stpk-V, a key member of powdery mildew resistance gene Pm21, confers powdery mildew resistance in wheat. Proc Natl Acad Sci USA 108:7727–7732. https://doi.org/10.1073/pnas.1016981108
Chen P, Qi L, Zhou B, Zhang S, Liu D (1995) Development and molecular cytogenetic analysis of wheat-Haynaldia villosa 6VS/6AL translocation lines specifying resistance to powdery mildew. Theor Appl Genet 91:1125–1128. https://doi.org/10.1007/BF00223930
Chen X, Shi A, Shang L, Leath S, Murphy JP (1997) The resistance reaction of H. villosa to powdery mildew isolates and its expression in wheat background. Acta Phytopathol Sin 27:17–22 (in Chinese with English abstract)
Chen F, Jia H, Zhang X, Qiao L, Li X, Zheng J, Guo H, Powers C, Yan L, Chang Z (2019) Positional cloning of PmCh135J reveals the origin and allelic variation of the Pm2 gene for powdery mildew resistance in wheat. Crop J 7:771–783. https://doi.org/10.1016/j.cj.2019.08.004
Copete-Parada A, Carmen P, Adoracion C (2021) Development and characterization of wheat-Agropyron cristatum introgression lines induced by ga-metocidal genes and wheat ph1b mutant. Agronomy 11:277. https://doi.org/10.3390/agronomy11020277
Cowger C, Mehra L, Arellano C, Meyers E, Murphy JP (2018) Virulence differences in Blumeria graminis f. sp. tritici from the central and eastern united states. Phytopathology 108:402–411. https://doi.org/10.1094/PHYTO-06-17-0211-R
Cowger C, Meyers E, Whetten R (2022) Sensitivity of the U.S. wheat powdery mildew population to quinone outside inhibitor fungicides and determination of the complete Blumeria graminis f. sp. tritici cytochrome b gene. Phytopathology 112:2. https://doi.org/10.1094/PHYTO-04-21-0132-R
Fedak G, Chi D, Wolfe D, Ouellet T, Cao WG, Han FP, Xue A (2021) Transfer of fusarium head blight resistance from Thinopyrum elongatum to bread wheat cultivar Chinese spring. Genome 64:997–1008. https://doi.org/10.1139/gen-2020-0151
Friebe B, Jiang J, Raupp WJ, Mclntosh RA, Gill BS (1996) Characterization of wheat-alien translocations conferring resistance to diseases and pests: current status. Euphytica 91:59–87. https://doi.org/10.1007/BF00035277
Gill KS, Gill BS (1996) A PCR-based screening assay of Ph1, the chromosome pairing regulator gene of wheat. Crop Sci 36:719–722. https://doi.org/10.2135/cropsci1996.0011183X003600030033x
Gill KS, Gill BS, Endo TR, Mukai Y (1993) Fine physical mapping of Ph1, a chromosome pairing regulator gene in polyploid wheat. Genetics 134:1231–1236. https://doi.org/10.1093/genetics/134.4.1231
Griffiths S, Sharp R, Foote TN, Bertin I, Wanous M, Reader S, Colas I, Moore G (2006) Molecular characterization of Ph1 as a major chromosome pairing locus in polyploid wheat. Nature 439:749–752. https://doi.org/10.1038/nature04434
Gyawali Y, Zhang W, Chao S, Xu S, Cai X (2019) Delimitation of wheat Ph1b deletion and development of Ph1b-specific DNA markers. Theor Appl Genet 132:195–204. https://doi.org/10.1007/s00122-018-3207-2
He F, Bao Y, Qi X, Ma Y, Li X, Wang H (2017) Molecular cytogenetic identification of a wheat-Thinopyrum ponticum translocation line resistant to powdery mildew. J Genet 96:165–169. https://doi.org/10.1007/s12041-017-0754-2
Hou L, Zhang X, Li X, Jia J, Yang H, Zhan H, Qiao L, Guo H, Chang Z (2015) Mapping of powdery mildew resistance gene PmCH89 in a putative wheat-Thinopyrum intermedium introgression line. Int J Mol Sci 16:17231–17244. https://doi.org/10.3390/ijms160817231
Ibba MI, Zhang M, Cai X, Morris CF (2019) Identification of a conserved ph1b-mediated 5DS-5BS crossing over site in soft-kernel durum wheat (Triticum turgidum subsp. durum) lines. Euphytica 215:200. https://doi.org/10.1007/s10681-019-2518-y
