Abstract
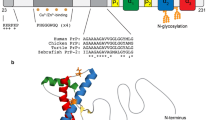
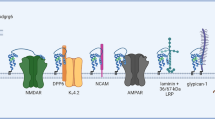
Cellular prion protein (PrPC) is a highly conserved glycoprotein, present both anchored in the cell membrane and soluble in the extracellular medium. It has a diversity of ligands and is variably expressed in numerous tissues and cell subtypes, most notably in the central nervous system (CNS). Its importance has been brought to light over the years both under physiological conditions, such as embryogenesis and immune system homeostasis, and in pathologies, such as cancer and neurodegenerative diseases. During development, PrPC plays an important role in CNS, participating in axonal growth and guidance and differentiation of glial cells, but also in other organs such as the heart, lung, and digestive system. In diseases, PrPC has been related to several types of tumors, modulating cancer stem cells, enhancing malignant properties, and inducing drug resistance. Also, in non-neoplastic diseases, such as Alzheimer’s and Parkinson’s diseases, PrPC seems to alter the dynamics of neurotoxic aggregate formation and, consequently, the progression of the disease. In this review, we explore in detail the multiple functions of this protein, which proved to be relevant for understanding the dynamics of organism homeostasis, as well as a promising target in the treatment of both neoplastic and degenerative diseases.



Similar content being viewed by others
Data availability
Not applicable.
References
Linden R, Cordeiro Y, Lima LMTR (2012) Allosteric function and dysfunction of the prion protein. Cell Mol Life Sci 69:1105–1124. https://doi.org/10.1007/s00018-011-0847-7
Castle AR, Gill AC (2017) Physiological functions of the cellular prion protein. Front Mol Biosci 4:1–25. https://doi.org/10.3389/fmolb.2017.00019
Falker C, Hartmann A, Guett I et al (2016) Exosomal cellular prion protein drives fibrillization of amyloid beta and counteracts amyloid beta-mediated neurotoxicity. J Neurochem 137:88–100. https://doi.org/10.1111/jnc.13514
Leblanc P, Arellano-Anaya ZE, Bernard E et al (2017) Isolation of exosomes and microvesicles from cell culture systems to study prion transmission. In: Methods in Mol Biol 153–176
Adle-Biassette H, Verney C, Peoc’h K, et al (2006) Immunohistochemical expression of prion protein (PrPC) in the human forebrain during development. J Neuropathol Exp Neurol 65:698–706. https://doi.org/10.1097/01.jnen.0000228137.10531.72
Linden R, Martins VR, Prado MAM et al (2008) Physiology of the prion protein. Physiol Rev 88:673–728. https://doi.org/10.1152/physrev.00007.2007
Moser M, Colello RJ, Pott U, Oesch B (1995) Developmental expression of the prion protein gene in glial cells. Neuron 14:509–517. https://doi.org/10.1016/0896-6273(95)90307-0
Watts JC, Bourkas MEC, Arshad H (2018) The function of the cellular prion protein in health and disease. Acta Neuropathol 135:159–178. https://doi.org/10.1007/s00401-017-1790-y
Arantes C, Nomizo R, Lopes MH et al (2009) Prion protein and its ligand stress inducible protein 1 regulate astrocyte development. Glia 57:1439–1449. https://doi.org/10.1002/glia.20861
Hartmann CA, Martins VR, Lima FRS (2013) High levels of cellular prion protein improve astrocyte development. FEBS Lett 587:238–244. https://doi.org/10.1016/j.febslet.2012.11.032
Lima FRS, Arantes CP, Muras AG et al (2007) Cellular prion protein expression in astrocytes modulates neuronal survival and differentiation. J Neurochem 103:2164–2176. https://doi.org/10.1111/j.1471-4159.2007.04904.x
Chadi S, Young R, Le Guillou S et al (2010) Brain transcriptional stability upon prion protein-encoding gene invalidation in zygotic or adult mouse. BMC Genomics 11:448. https://doi.org/10.1186/1471-2164-11-448
Halliez S, Passet B, Martin-Lannerée S, et al (2014) To develop with or without the prion protein. Front Cell Dev Biol 2:1–10. https://doi.org/10.3389/fcell.2014.00058
Weissmann C, Flechsig E (2003) PrP knock-out and PrP transgenic mice in prion research. Br Med Bull 66:43–60. https://doi.org/10.1093/bmb/66.1.43
Mallucci GR (2002) Post-natal knockout of prion protein alters hippocampal CA1 properties, but does not result in neurodegeneration. EMBO J 21:202–210. https://doi.org/10.1093/emboj/21.3.202
Manson JC, Clarke AR, Hooper ML et al (1994) 129/Ola mice carrying a null mutation in PrP that abolishes mRNA production are developmentally normal. Mol Neurobiol 8:121–127. https://doi.org/10.1007/BF02780662
Richt JA, Kasinathan P, Hamir AN et al (2007) Production of cattle lacking prion protein. Nat Biotechnol 25:132–138. https://doi.org/10.1038/nbt1271
White MD, Farmer M, Mirabile I et al (2008) Single treatment with RNAi against prion protein rescues early neuronal dysfunction and prolongs survival in mice with prion disease. Proc Natl Acad Sci U S A 105:10238–10243. https://doi.org/10.1073/pnas.0802759105
Yu G, Chen J, Xu Y et al (2009) Generation of goats lacking prion protein. Mol Reprod Dev 76:3
Büeler H, Fischer M, Lang Y et al (1992) Normal development and behaviour of mice lacking the neuronal cell-surface PrP protein. Nature 356:577–582. https://doi.org/10.1038/356577a0
Gerlai R (1996) Gene-targeting studies of mammalian behavior: is it the mutation or the background genotype? Trends Neurosci 19:177–181. https://doi.org/10.1016/S0166-2236(96)20020-7
Nuvolone M, Hermann M, Sorce S et al (2016) Strictly co-isogenic C57BL/6J-Prnp-/- mice: a rigorous resource for prion science. J Exp Med 213:313–327. https://doi.org/10.1084/jem.20151610
Norrby E (2011) Prions and protein-folding diseases. J Intern Med 270:1–14
Prusiner SB, Scott MR, DeArmond SJ, Cohen FE (1998) Prion protein biology. Cell 93:337–348
Cox TO, Gunther EC, Brody AH et al (2019) Anti-PrPC antibody rescues cognition and synapses in transgenic alzheimer mice. Ann Clin Transl Neurol 6:554–574. https://doi.org/10.1002/acn3.730
