Abstract
Background
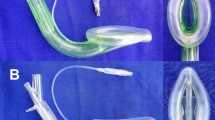
Supraglottic airway devices perform more poorly and have lower oropharyngeal leak pressure in edentulous patients than in patients with teeth. The Ambu Aura Gain is a newer second generation supraglottic airway device.
Objective
This randomized clinical trial assessed the oropharyngeal leak pressure in edentulous patients using the Ambu Aura Gain with a gastric tube for insertion guidance and without insertion guidance.
Material and methods
Patients with ASA (American Society of Anesthesiology) physical status I–III were recruited. Primary outcome was oropharyngeal leak pressure after insertion. Secondary outcome parameters were oropharyngeal leak pressure 15 min and 30 min after insertion, insertion time, insertion attempts and glottis view through flexible fiberscope.
Results
In this study 72 patients aged between 51 and 90 years (mean 73 years) were randomly allocated to the “with guidance” (n = 36) or the “without guidance” group (n = 36). Mean (SD) oropharyngeal leak pressure in “with guidance” and “without guidance” group was 24 cm H2O and 24 cm H2O (ns), respectively. A difference was found in mean insertion time with guidance versus without guidance group 52 s (45 s) vs. 26 s (15 s) (p < 0.001). No difference was found in any of the other secondary outcome parameters.
Conclusion
A guided insertion technique does not improve oropharyngeal leak pressure of the Ambu AuraGain™ in edentulous patients. As the only difference is an increase in insertion time this technique is of no benefit for this population.
Zusammenfassung
Hintergrund
Supraglottische Atemwegshilfen zeigen bei zahnlosen Patienten im Vergleich zu Patienten mit Zähnen deutliche Schwächen in der Performance und erreichen niedrigere oropharyngeale Verschlussdrücke.
Ziel der Arbeit
Die Ambu AuraGain™ ist eine relativ neue supraglottische Atemwegshilfe der zweiten Generation. Ziel der vorliegenden randomisierten Studie ist es, den oropharyngealen Verschlussdruck der Ambu AuraGain™ in Abhängigkeit von der Insertionstechnik (mit Magensonde als Führungsschiene oder konventionell ohne Führungsschiene) zu untersuchen.
Material und Methoden
Es wurden Patienten mit einem ASA(American Society of Anesthesiology)-Status I–III eingeschlossen. Als primäre Outcome-Parameter wurde der oropharyngeale Verschlussdruck unmittelbar nach Insertion gewählt. Sekundäre Outcome-Parameter waren oropharyngealer Verschlussdruck 15 min und 30 min nach Insertion, die Insertionszeit, Anzahl der Insertionsversuche und die fiberooptische Lageevaluation der Glottis.
Ergebnisse
Insgesamt 72 Patienten zwischen 51 und 90 Jahren (Mittelwert: 73) wurden nach Randomisierung der Gruppe mit Führung (n = 36) oder ohne Führung (n = 36) zugewiesen. Der mittlere (SD) oropharyngeale Verschlussdruck betrug sowohl in der Gruppe mit als auch in der Gruppe ohne Führung 24 cm H2O (ns). Ein deutlicher Unterschied ergab sich in der Insertionszeit (Gruppe mit vs ohne Führung 66 s [45] vs. 32 s [15]; p < 0.001). Auch in alle weiteren sekundären Endpunkten zeigten sich keine signifikanten Unterschiede zwischen den zwei Einführungstechniken.
Diskussion
Unsere Studie zeigt, dass eine geführte Insertion bei zahnlosen Patienten den oropharyngealen Verschlussdruck der Ambu AuraGain™ Larynxmaske nicht verbessert. Jedoch im Vergleich zu anderen untersuchten supraglottischen Atemwegshilfen ist der oropharyngeale Verschlussdruck der Ambu AuraGain™ bei diesem Patientenkollektiv deutlich höher.
