Abstract
Background
Diffusion-weighted MRI (DWI) is routinely used in abdominal imaging. In addition to neoplastic diseases, inflammatory changes can be delineated and diagnosed based on diffusion restriction in DWI. DWI is also increasingly used in the context of MRI of the small and large intestine.
Objective
This article focuses on the technical aspects of DWI and its role in the diagnosis of Crohn’s disease (CD) as well as in the grading of disease severity and in treatment monitoring.
Materials and methods
Guidelines, basic research papers, and review articles were analyzed.
Results
Diffusion-weighted MRI is a specialized MRI technique that visualizes the diffusion of water molecules in biological tissues. In the context of MRI of the small and large intestine, DWI facilitates the diagnosis of inflammatory bowel disease and assessment of treatment response. DWI enables detection of not only intra- and transmural changes, but also extramural pathologies and complications. However, DWI also has its limitations and challenges.
Conclusion
This article provides a comprehensive overview of the use of DWI for diagnostic evaluation of bowel wall changes and extramural complications in the setting of CD. It also summarizes the relevant evidence available in the literature.
Zusammenfassung
Hintergrund
Die diffusionsgewichtete MRT (DWI) wird mittlerweile routinemäßig in der abdominellen Bildgebung eingesetzt. Neben tumorlösen Veränderungen lassen sich auch entzündliche Veränderungen durch eine Diffusionsrestriktion abgrenzen und diagnostizieren. Auch im Rahmen der Magnetresonanztomographie (MRT) des Dünn- und Dickdarms wird die DWI zunehmend eingesetzt.
Fragestellung
Technische Aspekte der DWI, Rolle der DWI in der Diagnostik des M. Crohn, Graduierung des Schweregrads, Therapiemonitoring der Erkrankung wurden erörtert.
Material und Methoden
Leitlinien, Grundlagen- und Übersichtsarbeiten wurden analysiert.
Ergebnisse
Die DWI ist eine spezielle MRT-Technik, die die Diffusion von Wassermolekülen in biologischem Gewebe darstellt. Im Rahmen der MRT des Dünn- und Dickdarms ist die DWI für die Diagnose von entzündlichen Darmerkrankungen und deren Verlaufsbeurteilung unter Therapie hilfreich. Mit der DWI lassen sich nicht nur intra- und transmurale Veränderungen, sondern auch extramurale pathologische Veränderungen und Komplikationen darstellen. Allerdings hat die DWI auch ihre Limitationen und Schwierigkeiten.
Schlussfolgerung
Der vorliegende Artikel bietet einen umfassenden Überblick über den Einsatz der DWI für die diagnostische Bewertung von Darmwandveränderungen und extramuralen Komplikationen im Rahmen des M. Crohn und fasst die relevanten in der Literatur verfügbaren Erkenntnisse zusammen.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Magnetic resonance imaging (MRI) of the small and large intestine is a diagnostic method that enables assessment of the bowel wall as well as extraluminal changes, structures, and other abdominal organs. Due to the absence of radiation exposure, it is particularly suitable for patients with chronic inflammatory bowel diseases such as Crohn’s disease (also referred to as CD), who require frequent follow-up examinations and would otherwise be exposed to cumulative radiation from CT scans.
Diffusion-weighted MRI (DWI) is now routinely used in abdominal imaging. In comparison to morphological sequences, it provides additional information about cell integrity based on Brownian molecular motion. In cases of acute inflammatory bowel wall pathologies and complications such as abscesses, there is generally restricted diffusion. Therefore, DWI is increasingly being incorporated into the routine protocol of MRI of the small and large intestine. This article aims to explore the technical requirements and limitations of DWI, as well its role in the assessment of Crohn’s disease.
Background
Crohn’s disease is a chronic inflammatory condition that affects the walls of the digestive system, including various parts of the gastrointestinal (GI) tract. It is characterized by recurrent episodes of worsening symptoms and significantly compromises a patient’s quality of life. Crohn’s disease is often characterized by transmural involvement, leading to formation of strictures, fistulas, and abscesses [32]. When evaluating transmural involvement in Crohn’s disease, radiological assessment plays a crucial role as it provides valuable information beyond what can be observed through endoscopy, which is limited to examining the mucosal layer.