Linden R (2017) The biological function of the prion protein: a cell surface scaffold of signaling modules. Front Mol Neurosci 10:1–19. https://doi.org/10.3389/fnmol.2017.00077
Pan Y, Zhao L, Liang J et al (2006) Cellular prion protein promotes invasion and metastasis of gastric cancer. FASEB J 20:1886–1888. https://doi.org/10.1096/fj.06-6138fje
Wang Y, Yu S, Huang D et al (2016) Cellular prion protein mediates pancreatic cancer cell survival and invasion through association with and enhanced signaling of Notch1. Am J Pathol 186:2945–2956. https://doi.org/10.1016/j.ajpath.2016.07.010
Yang X, Zhang Y, Zhang L et al (2014) Prion protein and cancers. Acta Biochim Biophys Sin (Shanghai) 46:431–440. https://doi.org/10.1093/abbs/gmu019
Zhou L, Shang Y, Liu C et al (2014) Overexpression of PrPc, combined with MGr1-Ag/37LRP, is predictive of poor prognosis in gastric cancer. Int J Cancer 135:2329–2337. https://doi.org/10.1002/ijc.28883
Cashman NR, Loertscher R, Nalbantoglu J et al (1990) Cellular isoform of the scrapie agent protein participates in lymphocyte activation. Cell 61:185–192. https://doi.org/10.1016/0092-8674(90)90225-4
Isaacs JD, Jackson GS, Altmann DM (2006) The role of the cellular prion protein in the immune system. Clin Exp Immunol 146:1–8. https://doi.org/10.1111/j.1365-2249.2006.03194.x
Esiri MM, Carter J, Ironside JW (2000) Prion protein immunoreactivity in brain samples from an unselected autopsy population: findings in 200 consecutive cases. Neuropathol Appl Neurobiol 26:273–284. https://doi.org/10.1046/j.1365-2990.2000.00239.x
Harris DA, Lele P, Snider WD (1993) Localization of the mRNA for a chicken prion protein by in situ hybridization. Proc Natl Acad Sci U S A 90:4309–4313. https://doi.org/10.1073/pnas.90.9.4309
McLennan NF, Rennison KA, Bell JE, Ironside JW (2001) In situ hybridization analysis of PrP mRNA in human CNS tissues. Neuropathol Appl Neurobiol 27:373–383. https://doi.org/10.1046/j.0305-1846.2001.00343.x
Tremblay P, Bouzamondo-Bernstein E, Heinrich C et al (2007) Developmental expression of PrP in the post-implantation embryo. Brain Res 1139:60–67. https://doi.org/10.1016/j.brainres.2006.12.055
Bendheim PE, Brown HR, Rudelli RD et al (1992) Nearly ubiquitous tissue distribution of the scrapie agent precursor protein. Neurology 42:149–156. https://doi.org/10.1212/wnl.42.1.149
Manson J, West JD, Thomson V et al (1992) The prion protein gene: a role in mouse embryogenesis? Development 115:117–122. https://doi.org/10.1242/dev.115.1.117
Peralta OA, Huckle WR, Eyestone WH (2011) Expression and knockdown of cellular prion protein (PrPC) in differentiating mouse embryonic stem cells. Differentiation 81:68–77. https://doi.org/10.1016/j.diff.2010.09.181
Steele AD, Emsley JG, Özdinler PH et al (2006) Prion protein (PrP c ) positively regulates neural precursor proliferation during developmental and adult mammalian neurogenesis. Proc Natl Acad Sci 103:3416–3421. https://doi.org/10.1073/pnas.0511290103
Collinge J, Whittington MA, Sidle KCL et al (1994) Prion protein is necessary for normal synaptic function. Nature 370:295–297. https://doi.org/10.1038/370295a0
Miele G, Jeffrey M, Turnbull D et al (2002) Ablation of cellular prion protein expression affects mitochondrial numbers and morphology. Biochem Biophys Res Commun 291:372–377. https://doi.org/10.1006/bbrc.2002.6460
Málaga-Trillo E, Solis GP, Schrock Y et al (2009) Regulation of embryonic cell adhesion by the prion protein. PLoS Biol 7:0576–0590. https://doi.org/10.1371/journal.pbio.1000055
Amin L, Nguyen XTA, Rolle IG et al (2016) Characterization of prion protein function by focal neurite stimulation. J Cell Sci 129:3878–3891. https://doi.org/10.1242/jcs.183137
Nguyen XTA, Tran TH, Cojoc D, Legname G (2019) Copper binding regulates cellular prion protein function. Mol Neurobiol 56:6121–6133. https://doi.org/10.1007/s12035-019-1510-9
Bodrikov V, Solis GP, Stuermer CAO (2011) Prion protein promotes growth cone development through reggie/flotillin-dependent N-cadherin trafficking. J Neurosci 31:18013–18025. https://doi.org/10.1523/JNEUROSCI.4729-11.2011
Miyazawa K, Emmerling K, Manuelidis L (2010) Proliferative arrest of neural cells induces prion protein synthesis, nanotube formation, and cell-to-cell contacts. J Cell Biochem 111:239–247. https://doi.org/10.1002/jcb.22723
Santuccione A, Sytnyk V, Leshchyns’ka I, Schachner M, (2005) Prion protein recruits its neuronal receptor NCAM to lipid rafts to activate p59fyn and to enhance neurite outgrowth. J Cell Biol 169:341–354. https://doi.org/10.1083/jcb.200409127
Lee YJ, Baskakov IV (2013) The cellular form of the prion protein is involved in controlling cell cycle dynamics, self-renewal, and the fate of human embryonic stem cell differentiation. J Neurochem 124:310–322. https://doi.org/10.1111/j.1471-4159.2012.07913.x
Prodromidou K, Papastefanaki F, Sklaviadis T, Matsas R (2014) Functional cross-talk between the cellular prion protein and the neural cell adhesion molecule is critical for neuronal differentiation of neural stem/precursor cells. Stem Cells 32:1674–1687. https://doi.org/10.1002/stem.1663
Mehrabian M, Brethour D, Wang H et al (2015) The prion protein controls polysialylation of neural cell adhesion molecule 1 during cellular morphogenesis. PLoS ONE 10:e0133741. https://doi.org/10.1371/journal.pone.0133741
Halliez S, Martin-Lannerée S, Passet B et al (2015) Prion protein localizes at the ciliary base during neural and cardiovascular development and its depletion affects α-tubulin post-translational modifications. Sci Rep 5:17146. https://doi.org/10.1038/srep17146
Santos TG, Silva IG, B C-S, et al (2011) Enhanced neural progenitor/stem cells self-renewal via the interaction of stress-inducible protein 1 with the prion protein. Stem Cells 31:2136–2148. https://doi.org/10.1002/May
Skedsmo FS, Malachin G, Våge DI et al (2020) Demyelinating polyneuropathy in goats lacking prion protein. FASEB J 34:2359–2375. https://doi.org/10.1096/fj.201902588R
