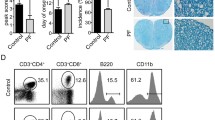
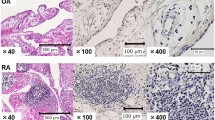
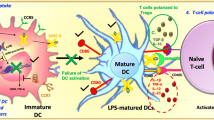
Abstract
ZSTK474 [2-(2-difluoromethylbenzimidazol-1-yl)-4,6-dimorpholino-1,3,5-triazine] is a novel phosphatidylinositol 3-kinase (PI3K) inhibitor that exhibits potent antitumor effects. Recent studies have shown that ZSTK474 is also with anti-inflammatory properties in collagen-induced arthritis. However, the effects of ZSTK474 on dendritic cells and inflammatory Th17 cell-mediated autoimmune central nervous system inflammation are not understood. In this study, we demonstrated that ZSTK474 suppressed human CD14+ monocyte-derived dendritic cell differentiation, maturation, and endocytosis, and further inhibited the stimulatory function of mature dendritic cell on allogeneic T cell proliferation. In addition, ZSTK474 inhibited the expression of dendritic cell-derived Th1 and Th17 cells polarizing cytokines interferon-γ/interleukin (IL)-12 and IL-6/IL-23. Furthermore, our results indicated that the in vivo administration of ZSTK474, which targets the dendritic cell and inflammatory Th1 and Th17 cell, led to a reduction of clinical score, central nervous system inflammation, and demyelination of mouse experimental autoimmune encephalomyelitis. Therefore, ZSTK474 significantly suppressed the human CD14+ monocyte-derived dendritic cell functions and ameliorated mouse experimental autoimmune encephalomyelitis. We further found that ZSTK474 inhibited the phosphorylation of PI3K downstream signaling Akt and glycogen synthase kinase 3 beta in the dendritic cell. These data suggested that ZSTK474 exerted potent anti-inflammatory and immunosuppressive properties via PI3K signaling and may serve as a potential therapeutic drug for multiple sclerosis and other autoimmune inflammatory diseases.
Key Messages
• ZSTK474 inhibits dendritic cell (DC) differentiation and maturation.
• ZSTK474 inhibits DC-derived Th1 and Th17-polarizing cytokines.
• ZSTK474 ameliorates EAE and suppresses DCs, Th1, and Th17 cells in EAE.
• ZSTK474 reduces CNS inflammation and demyelination of EAE mice.
• ZSTK474 could be a potential therapeutic drug for multiple sclerosis.








Similar content being viewed by others
Abbreviations
- PI3K:
-
Phosphatidylinositol 3-kinase
- DC:
-
Dendritic cell
- CNS:
-
Central nervous system
- EAE:
-
Experimental autoimmune encephalomyelitis
- MS:
-
Multiple sclerosis
- GSK3β:
-
Glycogen synthase kinase 3 beta
- mTOR:
-
Mammalian target of rapamycin
- APC:
-
Antigen-presenting cell
- TLR:
-
Toll-like receptor
- PKB:
-
Protein kinase B
- LPS:
-
Lipopolysaccharide
- GM-CSF:
-
Granulocyte macrophage-colony stimulating factor
- MFI:
-
Mean fluorescence intensity
References
Ruckle T, Schwarz M, Rommel C (2006) PI3Kgamma inhibition: towards an ‘aspirin of the 21st century’? Nat Rev Drug Discov 5:903–918
Fruman DA, Cantley LC (2002) Phosphoinositide 3-kinase in immunological systems. Semin Immunol 14:7–18
Rommel C, Camps M, Ji H (2007) PI3K delta and PI3K gamma: partners in crime in inflammation in rheumatoid arthritis and beyond? Nat Rev Immunol 7:191–201
Kong D, Dan S, Yamazaki K, Yamori T (2010) Inhibition profiles of phosphatidylinositol 3-kinase inhibitors against PI3K superfamily and human cancer cell line panel JFCR39. Eur J Cancer 46:1111–1121