Kang H, Zhang Z, Xu L, Qi W, Tang Y, Wang H, Zhu W, Li D, Zeng J, Wang Y, Fan X, Sha L, Zhang H, Zhou Y (2016) Characterization of wheat - Psathyrostachys huashanica small segment translocation line with enhanced kernels per spike and stripe rust resistance. Genome 59:221–229. https://doi.org/10.1139/gen-2015-0138
Kihara H (1919) Üeber cytologische Studien bei einigen Getreidearten. Spezies-Bastarde des Weizens und Weizenroggen-Bastard. Bot Mag 33:17–38. https://doi.org/10.15281/jplantres1887.33.386_17
Li H, Chen X, Xin MY, Xu H, Chen X, Jia X (2005) Development and identification of wheat-Haynaldia villosa T6DL·6VS chromosome translocation lines conferring resistance to powdery mildew. Plant Breed 124:203–205. https://doi.org/10.1111/j.1439-0523.2004.01062.x
Li Q, Lu Y, Pan C, Zhang J, Liu W, Yang X, Li X, Xi Y, Li L (2016) Characterization of a novel wheat-Agropyron cristatum 2P disomic addition line with powdery mildew resistance. Crop Sci 56:2390–2400. https://doi.org/10.2135/cropsci2015.10.0638
Li H, Dong Z, Ma C, Tian X, Qi Z, Wu N, Friebe B, Xiang Z, Xia Q, Liu W, Li T (2019a) Physical mapping of stem rust resistance gene Sr52 from Dasypyrum villosum based on Ph1b-induced homoeologous recombination. Int J Mol Sci 20:4887. https://doi.org/10.3390/ijms20194887
Li J, Yao X, Yang Z, Cheng X, Yuan F, Liu Y, Wu J, Yang Q, Zhao J, Chen X (2019b) Molecular cytogenetic characterization of a novel wheat-Psathyrostachys huashanica keng 5Ns (5D) disomic substitution line with stripe rust resistance. Mol Breed 39:109
Li S, Wang J, Wang K, Chen J, Wang K, Du L, Ni Z, Lin Z, Ye X (2019c) Development of PCR markers specific to Dasypyrum villosum genome based on transcriptome data and their application in breeding Triticum aestivum-D. villosum#4 alien chromosome lines. BMC Genom 20:289. https://doi.org/10.1186/s12864-019-5630-4
Li S, Jia Z, Wang K, Du L, Li H, Lin Z, Ye X (2020) Screening and functional characterization of candidate resistance genes to powdery mildew from Dasypyrum villosum#4 in a wheat line Pm97033. Theor Appl Genet 133:3067–3083. https://doi.org/10.1007/s00122-020-03655-4
Liu C, Li S, Wang M, Ye X, Lin Z (2017a) Developing of specific transcription sequences P21461 and P33259 on Dasypyrum villosum 6VS and application of molecular markers in identifying wheat–D. villosum breeding materials with powdery mildew resistance. Acta Agron Sin 43:983–992. https://doi.org/10.3724/SP.J.1006.2017.00983 (in Chinese with English abstract)
Liu C, Ye X, Wang M, Li S, Lin Z (2017b) Genetic behavior of Triticum aestivum-Dasypyrum villosum translocation chromosomes T6V#4S·6DL and T6V#2S·6AL carrying powdery mildew resistance. J Integr Agric 16:2136–2144. https://doi.org/10.1016/S2095-3119(16)61568-X
Lukaszewski AJ, Cowger C (2017) Re-engineering of the Pm21 transfer from Haynaldia villosa to bread wheat by induced homoeologous recombination. Crop Sci 57:2590–2594. https://doi.org/10.2135/cropsci2017.03.0192
Ma D, Hou L, Sun C, Zhang X, Yin J, Guo Q, Zhu Y (2019a) Molecular mapping of stripe rust resistance gene YrH9017 in wheat-Psathyrostachys huashanica introgression line H9017-14-16-5-3. J Integr Agric 18:108–114. https://doi.org/10.1016/S2095-3119(18)62048-9
Ma X, Xu Z, Wang J, Chen H, Ye X, Lin Z (2019b) Pairing and exchanging between Daypyrum villosum Chromosomes 6V#2 and 6V#4 in the hybrids of two different wheat alien substitution lines. Int J Mol Sci 20:6063. https://doi.org/10.3390/ijms20236063
Morris R, Sears ER (1967) The cytogenetics of wheat and its relatives. American Society of Agronomy Inc, Madison, pp 19–87
Paolacci AR, Tanzarella OA, Porceddu E, Ciaffi M (2009) Identification and validation of reference genes for quantitative RT-PCR normalization in wheat. BMC Mol Biol 10:11. https://doi.org/10.1186/1471-2199-10-11
