Abstract
Key message
We present the first genetic map of an allohexaploid Brassica species, based on segregating microsatellite markers in a doubled haploid mapping population generated from a hybrid between two hexaploid parents.
Abstract
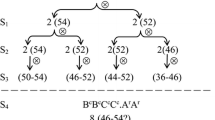
This study reports the first genetic map of trigenomic Brassica. A doubled haploid mapping population consisting of 189 lines was obtained via microspore culture from a hybrid H16-1 derived from a cross between two allohexaploid Brassica lines (7H170-1 and Y54-2). Simple sequence repeat primer pairs specific to the A genome (107), B genome (44) and C genome (109) were used to construct a genetic linkage map of the population. Twenty-seven linkage groups were resolved from 274 polymorphic loci on the A genome (109), B genome (49) and C genome (116) covering a total genetic distance of 3178.8 cM with an average distance between markers of 11.60 cM. This is the first genetic framework map for the artificially synthesized Brassica allohexaploids. The linkage groups represent the expected complement of chromosomes in the A, B and C genomes from the original diploid and tetraploid parents. This framework linkage map will be valuable for QTL analysis and future genetic improvement of a new allohexaploid Brassica species, and in improving our understanding of the genetic control of meiosis in new polyploids.





Similar content being viewed by others
References
Allender CJ, King GJ (2010) Origins of the amphiploid species Brassica napus L. investigated by chloroplast and nuclear molecular markers. BMC Plant Biol 10:54. doi:10.1186/1471-2229-10-54
Annisa, Chen S, Cowling WA (2013) Global genetic diversity in oilseed Brassica rapa. Crop Pasture Sci 64:993–1007. doi:10.1071/CP13206
Attia T, Busso C, Röbbelen G (1987) Digenomic triploids for an assessment of chromosome relationships in the cultivated diploid Brassica species. Genome 29:326–330
Axelsson T, Bowman CM, Sharpe AG, Lydiate DJ, Lagercrantz U (2000) Amphidiploid Brassica juncea contains conserved progenitor genomes. Genome 43:679–688. doi:10.1139/gen-43-4-679
Chalhoub B, Denoeud F, Liu S et al (2014) Early allopolyploid evolution in the post-Neolithic Brassica napus oilseed genome. Science 345:950–953. doi:10.1126/science.1253435
Chen S, Nelson MN, Ghamkhar K, Fu T, Cowling WA (2008) Divergent patterns of allelic diversity from similar origins: the case of oilseed rape (Brassica napus L.) in China and Australia. Genome 51:1–10. doi:10.1139/G07-095
Chen S, Zou J, Cowling WA, Meng J (2010) Allelic diversity in a novel gene pool of canola-quality Brassica napus enriched with alleles from B. rapa and B. carinata. Crop Pasture Sci 61:483–492
Chen S, Nelson MN, Chèvre AM et al (2011) Trigenomic bridges for Brassica improvement. Crit Rev Plant Sci 30:524–547. doi:10.1080/07352689.2011.615700
Chen S, Wan Z, Nelson MN, Chanhan JS, Redden R, Burton WA, Lin P, Salisbury PA, Fu T, Cowling WA (2013) Evidence from genome-wide simple sequence repeat markers for a polyphyletic origin and secondary centers of genetic diversity of Brassica juncea in China and India. J Hered 104:416–427. doi:10.1093/jhered/est015
Doyle J (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Geng XX, Chen S, Astarini IA, Yan GJ, Tian E, Meng JL, Li ZY, Ge HX, Nelson MN, Mason AS, Pradhan A, Zhou WJ, Cowling WA (2013) Doubled haploids of novel trigenomic Brassica derived from various interspecific crosses. Plant Cell Tissue Organ Cult 113:501–511
Guo S, Zou J, Li R, Long Y, Chen S, Meng J (2012) A genetic linkage map of Brassica carinata constructed with a doubled haploid population. Theor Appl Genet 125:1113–1124. doi:10.1007/s00122-012-1898-3
Guo Y, Chen S, Li Z, Cowling WA (2014) Center of origin and centers of diversity in an ancient crop, Brassica rapa (turnip rape). J Hered 105:555–565. doi:10.1093/jhered/esu021
Kresovich S, Szewc-McFadden AK, Bliek SM, McFerson JR (1995) Abundance and characterization of simple-sequence repeats (SSRs) isolated from a size-fractionated genomic library of Brassica napus L. (rapeseed). Theor Appl Genet 91:206–211. doi:10.1007/BF00220879
Lagercrantz U, Ellegren H, Andersson L (1993) The abundance of various polymorphic microsatellite motifs differs between plants and vertebrates. Nucleic Acids Res 21:1111–1115. doi:10.1093/nar/21.5.1111
Li Q, Wan J (2005) SSRHunter: development of a local searching software for SSR sites. Hereditas 27:808–810
Li MT, Li ZY, Zhang CY, Qian W, Meng JL (2005a) Reproduction and cytogenetic characterization of interspecific hybrids derived from crosses between Brassica carinata and B. rapa. Theor Appl Genet 110:1284–1289
Li MT, Zhang CY, Li ZY, Meng JL (2005b) The analysis of the biological characters in hexapod hybrids derived from Brassica carinata and Brassica rapa. Acta Agron Sin 31:1579–1585