Guénard V, Montag D, Schachner M, Martini R (1996) Onion bulb cells in mice deficient for myelin genes share molecular properties with immature, differentiated non-myelinating, and denervated Schwann cells. Glia 18:27–38. https://doi.org/10.1002/(SICI)1098-1136(199609)18:1%3c27::AID-GLIA3%3e3.0.CO;2-0
Kretzschmar HA, Prusiner SB, Stowring LE, DeArmond SJ (1986) Scrapie prion proteins are synthesized in neurons. Am J Pathol 122:1–5
Bribián A, Fontana X, Llorens F et al (2012) Role of the cellular prion protein in oligodendrocyte precursor cell proliferation and differentiation in the developing and adult mouse CNS. PLoS ONE 7:e33872. https://doi.org/10.1371/journal.pone.0033872
Brown DR, Besinger A, Herms JW, Kretzschmar HA (1998) Microglial expression of the prion protein. NeuroReport 9:1425–1429. https://doi.org/10.1097/00001756-199805110-00032
Garcia-Abreu J, Cavalcante LA, Neto VM (1995) Differential patterns of laminin expression in lateral and medial midbrain glia. NeuroReport 6:761–764. https://doi.org/10.1097/00001756-199503270-00014
Aguzzi A (2003) Prions and the immune system: a trip through intestine, spleen, lymph nodes and nerves. ATTI Della Accad Naz Dei Lincei Rend Lincei Sci Fis E Nat 14:293–337. https://doi.org/10.1007/BF02904488
Burthem J, Urban B, Pain A, Roberts DJ (2001) The normal cellular prion protein is strongly expressed by myeloid dendritic cells. Blood 98:3733–3738. https://doi.org/10.1182/blood.V98.13.3733
Salvesen Ø, Tatzelt J, Tranulis MA (2019) The prion protein in neuroimmune crosstalk. Neurochem Int 130:104335. https://doi.org/10.1016/j.neuint.2018.11.010
Kim S, Han S, Lee YE et al (2016) Prion protein-deficient mice exhibit decreased CD4 T and LTi cell numbers and impaired spleen structure. Immunobiology 221:94–102. https://doi.org/10.1016/j.imbio.2015.07.017
Dodelet VC, Cashman NR (1998) Prion protein expression in human leukocyte differentiation. Blood 91:1556–1561. https://doi.org/10.1182/blood.v91.5.1556
Dürig J, Giese A, Schulz-Schaeffer W et al (2000) Differential constitutive and activation-dependent expression of prion protein in human peripheral blood leucocytes. Br J Haematol 108:488–495. https://doi.org/10.1046/j.1365-2141.2000.01881.x
Zhang CC, Steele AD, Lindquist S, Lodish HF (2006) Prion protein is expressed on long-term repopulating hematopoietic stem cells and its important for their self-renewal. Proc Natl Acad Sci U S A 103:2184–2189. https://doi.org/10.1073/pnas.0510577103
Siberchicot C, Gault N, Déchamps N et al (2020) Prion protein deficiency impairs hematopoietic stem cell determination and sensitizes myeloid progenitors to irradiation. Haematologica 105:1216–1222. https://doi.org/10.3324/HAEMATOL.2018.205716
Jeon J-W, Park B-C, Jung J-G et al (2013) The soluble form of the cellular prion protein enhances phagocytic activity and cytokine production by human monocytes via activation of ERK and NF-κB. Immune Netw 13:148. https://doi.org/10.4110/in.2013.13.4.148
Seong YJ, Sung PS, Jang YS et al (2015) Activation of human natural killer cells by the soluble form of cellular prion protein. Biochem Biophys Res Commun 464:512–518. https://doi.org/10.1016/j.bbrc.2015.06.172
Richardson DD, Tol S, Valle-Encinas E et al (2015) The prion protein inhibits monocytic cell migration by stimulating β1 integrin adhesion and uropod formation. J Cell Sci 128:3018–3029. https://doi.org/10.1242/jcs.165365
Martínez del Hoyo G, López-Bravo M, Metharom P et al (2006) Prion protein expression by mouse dendritic cells is restricted to the nonplasmacytoid subsets and correlates with the maturation state. J Immunol 177:6137–6142. https://doi.org/10.4049/jimmunol.177.9.6137
Isaacs JD, Garden OA, Kaur G et al (2008) The cellular prion protein is preferentially expressed by CD4+ CD25+ Foxp3+ regulatory T cells. Immunology 125:313–319. https://doi.org/10.1111/j.1365-2567.2008.02853.x
Hu W, Nessler S, Hemmer B et al (2010) Pharmacological prion protein silencing accelerates central nervous system autoimmune disease via T cell receptor signalling. Brain 133:375–388. https://doi.org/10.1093/brain/awp298
Gregoire S, Logre C, Metharom P et al (2004) Identification of two immunogenic domains of the prion protein-PrP-which activate class II-restricted T cells and elicit antibody responses against the native molecule. J Leukoc Biol 76:125–134. https://doi.org/10.1189/jlb.1203656
Benestad SL, Austbø L, Tranulis MA et al (2012) Healthy goats naturally devoid of prion protein. Vet Res 43:87. https://doi.org/10.1186/1297-9716-43-87
Salvesen Ø, Espenes A, Reiten MR et al (2020) Goats naturally devoid of PrPC are resistant to scrapie. Vet Res 51:1. https://doi.org/10.1186/s13567-019-0731-2
Salvesen RMR, Espenes A et al (2017) LPS-induced systemic inflammation reveals an immunomodulatory role for the prion protein at the blood-brain interface. J Neuroinflammation 14:1–12. https://doi.org/10.1186/s12974-017-0879-5
Malachin G, Reiten MR, Salvesen Ø et al (2017) Loss of prion protein induces a primed state of type I interferon-responsive genes. PLoS ONE 12:1–16. https://doi.org/10.1371/journal.pone.0179881
de Almeida CJG, Chiarini LB, da Silva JP et al (2005) The cellular prion protein modulates phagocytosis and inflammatory response. J Leukoc Biol 77:238–246. https://doi.org/10.1189/jlb.1103531
Wang M, Zhao D, Yang Y et al (2014) The cellular prion protein negatively regulates phagocytosis and cytokine expression in murine bone marrow-derived macrophages. PLoS ONE 9:e102785. https://doi.org/10.1371/journal.pone.0102785
Onodera, (2010) Enhancement of phagocytotic activity by prion protein in PrP-deficient macrophage cells. Int J Mol Med 26:527–532. https://doi.org/10.3892/ijmm_00000495
Ding T, Zhou X, Kouadir M et al (2013) Cellular prion protein participates in the regulation of inflammatory response and apoptosis in BV2 microglia during infection with Mycobacterium bovis. J Mol Neurosci 51:118–126. https://doi.org/10.1007/s12031-013-9962-2