Qi L, Pumphrey MO, Friebe B, Zhang P, Qian C, Bowden RL, Rouse MN, Jin Y, Gill BS (2011) A novel robertsonian translocation event leads to transfer of a stem rust resistance gene (Sr52) effective against race Ug99 from Dasypyrum villosum into bread wheat. Theor Appl Genet 123:159–167. https://doi.org/10.1007/s00122-011-1574-z
Qu L, Foote T, Roberts M, Money T, Aragon-Alcaide L, Snape J, Moore G (1998) A simple PCR-based method for scoring the Ph1b deletion in wheat. Theor Appl Genet 96:371–375. https://doi.org/10.1007/s001220050751
Rahmatov M, Rouse MN, Nirmala J, Danilova T, Friebe B, Steffenson BJ, Johansson E (2016) A new 2DS·2RL Robertsonian translocation transfers stem rust resistance gene Sr59 into wheat. Theor Appl Genet 129:1383–1392. https://doi.org/10.1007/s00122-016-2710-6
Riley R, Chapman V (1958) Genetic control of the cytologically diploid behaviour of hexaploid wheat. Nature 182:713–715. https://doi.org/10.1038/182713a0
Riley R, Chapman V, Kimber G (1959) Genetic control of chromosome pairing in intergeneric hybrids with wheat. Nature 183:1244–1246. https://doi.org/10.1038/1831244a0
Roberts MA, Reader SM, Dalgliesh C, Miller TE, Foote TN, Fish LJ, Snape JW, Moore G (1999) Induction and characterization of Ph1 wheat mutants. Genetics 153:1909–1918. https://doi.org/10.1093/genetics/153.4.1909
Sears ER (1952) Homoeologous chromosomes in Triticum aestivum. Genetics 37:624
Sears ER (1976) Genetic control of chromosome pairing in wheat. Annu Rev Genet 10:31–51
Sears ER (1977) An induced mutant with homoeologous pairing in common wheat. Can J Genet Cytol 19:585–593. https://doi.org/10.1139/g77-063
Segal G, Liu B, Vega JM, Abbo S, Rodova M, Feldman M (1997) Identification of a chromosome-specific probe that maps within the Ph1 deletions in common and durum wheat. Theor Appl Genet 94:968–970. https://doi.org/10.1007/s001220050502
Sidhu GK, Rustgi S, Shafqat MN, von Wettstein D, Gill KS (2008) Fine structure mapping of a gene-rich region of wheat carrying Ph1, a suppressor of crossing over between homoeologous chromosomes. Proc Natl Acad Sci USA 105:15220–15220
Wan W, Xiao J, Li M, Tang X, Wen M, Cheruiyot A, Li Y, Wang H, Wang X (2020) Fine mapping of wheat powdery mildew resistance gene pm6 using 2B/2G homoeologous recombinants induced by the ph1b mutant. Theor Appl Genet 133:1265–1275. https://doi.org/10.1007/s00122-020-03546-8
Wang H, Zhao R, Yuan C, Zhang S, Xiao J, Wang X (2013) Transmission of T4DL 4VS chromosome through gametes and its genetic stability in different genetic background. J Triticeae Crops 33:13–17. https://doi.org/10.7606/j.issn.1009-1041.2013.01.03 (in Chinese with English abstract)
Wang Y, Quan W, Peng N, Wang C, Yang X, Liu X, Zhang H, Chen C, Ji W (2016) Molecular cytogenetic identification of a wheat-Aegilops geniculata Roth 7M(g) disomic addition line with powdery mildew resistance. Mol Breed 36:401
Wang B, Ma X, Ye X, Zhou Y, Ma Y, Lin Z (2021) Genotypic and phenotypic characterization of two Triticum aestivum L-Dasypyrum villosum translocations lines in the same wheat genetic background. Agronomy 11:399. https://doi.org/10.3390/agronomy11020399
Wani S, Mohan A, Singh G (2021) Physiological, molecular, and genetic perspective of wheat improvement. Springer, Cham
Wei W, Qin R, Song Y, Ning S, Guo L, Gu M (2002) Location and analysis of introgressed segments in the parthenogenetic progenies of Zea mays × Z. diploperennis by GISH. Acta Bot Sin 44:373–376 (in Chinese with English abstract)
Whelan ED, Conner PRL, Thomas JB (1986) Transmission of a wheat alien chromosome translocation with resistance to the wheat curl mite in common wheat, Triticum aestivum L. Can J Genet Cytol 28:294–297. https://doi.org/10.1023/A:1018320710129
