Abstract
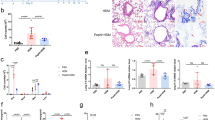
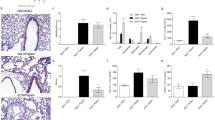
Lactobacilli prevent the early development of allergic diseases in children and experimental asthma in mice. However, the detailed mechanism underlying this action remains unknown. We aimed to explore the activation pathway in the host by Lactobacillus and identify its immunomodulation mechanism in allergic asthma. Continuous administration of 107 cfu, but not 109 cfu, of L. gasseri for 4 weeks prevented Dermatophagoides pteronyssinus (Der p)-induced airway hypersensitivity and inflammation in a mouse model of allergic asthma. DNA microarray analysis of the mesenteric and lung draining lymph nodes revealed a significant decrease in inflammatory chemokines and increase in gene expression in carbohydrate and lipid metabolism, particularly of PPARγ, in 107 cfu L. gasseri-administered mice compared with untreated mice. Compared with WT mice, Der p-sensitized PPARγL/+ mice showed increased airway hyperresponsiveness to methacholine, inflammatory cell infiltration, and inflammatory cytokine secretion in bronchoalveolar fluid. Moreover, the protective effects of L. gasseri were lost in Der p-induced airway inflammation in PPARγL/+ mice, and L. gasseri-induced PPARγ activation in BMDCs inhibited the development of allergic airway inflammation in both PPARγ WT and PPARγL/+ mice. L. gasseri may act via a novel PPARγ activation pathway in DCs to alleviate allergen-induced airway inflammation in allergic asthma.
Key messages
-
L. gasseri prevents mite allergen (Der p)-induced airway inflammation.
-
Prevention of airway inflammation occurs via activation of PPARγ in dendritic cells.
-
L. gasseri administration does not reverse Der p-induced airway inflammation in PPARγ(+/−) mice.
-
L. gasseri-induced PPARγ activation inhibits development of airway inflammation in WT and PPARγ(+/−) mice.





Similar content being viewed by others
References
Pawankar R (2014) Allergic diseases and asthma: a global public health concern and a call to action. World Allergy Organ J 7:12
Matinez FD, Vercelli D (2013) Asthma. Lancet 382:1360–1372
Jutel M, Agache I, Bonini S, Burks AW, Calderon M, Canonica W, Cox L, Demoly P, Frew AJ, O'Hehir R et al (2015) International consensus on allergy immunotherapy. J Allergy Clin Immunol 136:556–568
Integrative HMP (iHMP) Research Network Consortium (2014) The integrative human microbiome project: dynamic analysis of microbiome-host omics profiles during periods of human health and disease. Cell Host Microbe 16:276–289
Ding T, Schloss PD (2014) Dynamics and associations of microbial community types across the human body. Nature 509:357–360
Schaub B, Lauener R, von Mutius E (2006) The many faces of the hygiene hypothesis. J Allergy Clin Immunol 117:969–977
von Mutius E, Vercelli D (2010) Farm living: effects on childhood asthma and allergy. Nat Rev Immunol 10:861–868
Schaub B, Vercelli D (2015) Environmental protection from allergic diseases: from humans to mice and back. Cur Opin Immunol 36:88–93
Naik S, Bouladoux N, Linehan JL, Han SJ, Harrison OJ, Wilhelm C, Conlan S, Himmelfarb S, Byrd AL, Deming C et al (2015) Commensal-dendritic-cell interaction specifies a unique protective skin immune signature. Nature 520:104–108
Atarashi K, Tanoue T, Ando M, Kamada N, Nagano Y, Narushima S, Suda W, Imaoka A, Setoyama H, Nagamori T et al (2015) Th17 cell induction by adhesion of microbes to intestinal epithelial cells. Cell 163:367–380