Hu L, Zaloudek C, Mills G, Gray J, Jaffe R (2000) In vivo and in vitro ovarian carcinoma growth inhibition by a phosphatidylinositol 3-kinase inhibitor (LY294002). Clin Cancer Res 6:880–886
Ihle N, Williams R, Chow S, Chew W, Berggren M, Paine-Murrieta G, Minion D, Halter R, Wipf P, Abraham R et al (2004) Molecular pharmacology and antitumor activity of PX-866, a novel inhibitor of phosphoinositide-3-kinase signaling. Mol Cancer Ther 3:763–772
Yaguchi S, Fukui Y, Koshimizu I, Yoshimi H, Matsuno T, Gouda H, Hirono S, Yamazaki K, Yamori T (2006) Antitumor activity of ZSTK474: a new phosphatidylinositol 3-kinase inhibitor. J Natl Cancer Inst 98:545–556
Kong D, Yamori T (2007) ZSTK474 is an ATP-competitive inhibitor of class I phosphatidylinositol 3 kinase isoforms. Cancer Sci 98:1638–1642
Kong D, Yamori T (2008) Phosphatidylinositol 3-kinase inhibitors: promising drug candidates for cancer therapy. Cancer Sci 99:1734–1740
Kong D, Yaguchi S, Yamori T (2009) Effect of ZSTK474: a novel phosphatidylinositol 3-kinase inhibitor on DNA-dependent protein kinase. Biol Pharm Bull 32:297–300
Kong D, Yamori T (2009) Advances in development of phosphatidylinositol 3-kinase inhibitors. Curr Med Chem 16:2839–2854
Toyama S, Tamura N, Haruta K, Karakida T, Mori S, Watanabe T, Yamori T, Takasaki Y (2010) Inhibitory effects of ZSTK474: a novel phosphoinositide 3-kinase inhibitor, on osteoclasts and collagen-induced arthritis in mice. Arthritis Res Ther 12:R92
Krishnamoorthy G, Wekerle HEAE (2009) An immunologist’s magic eye. Eur J Immunol 39:2031–2035
Loma I, Heyman R (2011) Multiple sclerosis: pathogenesis and treatment. Curr Neuropharmacol 9(3):409–416
Hazeki K, Nigorikawa K, Hazeki O (2007) Role of phosphoinositide 3-kinase in innate immunity. Biol Pharm Bull 30:1617–1623
Yu Y, Nagai S, Wu H, Neish AS, Koyasu S, Gewirtz AT (2006) TLR5-mediated phosphoinositide 3-kinase activation negatively regulates flagellininduced proinflammatory gene expression. J Immunol 176:6194–6201
Hori S, Takahashi T, Sakaguchi S (2003) Control of autoimmunity by naturally arising regulatory CD4+ T cells. Adv Immunol 81:331–371
Sakaguchi S (2004) Naturally arising CD4+ regulatory T cells for immunologic self-tolerance and negative control of immune responses. Annu Rev Immunol 22:531–562
Dittel BN (2008) CD4 T cells: balancing the coming and going of autoimmune-mediated inflammation in the CNS. Brain Behav Immun 22(4):421–430
Stromnes IM, Goverman JM (2006) Active induction of experimental allergic encephalomyelitis. Nat Protoc 1(4):1810–1819
Ge Z, Da Y, Xue Z, Zhang K, Zhuang H, Peng M, Li Y, Li W, Simard A, Hao J et al (2013) Vorinostat, a histone deacetylase inhibitor, suppresses dendritic cell function and ameliorates experimental autoimmune encephalomyelitis. Exp Neurol 241:56–66
Xue Z, Ge Z, Zhang K, Sun R, Yang J, Han R, Peng M, Li Y, Li W, Zhang D et al (2014) Embelin suppresses dendritic cell functions and limits autoimmune encephalomyelitis through the TGF-β/β-catenin and STAT3 signaling pathways. Mol Neurobiol 49(2):1087–1101
Shakhar G, Lindquist RL, Skokos D, Dudziak D, Huang JH, Nussenzweig MC, Dustin ML (2005) Stable T cell-dendritic cell interactions precede the development of both tolerance and immunity in vivo. Nat Immunol 6:707–714