Shi F, Yang L, Kouadir M et al (2013) Prion protein participates in the regulation of classical and alternative activation of BV2 microglia. J Neurochem 124:168–174. https://doi.org/10.1111/jnc.12053
Pinheiro LP, Linden R, Mariante RM (2015) Activation and function of murine primary microglia in the absence of the prion protein. J Neuroimmunol 286:25–32. https://doi.org/10.1016/j.jneuroim.2015.07.002
Tsutsui S, Hahn JN, Johnson TA et al (2008) Absence of the cellular prion protein exacerbates and prolongs neuroinflammation in experimental autoimmune encephalomyelitis. Am J Pathol 173:1029–1041. https://doi.org/10.2353/ajpath.2008.071062
Gourdain P, Ballerini C, Nicot AB, Carnaud C (2012) Exacerbation of experimental autoimmune encephalomyelitis in prion protein (PrPc)-null mice: evidence for a critical role of the central nervous system. J Neuroinflammation 9:25. https://doi.org/10.1186/1742-2094-9-25
Aucouturier P, Carp RI, Carnaud C, Wisniewski T (2000) Prion diseases and the immune system. Clin Immunol 96:79–85
Mabbott NA, Alibhai JD, Manson J (2018) The role of the immune system in prion infection. In: Handbook of Clinical Neurology. Elsevier B.V., pp 85–107
Mabbott NA, Bradford BM, Pal R et al (2020) The effects of immune system modulation on prion disease susceptibility and pathogenesis. Int J Mol Sci 21:1–39
Peggion C, Stella R, Lorenzon P et al (2020) Microglia in prion diseases: angels or demons? Int J Mol Sci 21:1–16
Eklund CM, Kennedy RC, Hadlow WJ (1967) Pathogenesis of scrapie virus infection in the mouse. J Infect Dis 117:15–22. https://doi.org/10.1093/infdis/117.1.15
Keulen LJM, Schreuder BEC, Vromans MEW et al (2000) Pathogenesis of natural scrapie in sheep. Prion Diseases. Springer Vienna, Vienna, pp 57–71
Sigurdson CJ, Williams ES, Miller MW et al (1999) Oral transmission and early lymphoid tropism of chronic wasting disease PrPres in mule deer fawns (Odocoileus hemionus ). J Gen Virol 80:2757–2764. https://doi.org/10.1099/0022-1317-80-10-2757
Dickinson AG, Meikle VMH, Fraser H (1969) Genetical control of the concentration of ME7 scrapie agent in the brain of mice. J Comp Pathol 79:15–22. https://doi.org/10.1016/0021-9975(69)90021-8
Fraser H, Dickinson AG (1970) Pathogenesis of scrapie in the mouse: the role of the spleen. Nature 226:462–463. https://doi.org/10.1038/226462a0
Béringue V, Tixador P, Andréoletti O et al (2020) Host prion protein expression levels impact prion tropism for the spleen. PLOS Pathog 16:e1008283. https://doi.org/10.1371/journal.ppat.1008283
McCulloch L, Brown KL, Bradford BM et al (2011) Follicular dendritic cell-specific prion protein (PrPc) expression alone is sufficient to sustain prion infection in the spleen. PLoS Pathog 7:e1002402. https://doi.org/10.1371/journal.ppat.1002402
Brandner S, Klein MA, Aguzzi A (1999) A crucial role for B cells in neuroinvasive scrapie. Transfus Clin Biol 6:17–23. https://doi.org/10.1016/S1246-7820(99)80007-X
Kujala P, Raymond CR, Romeijn M et al (2011) Prion uptake in the gut: identification of the first uptake and replication sites. PLoS Pathog 7:e1002449. https://doi.org/10.1371/journal.ppat.1002449
Giese A, Brown DR, Groschup MH et al (1998) Role of microglia in neuronal cell death in prion disease. Brain Pathol 8:449–457. https://doi.org/10.1111/j.1750-3639.1998.tb00167.x
Muhleisen H, Gehrmann J, Meyermann R (1995) Reactive microgIia in Creutzfeldt-Jakob disease. Neuropathol Appl Neurobiol 21:505–517. https://doi.org/10.1111/j.1365-2990.1995.tb01097.x
Sasaki A, Hirato J, Nakazato Y (1993) Immunohistochemical study of microglia in the Creutzfeldt-Jakob diseased brain. Acta Neuropathol 86:337–344. https://doi.org/10.1007/BF00369445
Vidal E, Márquez M, Tortosa R et al (2006) Immunohistochemical approach to the pathogenesis of bovine spongiform encephalopathy in its early stages. J Virol Methods 134:15–29. https://doi.org/10.1016/j.jviromet.2005.11.010
Miyazono M, Kitamoto T, Iwaki T, Tateishi J (1992) Colocalization of prion protein and β protein in the same amyloid plaques in patients with Gerstmann-Sträussler Syndrome. Acta Neuropathol 83:333–339. https://doi.org/10.1007/BF00713522
Vincenti JE, Murphy L, Grabert K et al (2016) Defining the microglia response during the time course of chronic neurodegeneration. J Virol 90:3003–3017. https://doi.org/10.1128/jvi.02613-15
Gomez-Nicola D, Fransen NL, Suzzi S, Perry VH (2013) Regulation of microglial proliferation during chronic neurodegeneration. J Neurosci 33:2481–2493. https://doi.org/10.1523/JNEUROSCI.4440-12.2013
Williams AE, Lawson LJ, Perry VH, Fraser H (1994) Characterization of the microglial response in murine scrapie. Neuropathol Appl Neurobiol 20:47–55. https://doi.org/10.1111/j.1365-2990.1994.tb00956.x
Hay B, Prusiner SB, Lingappa VR (1987) Evidence for a secretory form of the cellular prion protein. Biochemistry 26:8110–8115. https://doi.org/10.1021/bi00399a014
Gatti JL, Métayer S, Moudjou M et al (2002) Prion protein is secreted in soluble forms in the epididymal fluid and proteolytically processed and transported in seminal plasma. Biol Reprod 67:393–400. https://doi.org/10.1095/biolreprod67.2.393
Robertson C, Booth SA, Beniac DR et al (2006) Cellular prion protein is released on exosomes from activated platelets. Blood 107:3907–3911. https://doi.org/10.1182/blood-2005-02-0802
Bignon Y, Poindessous V, Lazareth H et al (2020) The cellular prion protein is a stress protein secreted by renal tubular cells and a urinary marker of kidney injury. Cell Death Dis 11:243. https://doi.org/10.1038/s41419-020-2430-3
Vella L, Sharples R, Lawson V et al (2007) Packaging of prions into exosomes is associated with a novel pathway of PrP processing. J Pathol 211:582–590. https://doi.org/10.1002/path.2145