Kalliomaki M, Salminen S, Poussa T, Arvilommi H, Isolauri E (2013) Probiotics and prevention of atopic disease: 4-year follow-up of a randomised placebo-controlled trial. Lancet 361:1869–1871
Jan RL, Yeh KC, Hsieh MH, Lin YL, Kao HF, Li PH, Chang YS, Wang JY (2012) Lactobacillus gasseri suppresses Th17 pro-inflammatory response and attenuates allergen-induced airway inflammation in a mouse model of allergic asthma. Br J Nutr 108:130–139
Trompette A, Gollwitzer ES, Yadava K, Sichelstiel AK, Sprenger N, Ngom-Bru C, Blanchard C, Junt T, Nicod LP, Harris NL et al (2014) Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat Med 20:159–166
Voltan S, Martines D, Elli M, Brun P, Longo S, Porzionato A, Macchi V, D'Incà R, Scarpa M, Palù G, Voltan S et al (2008) Lactobacillus crispatus M247-derived H2O2 acts as a signal transducing molecule activating peroxisome proliferator activated receptor-gamma in the intestinal mucosa. Gastroenterology 135:1216–1227
Chen YS, Lin YL, Jan RL, Chen HH, Wang JY (2010) Randomized placebo-controlled trial of lactobacillus on asthmatic children with allergic rhinitis. Pediatr Pulmol 45:1111–1120
Tsai YS, Kim HJ, Takahashi N, Kim HS, Hagaman JR, Kim JK, Maeda N (2004) Hypertension and abnormal fat distribution but not insulin resistance in mice with P465L PPARγ. J Clin Invest 114:240–249
Takeda K, Hamelmann E, Joetham A, Shultz LD, Larsen GL, Irvin CG (1997) Development of eosinophilic airway inflammation and airway hyper-responsiveness in mast cell-deficient mice. J Exp Med 186:449–454
Huang HJ, Lin YL, Liu CF, Kao HF, Wang JY (2011) Mite allergen decreases DC-SIGN expression and modulates human dendritic cell differentiation and function in allergic asthma. Mucosal Immunol 4:519–527
Ecker RC, Rogojanu R, Streit M, Oesterreicher K, Steiner GE (2006) An improved method for discrimination of cell populations in tissue sections using microscopy-based multicolor tissue cytometry. Cytometry A 69:119–123
Lin Y-L, Chen S-H, Wang J-Y (2016) Critical role of IL-6 in dendritic cell-induced allergic inflammation of asthma. J Mol Med 94:51–59
Dennis G Jr, Sherman BT, Hosack DA, Yang J, Gao W, Lane HC, Lempicki RA (2003) DAVID: database for annotation, visualization, and integrated discovery. Genome Biol 4:P3
Gour N, Wills-Karp M (2015) IL-4 and IL-13 signaling in allergic airway disease. Cytokine 75:68–78
Lu J, Liu L, Zhu Y, Zhang Y, Wu Y, Wang G, Zhang D, Xu J, Xie X, Ke R et al (2014) PPAR-γ inhibits IL-13-induced collagen production in mouse airway fibroblasts. Eur J Pharmacol 737:133–139
Liu SH, Yang CN, Pan HC, Sung YJ, Liao KK, Chen WB, Lin WZ, Sheu ML (2010) IL-13 downregulates PPAR-gamma/heme oxygenase-1 via ER stress-stimulated calpain activation: aggravation of activated microglia death. Cell Mol Life Sci 67:1465–1476
Sazawal S, Hiremath G, Dhingra U, Malik P, Deb S, Black RE (2006) Efficacy of probiotics in prevention of acute diarrhoea: a meta-analysis of masked, randomised, placebo-controlled trials. Lancet Infect Dis 6:37482
Helwig U, Lammers KM, Rizzello F, Brigidi P, Rohleder V, Caramelli E, Gionchetti P, Schrezenmeir J, Foelsch UR, Schreiber S et al (2006) Lactobacilli, bifidobacteria and E. Coli nissle induce pro- and anti-inflammatory cytokines in peripheral blood mononuclear cells. World J Gastroenterol 12:5978–5986
Zhang Y, Li L, Guo C, Mu D, Feng B, Zuo X, Li Y (2016) Effects of probiotic type, dose and treatment duration on irritable bowel syndrome diagnosed by Rome III criteria: a meta-analysis. BMC Gastroenterol 16:62