Li H, Chen X, Yang Y, Xu J, Gu J, Fu J, Qian X, Zhang S, Wu J, Liu K (2011) Development and genetic mapping of microsatellite markers from whole genome shotgun sequences in Brassica oleracea. Mol Breed 28:585–596. doi:10.1007/s11032-010-9509-y
Lu GY, Yang GS, Fu TD (2001) Silver stained AFLP—a novel assay for DNA finger printing in Brassica napus. J Huazhong Agric Univ. doi:10.3321/j.issn:1000-2421.2001.05.002
Mason AS, Batley J (2015) Creating new interspecific hybrid and polyploid crops. Trends Biotechnol 33:436–441. doi:10.1016/j.tibtech.2015.06.004
Mason AS, Huteau V, Eber F, Coriton O, Yan G, Nelson MN, Cowling WA, Chèvre A (2010) Genome structure affects the rate of autosyndesis and allosyndesis in AABC, BBAC and CCAB Brassica interspecific hybrids. Chromosom Res 18:655–666. doi:10.1007/s10577-010-9140-0
Mason AS, Yan GJ, Cowling WA, Nelson MN (2012) A new method for producing allohexaploid Brassica through unreduced gametes. Euphytica 186:277–287. doi:10.1007/s10681-011-0537-4
Mason AS, Nelson MN, Takahira J et al (2014) The fate of chromosomes and alleles in an allohexaploid Brassica population. Genetics 197:273–283. doi:10.1534/genetics.113.159574
Mason AS, Takahira J, Atri C, Samans B, Hayward A, Cowling WA, Batley J, Nelson MN (2015) Microspore culture reveals complex meiotic behaviour in a trigenomic Brassica hybrid. BMC Plant Biol 15:173
Piquemal J, Cinquin E, Couton F, Rondeau C, Seignoret E, Doucet I, Perrer D, Villeger MJ, Vincourt P, Blanchard P (2005) Construction of an oilseed rape (Brassica napus L.) genetic map with SSR markers. Theor Appl Genet 111:1514–1523. doi:10.1007/s00122-005-0080-6
Plieske J, Struss D (2001) Microsatellite markers for genome analysis in Brassica. I. Development in Brassica napus and abundance in Brassicaceae species. Theor Appl Genet 102:689–694. doi:10.1007/s001220051698
Pradhan A, Plummer JA, Nelson MN, Cowling WA, Yan GJ (2010) Successful induction of trigenomic hexaploid Brassica from a triploid hybrid of B. napus L. and B. nigra (L.) Koch. Euphytica 176:87–98. doi:10.1007/s10681-010-0218-8
Saal B, Plieske J, Hu J, Quiros F, Struss D (2001) Microsatellite markers for genome analysis in Brassica. II. Assignment of rapeseed microsatellites to the A and C genomes and genetic mapping in Brassica oleracea L. Theor Appl Genet 102:695–699
Szewc-McFadden AK, Kresovich S, Bliek SM, Mitchell SE, McFerson JR (1996) Identification of polymorphic, conserved simple sequence repeats (SSRs) in cultivated Brassica species. Theor Appl Genet 93:534–538. doi:10.1007/s001220050311
Tian E, Jiang Y, Chen L, Zou J, Liu F, Meng J (2010) Synthesis of a Brassica trigenomic allohexaploid (B. carinata × B. rapa) de novo and its stability in subsequent generations. Theor Appl Genet 121:1431–1440. doi:10.1007/s00122-010-1399-1
U N (1935) Genome analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilization. Jpn J Bot 7:389–452
Uzunova MI, Ecke W (1999) Abundance, polymorphism and genetic mapping of microsatellites in oilseed rape (Brassica napus L.). Plant Breed 118:323–326. doi:10.1046/j.1439-0523.1999.00371.x
van Ooijen JW (2006) Joinmap 4: software for the calculation of genetic linkage maps in experimental populations. Kyazma BV, Wageningen
Xu J, Qian X, Wang X, Li R, Cheng X, Yang Y, Fu J, Zhang S, King GJ, Wu J, Liu K (2010) Construction of an integrated genetic linkage map for the A genome of Brassica napus using SSR markers derived from sequenced BACs in B. rapa. BMC Genomics 11:594. doi:10.1186/1471-2164-11-594
Zhou WJ, Zhang GQ, Tuvesson S, Dayteg C, Gertsson B (2006) Genetic survey of Chinese and Swedish oilseed rape (Brassica napus L.) by simple sequence repeats (SSRs). Genet Resour Crop Evol 53:443–447. doi:10.1007/s10722-004-7862-6
Acknowledgments
This work was supported partly by the Science and Technology Department of Zhejiang Province (2012C12902-1, 2011R50026-5), the Jiangsu Collaborative Innovation Center for Modern Crop Production, the Australia–China Special Fund, jointly managed by the Department of Innovation Industry Science and Research (Australia) and the Ministry of Science and Technology, and the National Natural Science Foundation (China).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Additional information
Communicated by C. F. Quiros.
S. Yang and S. Chen are equal first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, S., Chen, S., Geng, X.X. et al. The first genetic map of a synthesized allohexaploid Brassica with A, B and C genomes based on simple sequence repeat markers. Theor Appl Genet 129, 689–701 (2016). https://doi.org/10.1007/s00122-015-2657-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-015-2657-z