Altmeppen HC, Prox J, Puig B et al (2011) Lack of a-disintegrin-and-metalloproteinase ADAM10 leads to intracellular accumulation and loss of shedding of the cellular prion protein in vivo. Mol Neurodegener 6:36. https://doi.org/10.1186/1750-1326-6-36
Parkin ET, Watt NT, Turner AJ, Hooper NM (2004) Dual mechanisms for shedding of the cellular prion protein. J Biol Chem 279:11170–11178. https://doi.org/10.1074/jbc.M312105200
Yáñez-Mó M, Siljander PRM, Andreu Z et al (2015) Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles 4:27066. https://doi.org/10.3402/jev.v4.27066
Krämer-Albers E-M, Hill AF (2016) Extracellular vesicles: interneural shuttles of complex messages. Curr Opin Neurobiol 39:101–107. https://doi.org/10.1016/j.conb.2016.04.016
György B, Szabó TG, Pásztói M et al (2011) Membrane vesicles, current state-of-the-art: emerging role of extracellular vesicles. Cell Mol Life Sci 68:2667–2688. https://doi.org/10.1007/s00018-011-0689-3
Quek C, Hill AF (2017) The role of extracellular vesicles in neurodegenerative diseases. Biochem Biophys Res Commun 483:1178–1186. https://doi.org/10.1016/j.bbrc.2016.09.090
Fevrier B, Vilette D, Archer F et al (2004) Cells release prions in association with exosomes. Proc Natl Acad Sci 101:9683–9688. https://doi.org/10.1073/pnas.0308413101
Leng B, Sun H, Zhao J et al (2020) Plasma exosomal prion protein levels are correlated with cognitive decline in PD patients. Neurosci Lett 723:134866. https://doi.org/10.1016/j.neulet.2020.134866
Dias MVS, Teixeira BL, Rodrigues BR et al (2016) PRNP/prion protein regulates the secretion of exosomes modulating CAV1/caveolin-1-suppressed autophagy. Autophagy 12:2113–2128. https://doi.org/10.1080/15548627.2016.1226735
Abdulrahman BA, Abdelaziz DH, Schatzl HM (2018) Autophagy regulates exosomal release of prions in neuronal cells. J Biol Chem 293:8956–8968. https://doi.org/10.1074/jbc.RA117.000713
Barbieri G, Palumbo S, Gabrusiewicz K et al (2011) Silencing of cellular prion protein (PrPc) expression by DNA-antisense oligonucleotides induces autophagy-dependent cell death in glioma cells. Autophagy 7:840–853. https://doi.org/10.4161/auto.7.8.15615
Ryskalin L, Busceti CL, Biagioni F et al (2019) Prion protein in glioblastoma multiforme. Int J Mol Sci 20:1–15
Provenzano L, Ryan Y, Hilton DA et al (2017) Cellular prion protein (PrP C) in the development of Merlin-deficient tumours. Oncogene 36:6132–6142. https://doi.org/10.1038/onc.2017.200
Corsaro A, Bajetto A, Thellung S et al (2016) Cellular prion protein controls stem cell-like properties of human glioblastoma tumor-initiating cells. Oncotarget 7:38638–38657. https://doi.org/10.18632/oncotarget.9575
Iglesia RP, Prado MB, Cruz L et al (2017) Engagement of cellular prion protein with the co-chaperone Hsp70/90 organizing protein regulates the proliferation of glioblastoma stem-like cells. Stem Cell Res Ther 8:76. https://doi.org/10.1186/s13287-017-0518-1
Lee JH, Yun CW, Han YS et al (2018) Melatonin and 5-fluorouracil co-suppress colon cancer stem cells by regulating cellular prion protein-Oct4 axis. J Pineal Res 65:e12519. https://doi.org/10.1111/jpi.12519
Du L, Rao G, Wang H et al (2013) CD44-positive cancer stem cells expressing cellular prion protein contribute to metastatic capacity in colorectal cancer. Cancer Res 73:2682–2694. https://doi.org/10.1158/0008-5472.CAN-12-3759
Lin SC, Lin CH, Shih NC et al (2020) Cellular prion protein transcriptionally regulated by NFIL3 enhances lung cancer cell lamellipodium formation and migration through JNK signaling. Oncogene 39:385–398. https://doi.org/10.1038/s41388-019-0994-0
Keniry M, Pires MM, Mense S et al (2013) Survival factor NFIL3 restricts FOXO-induced gene expression in cancer. Genes Dev 27:916–927. https://doi.org/10.1101/gad.214049.113
Le Corre D, Ghazi A, Balogoun R et al (2019) The cellular prion protein controls the mesenchymal-like molecular subtype and predicts disease outcome in colorectal cancer. EBioMedicine 46:94–104. https://doi.org/10.1016/j.ebiom.2019.07.036
de Lacerda TCS, Costa-Silva B, Giudice FS et al (2016) Prion protein binding to HOP modulates the migration and invasion of colorectal cancer cells. Clin Exp Metastasis 33:441–451. https://doi.org/10.1007/s10585-016-9788-8
Ke J, Wu G, Zhang J et al (2020) Melanoma migration is promoted by prion protein via Akt-hsp27 signaling axis. Biochem Biophys Res Commun 523:375–381. https://doi.org/10.1016/j.bbrc.2019.12.042
Guo K, Kang NX, Li Y et al (2009) Regulation of HSP27 on NF-κB pathway activation may be involved in metastatic hepatocellular carcinoma cells apoptosis. BMC Cancer. https://doi.org/10.1186/1471-2407-9-100
Wang JH, Du JP, Li SJ et al (2012) Octarepeat peptides of prion are essential for multidrug resistance in gastric cancer cells. J Dig Dis 13:143–152. https://doi.org/10.1111/j.1751-2980.2011.00563.x
Wang Q, Qian J, Wang F, Ma Z (2012) Cellular prion protein accelerates colorectal cancer metastasis via the Fyn-SP1-SATB1 axis. In: Oncol Rep 2029–2034
Wang W, Wen Q, Luo J et al (2017) Suppression of β-catenin nuclear translocation by CGP57380 decelerates poor progression and potentiates radiation-induced apoptosis in nasopharyngeal carcinoma. Theranostics 7:2134–2149. https://doi.org/10.7150/THNO.17665
Han HJ, Russo J, Kohwi Y, Kohwi-Shigematsu T (2008) SATB1 reprogrammes gene expression to promote breast tumour growth and metastasis. Nature 452:187–193. https://doi.org/10.1038/nature06781
Shukla S, Sharma H, Abbas A et al (2013) Upregulation of SATB1 is associated with prostate cancer aggressiveness and disease progression. PLoS ONE. https://doi.org/10.1371/journal.pone.0053527
Yun C-W, Lee J-H, Go G et al (2021) Prion protein of extracellular vesicle regulates the progression of colorectal cancer. Cancers (Basel) 13:2144. https://doi.org/10.3390/cancers13092144
Liang J, Bai F, Luo G et al (2007) Hypoxia induced overexpression of PrPC in gastric cancer cell lines. Cancer Biol Ther 6:769–774. https://doi.org/10.4161/cbt.6.5.4001