MR enterography/enteroclysis (MRE), as a radiation-free imaging method, can visualize bowel wall changes as well as extraluminal structures and pathologies. Therefore, it is recommended in the European Crohnʼs and Colitis Organisation and European Society of Gastrointestinal and Abdominal Radiology (ECCO-ESGAR) Guideline for Diagnostic Assessment of 2018 for evaluating small bowel involvement in patients with newly diagnosed chronic inflammatory bowel disease [23]. The current German S3 guideline “Diagnosis and treatment of Crohn’s disease” by the German Society of Gastroenterology, Digestive and Metabolic Diseases (DGVS) recommends MRE for initial diagnosis of the small intestine [36]. In addition to initial diagnosis, imaging is also used to monitor and evaluate treatment effects of biologics or immunosuppressants.
Diffusion-weighted MRI can provide qualitative and quantitative information about tissue cellularity and membrane integrity. It is based on diffusion of water in the body and measures the movement of water molecules in the extracellular and intracellular space. Tissues with high cell density, such as in inflammatory processes, result in constriction of the extracellular space and higher density of hydrophobic cell membranes, leading to restricted diffusion. Abscesses for example are diffusion restricted due their high viscosity and inflammatory dense cellular composition. In addition to visual and qualitative evaluation of the diffusion-weighted sequences, the restriction of diffusion can also be assessed quantitatively using the apparent diffusion coefficient (ADC) value as visualized in the ADC map, which is generated from the diffusion-weighted image data with different b‑values [2]. Therefore, DWI is a promising tool for imaging assessment and quantification of intestinal inflammation in Crohn’s disease. It is particularly attractive for patients with intolerance to gadolinium-containing contrast agents or very severely impaired renal function, so that contrast agent administration should be critically considered according to European Society of Urogenital Radiology (ESUR) guidelines [14], since its use does not require contrast agent administration.
Technical aspects
Factors like bowel peristalsis, motion artifacts, and the presence of strictures can affect DWI image quality and interpretation. Optimizing imaging techniques, including appropriate patient preparation and sequence parameters, is crucial for obtaining reliable and accurate DWI results [17].
On the one hand, the bowel should be cleansed and emptied by fasting one day before the examination, except for clear liquids and regular medications. In case of additional evaluation of the colon, bowel cleansing as in colonoscopy is necessary. On the other hand, intraluminal contrast agents are used to achieve uniform and sufficient distension of the small intestine. This helps to reduce susceptibility artifacts caused by air and allows for better assessment of the intestinal wall, especially in relation to active inflammation of the terminal ileum [10, 18]. Biphasic contrast agents such as mannitol, sorbitol, and methylcellulose with high T2 signal and low T1 signal are predominantly used. In MRE, intraluminal contrast agents are administered orally: the intake of approximately 1000–1500 ml oral contrast agent should be spread over a period of 30–60 min through continuous drinking. In MR enteroclysis, the intraluminal contrast agent is administered via a nasojejunal tube, and the bowel is filled and monitored by MR fluoroscopy. However, due to the inconvenience for the patient and the long time required, this procedure is less commonly used [17]. Furthermore, to reduce bowel peristalsis and motion artifacts during image acquisition, international guidelines recommend administration of intravenous spasmolytics such as butylscopolamine or glucagon [10, 12, 37]. Park et al. showed that omitting butylscopolamine caused a greater loss in sensitivity of DWI-MRE than false-positive reduction for diagnosing bowel inflammation in CD and that DWI-MRE without butylscopolamine underestimates the extent of bowel inflammation in CD [29].
The recommended sequences for MRE include [10, 12, 37] steady-state free precession (SSFP) sequences, T2-weighted single-shot spin-echo sequences, and T1-weighted 3D gradient-echo sequences with contrast enhancement and fat saturation. The diffusion-weighted sequence can be integrated into this routine protocol. MRE using diffusion-weighted sequences can be performed on both 1.5 T and 3 T scanners using a combined body–phased-array surface coil with an integrated spine coil, whereby the 3 T magnet does not have a clear advantage over the 1.5 T magnet [15]. However, there is a lack of sufficiently validated clinical comparative studies.