Del Prete A, Vermi W, Dander E, Otero K, Barberis L, Luini W, Bernasconi S, Sironi M, Santoro A, Garlanda C et al (2004) Defective dendritic cell migration and activation of adaptive immunity in PI3Kgamma-deficient mice. EMBO J 23:3505–3515
Haylock-Jacobs S, Comerford I, Bunting M, Kara E, Townley S, Klingler-Hoffmann M, Vanhaesebroeck B, Puri KD, McColl SR (2011) PI3Kδ drives the pathogenesis of experimental autoimmune encephalomyelitis by inhibiting effector T cellapoptosis and promoting Th17 differentiation. J Autoimmun 36(3–4):278–287
Berod L, Heinemann C, Heink S, Escher A, Stadelmann C, Drube S, Wetzker R, Norgauer J, Kamradt T (2011) PI3Kgamma deficiency delays the onset of experimental autoimmune encephalomyelitis and ameliorates its clinical outcome. Eur J Immunol 41(3):833–844
Okkenhaug K, Bilancio A, Farjot G, Priddle H, Sancho S, Peskett E, Pearce W, Meek SE, Salpekar A, Waterfield MD et al (2002) Impaired B and T cell antigen receptor signaling in p110δ PI 3-kinase mutant mice. Science 297:1031–1034
Okkenhaug K, Patton DT, Bilancio A, Garçon F, Rowan WC, Vanhaesebroeck B (2006) The p110δ isoform of PI3K controls clonal expansion and differentiation of T helper cells. J Immunol 177(8):5122–5128
Oppmann B, Lesley R, Blom B, Timans JC, Xu Y, Hunte B, Vega F, Yu N, Wang J, Singh K et al (2000) Novel p19 protein engages IL-12p40 to form a cytokine IL-23 with biological activities similar as well as distinct from IL-12. Immunity 13:715–725
Yaldizli O, Putzki N (2009) Natalizumab in the treatment of multiple sclerosis. Ther Adv Neurol Disord 2(2):115–128
Cox AL, Thompson SA, Jones JL, Robertson VH, Hale G, Waldmann H, Compston DA, Coles AJ et al (2005) Lymphocyte homeostasis following therapeutic lymphocyte depletion in multiple sclerosis. Eur J Immunol 35:3332–3342
Stromnes IM, Goverman JM (2006) Passive induction of experimental allergic encephalomyelitis. Nat Protoc 1:1952–1960
Fletcher JM, Lalor SJ, Sweeney CM, Tubridy N, Mills KH et al (2010) T cells in multiple sclerosis and experimental autoimmune encephalomyelitis. Clin Exp Immunol 162(1):1–11
Mills KH (2008) Induction, function and regulation of IL-17-producing T cells. Eur J Immunol 38:2636–2649
Acknowledgments
We thank the Tianjin Blood Center for providing us with the buffy coat. We also thank the Experimental Animal Center of Tianjin Medical University (Tianjin, China) for their work in animal husbandry. This work is supported by the Ministry of Science and Technology of China through grant nos. 2012CB932503 and 2011CB933100; the National Natural Science Foundation of China through grant nos. 91029705, 81272317, and 81172864; and the Natural Science Foundation of Tianjin through grant no. 12JCZDJC23500.
Disclosure
The authors have nothing to disclose.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Zhenyi Xue, Wen Li, and Huafeng Wang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xue, Z., Li, W., Wang, H. et al. ZSTK474, a novel PI3K inhibitor, modulates human CD14+ monocyte-derived dendritic cell functions and suppresses experimental autoimmune encephalomyelitis. J Mol Med 92, 1057–1068 (2014). https://doi.org/10.1007/s00109-014-1158-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-014-1158-x