Robey IF, Stephen RM, Brown KS et al (2008) Regulation of the Warburg effect in early-passage breast cancer cells. Neoplasia 10:745–756. https://doi.org/10.1593/neo.07724
Sun W, Liu Y, Glazer CA et al (2010) TKTL1 is activated by promoter hypomethylation and contributes to head and neck squamous cell carcinoma carcinogenesis through increased aerobic glycolysis and HIF1 stabilization. Clin Cancer Res 16:857–866. https://doi.org/10.1158/1078-0432.CCR-09-2604
Li Q-Q, Sun Y-P, Ruan C-P et al (2011) Cellular prion protein promotes glucose uptake through the Fyn-HIF-2α-Glut1 pathway to support colorectal cancer cell survival. Cancer Sci 102:400–406. https://doi.org/10.1111/j.1349-7006.2010.01811.x
Zhuang D, Liu Y, Mao Y et al (2012) TMZ-induced PrPc/par-4 interaction promotes the survival of human glioma cells. Int J Cancer 130:309–318. https://doi.org/10.1002/ijc.25985
Meslin F, Conforti R, Mazouni C et al (2007) Efficacy of adjuvant chemotherapy according to prion protein expression in patients with estrogen receptor-negative breast cancer. Ann Oncol 18:1793–1798. https://doi.org/10.1093/annonc/mdm406
Cheng Y, Tao L, Xu J et al (2014) CD44/Cellular prion protein interact in multidrug resistant breast cancer cells and correlate with responses to neoadjuvant chemotherapy in breast cancer patients. Mol Carcinog 53:686–697. https://doi.org/10.1002/mc.22021
Wiegmans AP, Saunus JM, Ham S et al (2019) Secreted cellular prion protein binds doxorubicin and correlates with anthracycline resistance in breast cancer. JCI Insight 4:e124092. https://doi.org/10.1172/jci.insight.124092
Liang J, Ge F, Guo C et al (2009) Inhibition of PI3K/Akt partially leads to the inhibition of PrPC-induced drug resistance in gastric cancer cells. FEBS J 276:685–694. https://doi.org/10.1111/j.1742-4658.2008.06816.x
Du J, Pan Y, Shi Y et al (2005) Overexpression and significance of prion protein in gastric cancer and multidrug-resistant gastric carcinoma cell line SGC7901/ADR. Int J Cancer 113:213–220. https://doi.org/10.1002/ijc.20570
Sakudo A, Lee D, Nishimura T et al (2005) Octapeptide repeat region and N-terminal half of hydrophobic region of prion protein (PrP) mediate PrP-dependent activation of superoxide dismutase. Biochem Biophys Res Commun 326:600–606. https://doi.org/10.1016/j.bbrc.2004.11.092
Ju H-Q, Gocho T, Aguilar M et al (2015) Mechanisms of overcoming intrinsic resistance to gemcitabine in pancreatic ductal adenocarcinoma through the redox modulation. Mol Cancer Ther 14:788–798. https://doi.org/10.1158/1535-7163.MCT-14-0420
Luo G, Wang W, Wu Q et al (2017) MGr1-Antigen/37 kDa laminin receptor precursor promotes cellular prion protein induced multi-drug-resistance of gastric cancer. Oncotarget 8:71630–71641. https://doi.org/10.18632/oncotarget.17795
Bernardino-Sgherri J, Siberchicot C, Auvré F et al (2021) Tumor resistance to radiotherapy is triggered by an ATM/TAK1-dependent-increased expression of the cellular prion protein. Oncogene 40:3460–3469. https://doi.org/10.1038/s41388-021-01746-0
Cha S, Sin MJ, Kim MJ et al (2021) Involvement of cellular prion protein in invasion and metastasis of lung cancer by inducing treg cell development. Biomolecules 11:1–12. https://doi.org/10.3390/biom11020285
Tang Z, Ma J, Zhang W et al (2016) The role of prion protein expression in predicting gastric cancer prognosis. J Cancer 7:984–990. https://doi.org/10.7150/jca.14237
McDonald R (1989) Modern biological theories of aging. J Nutr 119:952–953. https://doi.org/10.1093/jn/119.6.952
Goh AX-H, Li C, Sy M-S, Wong B-S (2007) Altered prion protein glycosylation in the aging mouse brain. J Neurochem 100:841–854. https://doi.org/10.1111/j.1471-4159.2006.04268.x
Williams WM, Stadtman ER, Moskovitz J (2004) Ageing and exposure to oxidative stress in vivo differentially affect cellular levels of PrPc in mouse cerebral microvessels and brain parenchyma. Neuropathol Appl Neurobiol 30:161–168. https://doi.org/10.1111/j.1365-2990.2003.00523.x
Brown DR, Clive C, Haswell SJ (2001) Antioxidant activity related to copper binding of native prion protein. J Neurochem 76:69–76. https://doi.org/10.1046/j.1471-4159.2001.00009.x
Brown DR, Wong B-S, Hafiz F et al (1999) Normal prion protein has an activity like that of superoxide dismutase. Biochem J 344:1. https://doi.org/10.1042/0264-6021:3440001
Gasperini L, Meneghetti E, Legname G, Benetti F (2016) In absence of the cellular prion protein, alterations in copper metabolism and copper-dependent oxidase activity affect iron distribution. Front Neurosci 10:437. https://doi.org/10.3389/fnins.2016.00437
Benvegnù S, Gasperini L, Legname G (2011) Aged PrP null mice show defective processing of neuregulins in the peripheral nervous system. Mol Cell Neurosci 47:28–35. https://doi.org/10.1016/j.mcn.2011.02.005
Küffer A, Lakkaraju AKK, Mogha A et al (2016) The prion protein is an agonistic ligand of the G protein-coupled receptor Adgrg6. Nature 536:464–468. https://doi.org/10.1038/nature19312
Feigin VL, Stark BA, Johnson CO et al (2021) Global, regional, and national burden of stroke and its risk factors, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol 20:795–820. https://doi.org/10.1016/S1474-4422(21)00252-0
McLennan NF, Brennan PM, McNeill A et al (2004) Prion protein accumulation and neuroprotection in hypoxic brain damage. Am J Pathol 165:227–235. https://doi.org/10.1016/S0002-9440(10)63291-9
Doeppner TR, Kaltwasser B, Schlechter J et al (2015) Cellular prion protein promotes post-ischemic neuronal survival, angioneurogenesis and enhances neural progenitor cell homing via proteasome inhibition. Cell Death Dis 6:e2024–e2024. https://doi.org/10.1038/cddis.2015.365
Weise J, Doeppner TR, Müller T et al (2008) Overexpression of cellular prion protein alters postischemic Erk1/2 phosphorylation but not Akt phosphorylation and protects against focal cerebral ischemia. Restor Neurol Neurosci 26:57–64