For intestinal imaging, diffusion-weighted images with at least two different b‑values (b = 0–100 and b = 600–1100 s/mm2) are generated, from which the ADC map is generated. Most studies used either a single b‑value (excluding b = 0) or multiple b‑values in the range of 500 to 1000 s/mm2 empirically [4, 5, 8, 11, 22, 34, 35].
As a routine part of the authors’ MRE examination, they perform a DWI scan during free breathing. Parallel imaging is crucial and diminishes susceptibility, chemical shift, and motion artifacts [27]. In addition, the use of compressed sensing can lead to a reduction in the acquisition time. The DWI scan is performed in the transverse plane. To enable the required broad coverage from the upper abdomen to the pelvis, two separate transverse scans (upper scan for the abdomen and lower scan for the pelvis) are created to obtain high-resolution images. Each scan takes about 3–3.5 min.
Detection of acute inflammatory intestinal changes in Crohn’s disease using DWI and its diagnostic performance
Several radiological assessment tools are used to evaluate transmural involvement and assess disease activity in Crohn’s disease, including the Magnetic Resonance Index of Activity (MaRIA) and the Clermont score. The MaRIA is a commonly cited enterographic index for evaluating the activity of Crohn’s disease. It assesses various specific features observed on MRE images, including the presence of bowel wall thickening, edema, ulceration, measurements of wall signal intensity before and after intravenous contrast administration, and relative contrast enhancement of the intestinal wall. Each of these features is assigned a score based on its severity, and the individual scores are then combined to generate an overall MaRIA score [31, 32]. The Clermont score published by Buisson et al. is a DWI-based index for quantification of disease activity, which combines conventional MRE parameters derived from the MaRIA index such as bowel wall thickness [6, 32] in ileal Crohn’s disease with DWI parameters such as ADC measurements. An external validation by the same group found a strong correlation between the Clermont score and the MaRIA score in Crohn’s disease affecting the ileum. However, this correlation was not as strong in Crohn’s disease affecting the colon [16].
Numerous studies have investigated the diagnostic performance of DWI in detecting acute inflammatory intestinal changes in patients with Crohn’s disease [13, 25, 27]. These studies have demonstrated that DWI can detect and characterize active inflammation, including increased bowel wall thickness, edema, and the presence of ulcers.
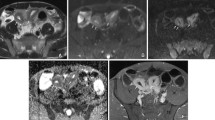
Increased bowel wall thickness is a key finding of active Crohn’s disease in DWI. The inflamed bowel wall appears hyperintense on DWI due to increased cellularity and hence restricted diffusion (Fig. 1). DWI is sensitive to the presence of edema within the bowel wall, which is a common feature of active inflammation in Crohn’s disease. Edema appears as hyperintense signal in DWI due to increased water content. Detecting and characterizing edema in DWI helps to assess the extent and severity of inflammation. Ulceration, a characteristic finding in Crohn’s disease, can also be identified using DWI. Ulcers manifest as focal areas of hyperintensity in DWI due to restricted diffusion caused by cellular debris and inflammatory exudates. Identifying and analyzing ulcers in DWI provides valuable information about the location and severity of inflammation.
