Abstract.
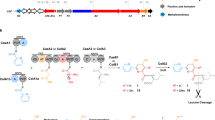
Nonribosomal peptides and polyketides represent a large class of natural products that show an extreme structural diversity and broad pharmacological relevance. They are synthesized from simple building blocks such as amino or carboxy acids and malonate derivatives on multimodular enzymes called nonribosomal peptide synthetases (NRPSs) and polyketide synthases (PKSs), respectively. Although utilizing different substrates, NRPSs and PKSs show striking similarities in the modular architecture of their catalytic domains and product assembly-line mechanism. Among these compounds are well known antibiotics (penicillin, vancomycin and erythromycin) as well as potent immunosuppressive agents (cyclosporin, rapamycin and FK 506). This review focuses on the modular organization of NRPSs, PKSs and mixed NRPS/PKS systems and how modules and domains that build up the biosynthetic templates can be exploited for the rational design of recombinant enzymes capable of synthesizing novel compounds.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Schwarzer, D., Marahiel, M. Multimodular biocatalysts for natural product assembly. Naturwissenschaften 88, 93–101 (2001). https://doi.org/10.1007/s001140100211
Issue Date:
DOI: https://doi.org/10.1007/s001140100211