Jeong J-K, Seo J-S, Moon M-H et al (2012) Hypoxia-inducible factor-1 alpha regulates prion protein expression to protect against neuron cell damage. Neurobiol Aging 33:1006.e1-1006.e10. https://doi.org/10.1016/j.neurobiolaging.2011.09.037
Schmitz M, Greis C, Ottis P et al (2014) Loss of prion protein leads to age-dependent behavioral abnormalities and changes in cytoskeletal protein expression. Mol Neurobiol 50:923–936
Rial D, Duarte FS, Xikota JC et al (2009) Cellular prion protein modulates age-related behavioral and neurochemical alterations in mice. Neuroscience 164:896–907. https://doi.org/10.1016/j.neuroscience.2009.09.005
Leighton PLA, Nadolski NJ, Morrill A et al (2018) An ancient conserved role for prion protein in learning and memory. Biol Open 7:bio025734. https://doi.org/10.1242/bio.025734
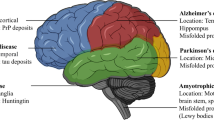
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259. https://doi.org/10.1007/BF00308809
Di Lazzaro V (2018) Emergence of neurophysiological biomarkers of Alzheimer disease. J Neurol Neurosurg Psychiatry 89:1235–1235. https://doi.org/10.1136/jnnp-2018-318867
Benilova I, Karran E, De Strooper B (2012) The toxic Aβ oligomer and Alzheimer’s disease: An emperor in need of clothes. Nat Neurosci 15:349–357
Kayed R, Lasagna-Reeves CA (2012) Molecular mechanisms of amyloid oligomers toxicity. J Alzheimer’s Dis 33:S67–S78. https://doi.org/10.3233/JAD-2012-129001
Haas LT, Kostylev MA, Strittmatter SM (2014) Therapeutic molecules and endogenous ligands regulate the interaction between brain cellular prion protein (PrPC) and metabotropic glutamate receptor 5 (mGluR5). J Biol Chem 289:28460–28477. https://doi.org/10.1074/jbc.M114.584342
Um JW, Nygaard HB, Heiss JK et al (2012) Alzheimer amyloid-Î 2 oligomer bound to postsynaptic prion protein activates Fyn to impair neurons. Nat Neurosci 15:1227–1235. https://doi.org/10.1038/nn.3178
Younan ND, Sarell CJ, Davies P et al (2013) The cellular prion protein traps Alzheimer’s Aβ in an oligomeric form and disassembles amyloid fibers. FASEB J 27:1847–1858. https://doi.org/10.1096/fj.12-222588
Pagano K, Galante D, D’Arrigo C et al (2019) Effects of prion protein on Aβ42 and pyroglutamate-modified AβpΕ3-42 oligomerization and toxicity. Mol Neurobiol 56:1957–1971. https://doi.org/10.1007/s12035-018-1202-x
Ulbrich S, Janning P, Seidel R et al (2018) Alterations in the brain interactome of the intrinsically disordered N-terminal domain of the cellular prion protein (PrPC) in Alzheimer’s disease. PLoS ONE 13:1–15. https://doi.org/10.1371/journal.pone.0197659
Laurén J, Gimbel DA, Nygaard HB et al (2009) Cellular prion protein mediates impairment of synaptic plasticity by amyloid-Β oligomers. Nature 457:1128–1132. https://doi.org/10.1038/nature07761
Madhu P, Mukhopadhyay S (2020) Preferential recruitment of conformationally distinct amyloid-β oligomers by the intrinsically disordered region of the human prion protein. ACS Chem Neurosci 11:86–98. https://doi.org/10.1021/acschemneuro.9b00646
Harris DA, Huber MT, van Dijken P et al (1993) Processing of a cellular prion protein: identification of N- and C-terminal cleavage sites. Biochemistry 32:1009–1016. https://doi.org/10.1021/bi00055a003
Scott-McKean JJ, Surewicz K, Choi JK et al (2016) Soluble prion protein and its N-terminal fragment prevent impairment of synaptic plasticity by Aβ oligomers: implications for novel therapeutic strategy in Alzheimer’s disease. Neurobiol Dis 91:124–131. https://doi.org/10.1016/j.nbd.2016.03.001
Balducci C, Beeg M, Stravalaci M et al (2010) Synthetic amyloid-β oligomers impair long-term memory independently of cellular prion protein. Proc Natl Acad Sci 107:2295–2300. https://doi.org/10.1073/pnas.0911829107
Rial D, Piermartiri TC, Duarte FS et al (2012) Overexpression of cellular prion protein (PrPC) prevents cognitive dysfunction and apoptotic neuronal cell death induced by amyloid-β (Aβ1-40) administration in mice. Neuroscience 215:79–89. https://doi.org/10.1016/j.neuroscience.2012.04.034
Gunther EC, Smith LM, Kostylev MA et al (2019) Rescue of transgenic Alzheimer’s pathophysiology by polymeric cellular prion protein antagonists. Cell Rep 26:145-158.e8. https://doi.org/10.1016/j.celrep.2018.12.021
Chung E, Ji Y, Sun Y et al (2010) Anti-PrPCmonoclonal antibody infusion as a novel treatment for cognitive deficits in an Alzheimer’s disease model mouse. BMC Neurosci 11:130. https://doi.org/10.1186/1471-2202-11-130
Salazar SV, Gallardo C, Kaufman AC et al (2017) Conditional deletion of Prnp rescues behavioral and synaptic deficits after disease onset in transgenic Alzheimer’s disease. J Neurosci 37:9207–9221. https://doi.org/10.1523/JNEUROSCI.0722-17.2017
Whitehouse IJ, Jackson C, Turner AJ, Hooper NM (2010) Prion protein is reduced in aging and in sporadic but not in familial Alzheimer’s disease. J Alzheimer’s Dis 22:1023–1031. https://doi.org/10.3233/JAD-2010-101071
Llorens F, Ansoleaga B, Garcia-Esparcia P et al (2013) PrP mRNa and protein expression in brain and PrPc in CSF in Creutzfeldt-Jakob disease MM1 and VV2. Prion 7:383–393. https://doi.org/10.4161/pri.26416
Kessels HW, Nguyen LN, Nabavi S, Malinow R (2010) The prion protein as a receptor for amyloid-β. Nature 466:E3–E4. https://doi.org/10.1038/nature09217
Calella AM, Farinelli M, Nuvolone M et al (2010) Prion protein and Aβ-related synaptic toxicity impairment. EMBO Mol Med 2:306–314. https://doi.org/10.1002/emmm.201000082
Avila J, Lucas JJ, Pérez M, Hernández F (2004) Role of tau protein in both physiological and pathological conditions. Physiol Rev 84:361–384
Ondrejcak T, Klyubin I, Corbett GT et al (2018) Cellular prion protein mediates the disruption of hippocampal synaptic plasticity by soluble tau in vivo. J Neurosci 38:10595–10606. https://doi.org/10.1523/JNEUROSCI.1700-18.2018
Gomes LA, Hipp SA, Rijal Upadhaya A et al (2019) Aβ-induced acceleration of Alzheimer-related τ-pathology spreading and its association with prion protein. Acta Neuropathol 138:913–941. https://doi.org/10.1007/s00401-019-02053-5