33-year-old male with Crohn’s disease. In the T2-weighted single-shot fast spin-echo image (a), there is long-segmented wall thickening (arrow) with ulcerations in the distal ileum. The acute inflammation (arrow) exhibits a pronounced uptake of contrast agent in T1-weighted gradient-echo image with fat saturation (b). In the diffusion-weighted sequence, a strong diffusion restriction due to the inflammation, resulting in a high signal intensity (arrow) in the image with a b-value of 800 s/mm2 (c) and a corresponding low signal intensity (arrow) in the apparent diffusion coefficient (ADC) map (d) is seen
In a recent study, Bae et al. showed that a modified simplified magnetic resonance index of activity (sMARIA) that uses DWI instead of ulcers in its calculation showed diagnostic performance comparable with sMARIA using conventional MRE with contrast-enhanced sequences. In addition, DWI can improve the diagnostic performance of non-contrast MRE in assessing active inflammation in Crohn’s disease [1]. According to a systematic review and meta-analysis by Choi et al., the diagnostic performance of diffusion-weighted imaging (DWI) enterography for detecting active bowel inflammation in Crohn’s disease varies widely [11]. Sensitivities ranged from 68 to 100%, while specificities ranged from 51 to 100%. The accuracy of DWI enterography may have been overestimated in certain studies due to biases such as not adequately blinding the readers or using conventional contrast-enhanced MR enterography as the reference standard [11]. When unbiased studies were considered, the meta-analytic summary estimates showed a sensitivity of 79% and specificity of 61% for DWI enterography. Notably, false-positive results on DWI enterography tend to occur in bowel loops with inadequate fluid distension, especially in the colorectum and jejunum [19, 21, 26]. False positives can also arise from incidental findings such as lymphoid nodular hyperplasia of the terminal ileum [11]. The criteria for defining abnormally restricted intestinal mural diffusion on DWI enterography vary among studies, and quantitative thresholds for distinguishing inflamed from noninflamed bowel have yet to be validated and standardized [11].
The literature presents conflicting results regarding the direct comparison between DWI and contrast-enhanced (CE-MRE): Seo et al. demonstrated high concordance between DWI combined with T2-weighted sequences and CE-MRE in identifying inflamed bowel segments [34]. However, Shenoy-Bhangle et al. found that DWI had lower accuracy but higher sensitivity compared to CE-MRE in children [35].
Overall, studies primarily show agreement that the combination of DWI and CE-MRE has a positive impact on the detection of inflamed bowel segments. The addition of DWI seems to improve accuracy [30, 35], sensitivity [35], and diagnostic confidence [20]. Therefore, DWI plays an important role especially when contrast administration is contraindicated.
The results indicate that DWI enterography holds promise for detecting active bowel inflammation in Crohn’s disease, but its diagnostic performance is influenced by factors such as bias, fluid distention, and the lack of standardized criteria. Further research is needed to improve the accuracy and reproducibility of DWI enterography in identifying complications and monitoring disease activity in Crohn’s disease.
Detection of complications in the context of Crohn’s disease
Crohn’s disease is a chronic inflammatory bowel disease that can lead to various complications including fistulas and abscesses. These complications significantly impact the quality of life for individuals with Crohn’s disease and require prompt diagnosis and appropriate management. Fistulas and abscesses in Crohn’s disease are primarily attributed to the transmural inflammation and ulceration that characterize the condition [32]. The chronic inflammation weakens the intestinal walls, leading to the formation of abnormal connections between different segments of the gastrointestinal tract or between the intestine and adjacent organs or structures.
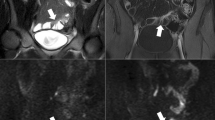
Diffusion-weighted imaging (DWI) has emerged as a valuable tool in the detection and characterization of abscesses and fistulas in Crohn’s disease. Fistulas manifest as areas of hyperintensity on DWI, connecting the affected bowel segment to adjacent structures (Fig. 2). Abscesses, on the other hand, exhibit hyperintense signals with restricted diffusion due to the presence of purulent material (Fig. 3). DWI can help delineate the extent and involvement of these complications, thus assisting in treatment planning [26, 33].