Minelli EB, Benini A (2008) Relationship between number of bacteria and their probiotic effects. Microb Ecol Health Dis 20:180–183
Kwon HK, Lee CG, So JS, Chae CS, Hwang JS, Sahoo A, Nam JH, Rhee JH, Hwang KC, Im SH (2010) Generation of regulatory dendritic cells and CD4+Foxp3+ T cells by probiotics administration suppresses immune disorders. Proc Natl Acad Sci U S A 107:2159–2164
Alexander KL, Targan SR, Elson CO (2014) Microbiota activation and regulation of innate and adaptive immunity. Immunol Rev 260:206–220
Hammad H, de Heer HJ, Soullié T, Angeli V, Trottein F, Hoogsteden HC, Lambrecht BN (2004) Activation of peroxisome proliferator-activated receptor-gamma in dendritic cells inhibits the development of eosinophilic airway inflammation in a mouse model of asthma. Am J Pathol 164:263–271
Thaiss CA, Zmora N, Levy M, Elinav E (2016) The microbiome and innate immunity. Nature 535:65–74
Abrahamsson TR, Jakobsson HE, Andersson AF, Bjorksten B, Engstrand L, Jenmalm MC (2012) Low diversity of the gut microbiota in infants with atopic eczema. J Allergy Clin Immunol 129:434–440
Bisgaard H, Li N, Bonnelykke K, Chawes BL, Skov T, Paludan-Müller G, Stokholm J, Smith B, Krogfelt KA (2011) Reduced diversity of the intestinal microbiota during infancy is associated with increased risk of allergic disease at school age. J Allergy Clin Immunol 128:646–652
Olszak T, An D, Zeissig S, Vera MP, Richter J, Franke A, Glickman JN, Siebert R, Baron RM, Kasper DL et al (2012) Microbial exposure during early life has persistent effects on natural killer T cell function. Science 336:489–493
Arnold IC, Dehzad N, Reuter S, Martin H, Becher B, Taube C, Müller A (2011) Helicobacter pylori infection prevents allergic asthma in mouse models through the induction of regulatory T cells. J Clin Invest 121:3088–3093
Zhang B, An J, Shimada T, Liu S, Maeyama K (2012) Oral administration of enterococcus faecalis FK-23 suppresses Th17 cell development and attenuates allergic airway responses in mice. Int J Mol Med 30:248–254
Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, deRoos P, Liu H, Cross JR, Pfeffer K, Coffer PJ et al (2013) Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 504:451–455
Smith PM, Howitt MR, Panikov N, Michaud M, Gallini CA, Bohlooly-Y M, Glickman JN, Garrett WS (2013) The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 341:569–573
Mazmanian SK, Liu CH, Tzianabos AO, Kasper DL (2005) An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122:107–118
Shen Y, Giardino-Torchia ML, Lawson GW, Karp CL, Ashwell JD, Mazmanian SK (2012) Outer membrane vesicles of a human commensal mediate immune regulation and disease protection. Cell Host Microbe 12:509–520
Calder PC (2008) Polyunsaturated fatty acids, inflammatory processes and inflammatory bowel diseases. Mol Nutr Food Res 52:885–897
Kelly D, Campbell JI, King TP, Grant G, Jansson EA, Coutts AG, Pettersson S, Conway S (2004) Commensal anaerobic gut bacteria attenuate inflammation by regulating nuclear cytoplasmic shuttling of PPAR-gamma and RelA. Nat Immunol 5:104–112
Bassaganya-Riera J, Viladomiu M, Pedragosa M, De Simone C, Carbo A, Shaykhutdinov R, Jobin C, Arthur JC, Corl BA, Vogel H et al (2012) Probiotic bacteria produce conjugated linoleic acid locally in the gut that targets macrophage pPARγ to suppress colitis. PLoS One 7:e31238
Benayoun L, Letuve S, Druilhe A, Boczkowski J, Dombret MC, Mechighel P, Megret J, Leseche G, Aubier M, Pretolani M (2001) Regulation of peroxisome proliferator-activated receptor gamma expression in human asthmatic airways: relationship with proliferation, apoptosis, and airway remodeling. Am J Respir Crit Care Med 164:1487–1494