Larson M, Sherman MA, Amar F et al (2012) The complex PrPc-Fyn couples human oligomeric Aβ with pathological tau changes in Alzheimer’s disease. J Neurosci 32:16857–16871. https://doi.org/10.1523/JNEUROSCI.1858-12.2012
Vergara C, Ordóñez-Gutiérrez L, Wandosell F et al (2015) Role of PrPC expression in tau protein levels and phosphorylation in Alzheimer’s disease evolution. Mol Neurobiol 51:1206–1220. https://doi.org/10.1007/s12035-014-8793-7
De Cecco E, Celauro L, Vanni S et al (2020) The uptake of tau amyloid fibrils is facilitated by the cellular prion protein and hampers prion propagation in cultured cells. J Neurochem 155:577–591. https://doi.org/10.1111/jnc.15040
Pickett EK, Herrmann AG, McQueen J et al (2019) Amyloid beta and tau cooperate to cause reversible behavioral and transcriptional deficits in a model of Alzheimer’s disease. Cell Rep 29:3592-3604.e5. https://doi.org/10.1016/j.celrep.2019.11.044
Lidón L, Vergara C, Ferrer I et al (2020) Tau protein as a new regulator of cellular prion protein transcription. Mol Neurobiol 57:4170–4186. https://doi.org/10.1007/s12035-020-02025-x
Schmitz M, Wulf K, Signore SC et al (2013) Impact of the cellular prion protein on amyloid-β and 3PO-tau processing. J Alzheimer’s Dis 38:551–565. https://doi.org/10.3233/JAD-130566
de Lau LM, Breteler MM (2006) Epidemiology of Parkinson’s disease. Lancet Neurol 5:525–535. https://doi.org/10.1016/S1474-4422(06)70471-9
Hoehn MM, Yahr MD (1967) Parkinsonism: onset, progression, and mortality. Neurology 17:427–427. https://doi.org/10.1212/WNL.17.5.427
Chaudhuri KR, Healy DG, Schapira AH (2006) Non-motor symptoms of Parkinson’s disease: diagnosis and management. Lancet Neurol 5:235–245. https://doi.org/10.1016/S1474-4422(06)70373-8
Rietdijk CD, Perez-Pardo P, Garssen J et al (2017) Exploring Braak’s hypothesis of Parkinson’s disease. Front Neurol 8:37. https://doi.org/10.3389/fneur.2017.00037
Sarnataro D (2018) Attempt to untangle the prion-like misfolding mechanism for neurodegenerative diseases. Int J Mol Sci 19:3081. https://doi.org/10.3390/ijms19103081
Li J-Y, Englund E, Holton JL et al (2008) Lewy bodies in grafted neurons in subjects with Parkinson’s disease suggest host-to-graft disease propagation. Nat Med 14:501–503. https://doi.org/10.1038/nm1746
Ugalde CL, Finkelstein DI, Lawson VA, Hill AF (2016) Pathogenic mechanisms of prion protein, amyloid-β and α-synuclein misfolding: the prion concept and neurotoxicity of protein oligomers. J Neurochem 139:162–180. https://doi.org/10.1111/jnc.13772
Aulić S, Masperone L, Narkiewicz J et al (2017) α-Synuclein amyloids hijack prion protein to gain cell entry, facilitate cell-to-cell spreading and block prion replication. Sci Rep 7:10050. https://doi.org/10.1038/s41598-017-10236-x
Ferreira DG, Temido-Ferreira M, Vicente Miranda H et al (2017) α-synuclein interacts with PrPC to induce cognitive impairment through mGluR5 and NMDAR2B. Nat Neurosci 20:1569–1579. https://doi.org/10.1038/nn.4648
Thom T, Schmitz M, Fischer AL et al (2022) Cellular prion protein mediates α-synuclein uptake, localization, and toxicity in vitro and in vivo. Mov Disord 37:39–51. https://doi.org/10.1002/mds.28774
La Vitola P, Beeg M, Balducci C et al (2019) Cellular prion protein neither binds to alpha-synuclein oligomers nor mediates their detrimental effects. Brain 142:249–254. https://doi.org/10.1093/brain/awy318
Corbett GT, Wang Z, Hong W et al (2020) PrP is a central player in toxicity mediated by soluble aggregates of neurodegeneration-causing proteins. Acta Neuropathol 139:503–526. https://doi.org/10.1007/s00401-019-02114-9
Nicoll AJ, Panico S, Freir DB et al (2013) Amyloid-β nanotubes are associated with prion protein-dependent synaptotoxicity. Nat Commun 4:2416. https://doi.org/10.1038/ncomms3416
Shirasaka M, Kuwata K, Honda R (2020) α-Synuclein chaperone suppresses nucleation and amyloidogenesis of prion protein. Biochem Biophys Res Commun 521:259–264. https://doi.org/10.1016/j.bbrc.2019.10.120
De Cecco E, Legname G (2018) The role of the prion protein in the internalization of α-synuclein amyloids. Prion 12:23–27. https://doi.org/10.1080/19336896.2017.1423186
Rösener NS, Gremer L, Wördehoff MM et al (2020) Clustering of human prion protein and α-synuclein oligomers requires the prion protein N-terminus. Commun Biol 3:1–12. https://doi.org/10.1038/s42003-020-1085-z
Acknowledgements
We thank Sidney Pratt, Canadian, MAT (The Johns Hopkins University), RSAdip—TESL (Cambridge University) for carefully reviewing the English text of this paper.
Funding
This work was supported by Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ; grant number: E-26/200–813/2021); Instituto Nacional de Neurociência Translacional (INNT; grant number: 465346/2014–6); Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; grant number: 310350/2020–4), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES; grant number: 88881.370888/2019–01), and PhD-Program on Morphological Sciences from Universidade Federal do Rio de Janeiro (UFRJ).
Author information
Authors and Affiliations
Contributions
Conceptualization: Izabella Grimaldi and Flavia Lima. Literature search: Izabella Grimaldi, Ana Clara Campanelli, Felipe Saceanu Leser, José Marcos Janeiro, and Bárbara Gomes da Rosa. Writing—original draft preparation: Izabella Grimaldi, Ana Clara Campanelli, José Marcos Janeiro, Felipe Saceanu Leser, and Bárbara Gomes da Rosa. Writing—review and editing: Izabella Grimaldi, Felipe Saceanu Leser, Luciana Romão,and Flavia Lima. Supervision: Flavia Lima and Luciana Romão. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Grimaldi, I., Leser, F.S., Janeiro, J.M. et al. The multiple functions of PrPC in physiological, cancer, and neurodegenerative contexts. J Mol Med 100, 1405–1425 (2022). https://doi.org/10.1007/s00109-022-02245-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-022-02245-9