42-year-old male with Crohn’s disease and enteroenteric fistula. Axial T2-weighted single-shot fast spin-echo image (a) shows a hypointense linear structure (arrow) connecting inflamed loops of the small bowel consistent with enteroenteric fistula. The fistula (arrow) shows bright signal on diffusion-weighted MRI image (b; b‑value = 800 s/mm2), indicating restricted diffusion
29-year-old male with Crohn’s disease and abdominal abscess. Transverse T2-weighted single-shot fast spin-echo image without fat saturation (a) shows loculated collection with thick rim (arrow). In transverse post-contrast fat-suppressed T1-weighted gradient-echo image (b), perilesional enhancement (arrow) is noted around the collection. The collection (arrow) demonstrates restricted diffusion with bright signal in single-shot echo planar diffusion-weighted image (b = 800 mm2/s; c) and low signal in the apparent diffusion coefficient map (d)
Several studies have investigated the sensitivity and specificity of DWI in identifying these complications. Varying sensitivity rates for DWI in detecting abscesses and fistulas in Crohn’s disease have been reported. For abscess detection, sensitivities ranging from 76 to 100% have been found in different studies [3, 26]. The variability in sensitivity can be attributed to factors such as the size and location of the abscess, the experience of the radiologist, and the specific DWI protocol used. In the case of fistulas, DWI has shown high sensitivity rates, often exceeding 90% [3, 9]. This is particularly evident for perianal fistulas, where DWI has demonstrated excellent sensitivity in detecting the presence and extent of fistulous tracts. However, sensitivity rates may vary depending on the location and complexity of fistulas within the gastrointestinal tract. Overall, DWI has shown promising diagnostic performance in the detection of abscesses and fistulas in Crohn’s disease. When combined with conventional imaging techniques such as MRE, DWI can enhance the accuracy and confidence in diagnosing these complications.
Monitoring of inflammatory activity
The correlation between the degree of mural diffusion restriction assessed through DWI enterography and the severity of bowel inflammation in Crohn’s disease suggests that quantitative assessment using DWI enterography might be useful. A reduction in DWI hyperintensity and an increase in ADC values suggest a decrease in inflammatory activity and good response to treatment. Quantitative assessment aims to stratify patients based on inflammation severity, which would be important for personalized treatment approaches and monitoring changes in inflammation after treatment. While some studies have shown promising results [6, 7, 24], the technical reproducibility of quantitative measurements in the bowel wall needs further clarification. Variability in ADC measurements due to different scanning techniques, inter-scanner variability, and within-scanner variability adds complexity to the interpretation of quantitative parameters obtained from DWI enterography [28].
Moreover, the measurement of quantitative parameters in the narrow bowel wall, particularly in patients with multiple diseased segments, can be challenging and may introduce observer variability.
The role of DWI enterography in monitoring inflammatory activity in Crohn’s disease can be better evaluated through longitudinal follow-up studies that directly compare imaging results before and after treatment in the same patient. Most existing evidence is from cross-sectional studies, and there are limited published data on longitudinal monitoring.
Quantitative assessment using DWI enterography shows potential in stratifying patients based on inflammation severity and monitoring changes for inflammatory activity. However, the technical reproducibility of quantitative measurements and the need for longitudinal studies to evaluate posttherapeutic monitoring warrant further investigation.
Practical conclusion
-
Diffusion-weighted MRI (DWI) is a promising tool in the diagnosis of Crohn’s disease and the assessment of disease severity and treatment response. Therefore, DWI may be considered as a valuable adjunct to contrast-enhanced MR enterography.
-
DWI can detect and characterize active inflammation, including increased bowel wall thickness, edema, and ulcers and helps in detection of complications such as fistulas and abscesses.
-
Further research is needed to validate and standardize quantitative thresholds to determine the adequate role of DWI in the evaluation of bowel inflammation in Crohn’s disease.
-
Patient preparation, including bowel cleansing, and the use of intraluminal contrast agents are indispensable for obtaining reliable DWI findings.
-
Be aware of the limitations and potential false-positive results of DWI, especially in bowel loops with inadequate distention and incidental findings.