Ogawa S, Lozach J, Benner C, Pascual G, Tangirala RK, Westin S, Hoffmann A, Subramaniam S, David M, Rosenfeld MG et al (2005) Molecular determinants of crosstalk between nuclear receptors and toll-like receptors. Cell 122:707–721
Kiss M, Czimmerer Z, Nagy L (2013) The role of lipid-activated nuclear receptors in shaping macrophage and dendritic cell function: from physiology to pathology. J Allergy Clin Immunol 132:264–286
Park SJ, Lee KS, Kim SR, Min KH, Choe YH, Moon H, Chae HJ, Yoo WH, Lee YC (2009) Peroxisome proliferator-activated receptor agonist down-regulates IL-17 expression in a murine model of allergic airway inflammation. J Immunol 183:3259–3267
Richards DB, Bareille P, Lindo EL, Quinn D, Farrow SN (2010) Treatment with a peroxisomal proliferator activated receptor gamma agonist has a modest effect in the allergen challenge model in asthma: a randomized controlled trial. Respir Med 104:668–674
Serhan CN, Devchand PR (2001) Novel anti-inflammatory target for asthma. A role for pPAR gamma? Am J Respir Cell Mol Biol 24:658–661
Woerly G, Honda K, Loyens M, Papin JP, Auwerx J, Staels B, Capron M, Dombrowicz D (2003) Peroxisome proliferator activated receptor alpha and gamma down regulate allergic inflammatory and eosinophil activation. J Exp Med 198:411–421
Selle K, Klaenhammer TR (2013) Genomic and phenotypic evidence for probiotic influences of lactobacillus gasseri on human health. FEMS Microbiol Rev 37:915–935
Kawase M, He F, Kubota A, Harata G, Hiramatsu M (2007) Orally administrated lactobacillus gasseri TMC0356 and lactobacillus GG alleviated nasal blockage of guinea pig with allergic rhinitis. Microbiol Immunol 51:1109–1114
Morita H, He F, Kawase M, Kubota A, Hiramatsu M, Kurisaki JI, Salminen S (2006) Preliminary human study for possible alteration of serum immunoglobulin E production in perennial allergic rhinitis with fermented milk prepared with lactobacillus gasseri TMC0356. Microbiol Immunol 50:701–706
Martınez-Canavate A, Sierra S, Lara-Villoslada F, Romero J, Maldonado J, Boza J, Xaus J, Olivares M (2009) A probiotic dairy product containing L. Gasseri CECT5714 and L. coryniformis CECT5711 induces immunological changes in children suffering from allergy. Pediatr Allergy Immunol 20:592–600
Acknowledgements
This work was supported by Ministry of Science and Technology (MOST) of Taiwan and research grants 99RC07, 101CB02, 102CB03, and 103CB01 from South Taiwan Science Park, MOST. The authors thank Dr. Yau-Sheng Tsai, Institute of Clinical Medicine, College of Medicine, National Chen Kung University, for providing PPARγ mice. We acknowledge Wallace Academic Editing for English proofreading.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The animal research in this study is according to the affidavit of approval of animal use protocol National Cheng Kung University, and the Institutional Animal Care and Use Committee (IACUC) approval No is 101096.
Competing interests
The authors declare that they have no competing interests.
Electronic supplementary material
ESM 1
(PDF 565 kb)
Rights and permissions
About this article
Cite this article
Hsieh, MH., Jan, RL., Wu, L.SH. et al. Lactobacillus gasseri attenuates allergic airway inflammation through PPARγ activation in dendritic cells. J Mol Med 96, 39–51 (2018). https://doi.org/10.1007/s00109-017-1598-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-017-1598-1