Xing L, Yuan L, Lv Z, Wang Q, Yin C, Huang Z, Liu J, Cao S, Zhang R, Chen P, Karafiatova M, Vrana J, Bartos J, Dolezel J, Cao A (2021) Long-range assembly of sequences helps to unravel the genome structure and small variation of the wheat-Haynaldia villosa translocated chromosome 6VS·6AL. Plant Biotechnol J 19:1567–1578. https://doi.org/10.1111/pbi.13570
Zhan H, Zhang X, Li G, Pan Z, Hu J, Li X, Qiao L, Jia J, Guo H, Chang Z, Yang Z (2015) Molecular characterization of a new wheat-Thinopyrum intermedium translocation line with resistance to powdery mildew and stripe rust. Int J Mol Sci 16:2162–2173. https://doi.org/10.3390/ijms16012162
Zhang M, Zhang W, Zhu X, Sun Q, Yan C, Xu SS, Fiedler J, Cai X (2020) Dissection and physical mapping of wheat chromosome 7B by inducing meiotic recombination with its homoeologues in Aegilops speltoides and Thinopyrum elongatum. Theor Appl Genet 133:3455–3467. https://doi.org/10.1007/s00122-020-03680-3
Zhang R, Xiong C, Mu H, Yao R, Meng X, Kong L, Xing L, Wu J, Feng Y, Cao A (2021a) Pm67, a new powdery mildew resistance gene transferred from Dasypyrum villosum chromosome 1V to common wheat (Triticum aestivum L.). Crop J 9:882–888. https://doi.org/10.1016/j.cj.2020.09.012
Zhang S, Fan C, Luo J, Huang L, Xie D, Li Y, Chen Z, Jiang B, Ning S, Yuan Z, Huang L, Zhang L, Liu D, Hao M (2021b) KASP markers to detect sub-chromosomal arm translocations between 6VS of Haynaldia villosa and 6AS of wheat. Euphytica 217:10. https://doi.org/10.1007/s10681-020-02744-1
Zhang X, Wang W, Liu C, Zhu S, Gao H, Xu H, Zhang L, Song J, Song W, Liu K, He H, Ma P (2021c) Diagnostic kompetitive allele-specific PCR markers of wheat broad-spectrum powdery mildew resistance genes Pm21, PmV, and Pm12 developed for high-throughput marker-assisted selection. Plant Dis 105:2844–2850. https://doi.org/10.1094/PDIS-02-21-0308-RE
Zhao R, Wang H, Xiao J, Bie T, Cheng S, Jia Q, Yuan C, Zhang R, Cao A, Chen P, Wang X (2013) Induction of 4VS chromosome recombinants using the CS ph1b mutant and mapping of the wheat yellow mosaic virus resistance gene from Haynaldia villosa. Theor Appl Genet 126:2921–2930. https://doi.org/10.1007/s00122-013-2181-y
Zhao R, Liu B, Jiang Z, Chen T, Wang L, Ji Y, Hu Z, He H, Bie T (2019) Comparative analysis of genetic effects of wheat-Dasypyrum villosum translocations T6V#2S·6AL and T6V#4S·6DL. Plant Breed 138:503–512. https://doi.org/10.1111/pbr.12711
Acknowledgements
The authors thank Dr. Fangpu Han, Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, for his guidance on genomic in situ hybridization of wheat chromosomes, and Ms. Yiping Yan, Institute of Crop Sciences, Chinese Academy of Agricultural Sciences, for her assistance in making hybridization and disease resistance investigation.
Funding
This study was financially supported by the National Key Research and Development Program of China (Grant Nos. 2016YFD0102002 and 2016YFD0102001).
Author information
Authors and Affiliations
Contributions
XLM, YNC, and JNC conducted the experiments. XLM and ZSL analyzed the data. XLM, ZSL, and XGY wrote the manuscript. YNC prepared the files of figures and tables. MY and BCW participated in partial ph1b genotyping experiments. ZSL and XGY conceived and directed the project.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Ethics approval
Not applicable.
Additional information
Communicated by Steven S. Xu.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ma, X., Chang, Y., Chen, J. et al. Development of wheat-Dasypyrum villosum T6V#4S·6AL translocation lines with enhanced inheritance for powdery mildew resistance. Theor Appl Genet 135, 2423–2435 (2022). https://doi.org/10.1007/s00122-022-04124-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-022-04124-w