References
Bae H, Seo N, Kang EA et al (2023) Validation of the simplified magnetic resonance index of activity by using DWI without gadolinium enhancement to evaluate bowel inflammation in Crohn’s disease. Eur Radiol 33:3266–3275
Baliyan V, Das CJ, Sharma R et al (2016) Diffusion weighted imaging: technique and applications. World J Radiol 8:785–798
Barat M, Hoeffel C, Bouquot M et al (2019) Preoperative evaluation of small bowel complications in Crohn’s disease: comparison of diffusion-weighted and contrast-enhanced MR imaging. Eur Radiol 29:2034–2044
Bhatnagar G, Dikaios N, Prezzi D et al (2015) Changes in dynamic contrast-enhanced pharmacokinetic and diffusion-weighted imaging parameters reflect response to anti-TNF therapy in Crohn’s disease. BJR 88:20150547
Buisson A, Hordonneau C, Goutte M et al (2015) Diffusion-weighted magnetic resonance imaging is effective to detect ileocolonic ulcerations in Crohn’s disease. Aliment Pharmacol Ther 42:452–460
Buisson A, Joubert A, Montoriol PF et al (2013) Diffusion-weighted magnetic resonance imaging for detecting and assessing ileal inflammation in Crohn’s disease. Aliment Pharmacol Ther 37:537–545
Caruso A, D’incà R, Scarpa M et al (2014) Diffusion-weighted magnetic resonance for assessing ileal Crohn’s disease activity. Inflamm Bowel Dis 20:1575–1583
Catalano OA, Gee MS, Nicolai E et al (2016) Evaluation of quantitative PET/MR enterography biomarkers for discrimination of inflammatory strictures from fibrotic strictures in Crohn disease. Radiology 278:792–800
Cavusoglu M, Duran S, Sözmen Cılız D et al (2017) Added value of diffusion-weighted magnetic resonance imaging for the diagnosis of perianal fistula. Diagn Interv Imaging 98:401–408
Chatterji M, Fidler JL, Taylor SA et al (2021) State of the art MR enterography technique. Top Magn Reson Imaging 30:3–11
Choi SH, Kim KW, Lee JY et al (2016) Diffusion-weighted magnetic resonance enterography for evaluating bowel inflammation in Crohn’s disease: a systematic review and meta-analysis. Inflamm Bowel Dis 22:669–679
Costa-Silva L, Brandão AC (2013) MR enterography for the assessment of small bowel diseases. Magn Reson Imaging Clin N Am 21:365–383
Dogan I, Tastekin D, Karabulut S et al (2022) Capecitabine and temozolomide (CAPTEM) is effective in metastatic well-differentiated gastrointestinal neuroendocrine tumors. J Dig Dis 23:493–499
European Society of Urogenital Radiology TH, Stacul E, Bellin M et al (2018) ESUR guidelines on contrast agents 10.0. Contrast Media Safety Committee. https://www.esur.org/esur-guidelines-on-contrast-agents/. Accessed 18.07.2023
Fiorino G, Bonifacio C, Padrenostro M et al (2013) Comparison between 1.5 and 3.0 Tesla magnetic resonance enterography for the assessment of disease activity and complications in ileo-colonic Crohn’s disease. Dig Dis Sci 58:3246–3255
Hordonneau C, Buisson A, Scanzi J et al (2014) Diffusion-weighted magnetic resonance imaging in ileocolonic Crohn’s disease: validation of quantitative index of activity. Am J Gastroenterol 109:89–98
Ingenerf M, Schmid-Tannwald C (2023) Magnetic resonance enterography/enteroclysis : Technical aspects and indications. Radiol (heidelb) 63:429–434
Jesuratnam-Nielsen K, Løgager VB, Rezanavaz-Gheshlagh B et al (2015) Plain magnetic resonance imaging as an alternative in evaluating inflammation and bowel damage in inflammatory bowel disease—a prospective comparison with conventional magnetic resonance follow-through. Scand J Gastroenterol 50:519–527
Kim KJ, Lee Y, Park SH et al (2015) Diffusion-weighted MR enterography for evaluating Crohn’s disease: how does it add diagnostically to conventional MR enterography? Inflamm Bowel Dis 21:101–109
Kinner S, Blex S, Maderwald S et al (2014) Addition of diffusion-weighted imaging can improve diagnostic confidence in bowel MRI. Clin Radiol 69:372–377
Kiryu S, Dodanuki K, Takao H et al (2009) Free-breathing diffusion-weighted imaging for the assessment of inflammatory activity in Crohn’s disease. J Magn Reson Imaging 29:880–886
Kovanlikaya A, Beneck D, Rose M et al (2015) Quantitative apparent diffusion coefficient (ADC) values as an imaging biomarker for fibrosis in pediatric Crohn’s disease: preliminary experience. Abdom Imaging 40:1068–1074
Maaser C, Sturm A, Vavricka SR et al (2019) ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications. J Crohns Colitis 13:144–164
Neubauer H, Pabst T, Dick A et al (2013) Small-bowel MRI in children and young adults with Crohn disease: retrospective head-to-head comparison of contrast-enhanced and diffusion-weighted MRI. Pediatr Radiol 43:103–114
Ninivaggi V, Missere M, Restaino G et al (2016) MR-enterography with diffusion weighted imaging: ADC values in normal and pathological bowel loops, a possible threshold ADC value to differentiate active from inactive Crohn’s disease. Eur Rev Med Pharmacol Sci 20:4540–4546
Oto A, Schmid-Tannwald C, Agrawal G et al (2011) Diffusion-weighted MR imaging of abdominopelvic abscesses. Emerg Radiol 18:515–524
Oto A, Zhu F, Kulkarni K et al (2009) Evaluation of diffusion-weighted MR imaging for detection of bowel inflammation in patients with Crohn’s disease. Acad Radiol 16:597–603
Park SH (2016) DWI at MR Enterography for Evaluating Bowel Inflammation in Crohn Disease. Ajr Am J Roentgenol 207:40–48
Park SH, Huh J, Park SH et al (2017) Diffusion-weighted MR enterography for evaluating Crohn’s disease: Effect of anti-peristaltic agent on the diagnosis of bowel inflammation. Eur Radiol 27:2554–2562
Qi F, Jun S, Qi QY et al (2015) Utility of the diffusion-weighted imaging for activity evaluation in Crohn’s disease patients underwent magnetic resonance enterography. BMC Gastroenterol 15:12
Rimola J, Rodriguez S, García-Bosch O et al (2009) Magnetic resonance for assessment of disease activity and severity in ileocolonic Crohn’s disease. Gut 58:1113–1120
Rozendorn N, Amitai MM, Eliakim RA et al (2018) A review of magnetic resonance enterography-based indices for quantification of Crohn’s disease inflammation. Therap Adv Gastroenterol 11:1756284818765956
Schmid-Tannwald C, Agrawal G, Dahi F et al (2012) Diffusion-weighted MRI: role in detecting abdominopelvic internal fistulas and sinus tracts. J Magn Reson Imaging 35:125–131
Seo N, Park SH, Kim KJ et al (2016) MR Enterography for the evaluation of small-bowel inflammation in Crohn disease by using diffusion-weighted imaging without intravenous contrast material: a prospective Noninferiority study. Radiology 278:762–772
Shenoy-Bhangle AS, Nimkin K, Aranson T et al (2016) Value of diffusion-weighted imaging when added to magnetic resonance enterographic evaluation of Crohn disease in children. Pediatr Radiol 46:34–42
Sturm A, Atreya R, Bettenworth D et al (2022) Aktualisierte S3-Leitlinie „Diagnostik und Therapie des Morbus Crohn“ der Deutschen Gesellschaft für Gastroenterologie, Verdauungs- und Stoffwechselkrankheiten (DGVS) – August 2021 – AWMF-Registernummer: 021-004. Z Gastroenterol 60:332–418
Taylor SA, Avni F, Cronin CG et al (2017) The first joint ESGAR/ESPR consensus statement on the technical performance of cross-sectional small bowel and colonic imaging. Eur Radiol 27:2570–2582
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M. Ingenerf and C. Schmid-Tannwald declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies mentioned were in accordance with the ethical standards indicated in each case.
The supplement containing this article is not sponsored by industry.
Additional information

Scan QR code & read article online
Rights and permissions
About this article
Cite this article
Ingenerf, M., Schmid-Tannwald, C. Diffusion-weighted imaging in Crohn’s disease. Radiologie 63 (Suppl 2), 27–33 (2023). https://doi.org/10.1007/s00117-023-01191-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00117-023-01191-y
Keywords
- Magnetic resonance imaging
- Inflammatory bowel diseases
- Intestinal fistula
- Abscess
- Gastrointestinal tract