Similar content being viewed by others
References
Cited Literature
Beydeş T, Küçükgüçlü S, Özbilgin Ş, Kuvaki B, Ademoğlu M, Sarı M (2016) Comparison of laryngeal mask Airway Supreme(TM) versus Unique(TM) in edentulous geriatric patients. Turk J Anaesthesiol Reanim 44(1):32–36. https://doi.org/10.5152/TJAR.2016.22129
Brimacombe J (2004) Laryngeal mask anesthesia. Principles and practice, 2nd edn. WB Saunders, London
Brimacombe J, Berry A (1993) A proposed fiber-optic scoring system to standardize the assessment of laryngeal mask airway position. Anesth Analg 76(2):457
Brimacombe J, Keller C (2002) The ProSeal laryngeal mask airway. Anesthesiol Clin North Am 20(4):871–891. https://doi.org/10.1016/s0889-8537(02)00044-5
Brimacomb J, Keller C, Kurian S, Myles J (2002) Reliability of epigastric auscultation to detect gastric insufflation. Br J Anaesth 88(1):127–129. https://doi.org/10.1093/bja/88.1.127
Brimacombe J, Keller C, Judd DV (2004) Gum elastic bougie-guided insertion of the ProSeal laryngeal mask airway is superior to the digital and introducer tool techniques. Anesthesiology 100(1):25–29. https://doi.org/10.1097/00000542-200401000-00008
Cholmakow-Bodechtel C, Füßl-Grünig E, Geyer S, Hertrampf K, Hoffmann T, Holtfreter T et al (2016) DMS V Fünfte deutsche Mundgesundheitsstudie vom Institut der Deutschen Zahnärzte. Deutscher Zahnärzte Verlag DÄV, Köln
Dixon GS, Thomson WM, Kruger E (1999) The West Coast Study I: self-reported dental health and the use of dental services. N Z Dent J 95(420):38–43
Genez M, Küçükgüçlü S, Özbilgin Ş, Kuvaki B, Beydeş T, Aksoy Sari M (2017) A comparison of usage of the laryngeal mask Unique™ in denticulate and edentulate geriatric patients. Turk J Med Sci 47(3):854–860. https://doi.org/10.3906/sag-1603-206
Keller C, Brimacombe J (1998) Bronchial mucus transport velocity in paralyzed anesthetized patients: a comparison of the laryngeal mask airway and cuffed tracheal tube. Anesth Analg 86(6):1280–1282. https://doi.org/10.1097/00000539-199806000-00028
Keller C, Brimacombe JR, Keller K, Morris R (1999) Comparison of four methods for assessing airway sealing pressure with the laryngeal mask airway in adult patients. Br J Anaesth 82(2):286–287. https://doi.org/10.1093/bja/82.2.286
Keller C, Brimacombe J, Bittersohl J, Lirk P, von Goedecke A (2004) Aspiration and the laryngeal mask airway: three cases and a review of the literature. Br J Anaesth 93(4):579–582. https://doi.org/10.1093/bja/aeh228
Kim GW, Kim JY, Kim SJ, Moon YR, Park EJ, Park SY (2019) Conditions for laryngeal mask airway placement in terms of oropharyngeal leak pressure: a comparison between blind insertion and laryngoscope-guided insertion. BMC Anesthesiol 19(1):4. https://doi.org/10.1186/s12871-018-0674-6
Lopez-Gil M, Brimacombe J, Alvarez M (1996) Safety and efficacy of the laryngeal mask airway. A prospective survey of 1400 children. Anaesthesia 51(10):969–972. https://doi.org/10.1111/j.1365-2044.1996.tb14968.x
Moser B, Audige L, Keller C, Brimacombe J, Gasteiger L, Bruppacher HR (2018) A prospective, randomized trial of the Ambu AuraGain™laryngeal mask versus LMA protector airway in paralyzed, anesthetized adult men. Minerva Anestesiol 84(6):684–692
Oczenski W, Krenn H, Dahaba AA, Binder M, El-Schahawi-Kienzl I, Jellinek H (1999) Hemodynamic and catecholamine stress responses to insertion of the Combitube, laryngeal mask airway or tracheal intubation. Anesth Analg 88(6):1389–1394. https://doi.org/10.1097/00000539-199906000-00035 (published correction appears in Anesth Analg. 2017 Mar;124(3):1024)
Pennant JH, White PF (1993) The laryngeal mask airway. Its uses in anesthesiology. Anesthesiology 79(1):144–163. https://doi.org/10.1097/00000542-199307000-00021
Robbins J (1996) Normal swallowing and aging. Semin Neurol 16(4):309–317. https://doi.org/10.1055/s-2008-1040989
Robbins J (1999) Old swallowing and dysphagia: thoughts on intervention and prevention. Nutr Clin Pract 14(5):21–26
Stögermüller B, Ofner S, Ziegler B, Keller C, Moser B, Gasteiger L (2019) Ambu® Aura Gain™ versus Ambu® Aura Once™ in children: a randomized, crossover study assessing oropharyngeal leak pressure and fibreoptic position. Can J Anaesth 66(1):57–62. https://doi.org/10.1007/s12630-018-1235-7
Tiefenthaler W, Eschertzhuber S, Brimacombe J, Fricke E, Keller C, Kaufmann M (2013) A randomised, non-crossover study of the Guardian CPV Laryngeal Mask versus the LMA Supreme in paralysed, anaesthetised female patients. Anaesthesia 68(6):600–604. https://doi.org/10.1111/anae.12178
Timmermann A, Bergner UA, Russo SG (2015) Laryngeal mask airway indications: new frontiers for second-generation supraglottic airways. Curr Opin Anaesthesiol 28(6):717–726. https://doi.org/10.1097/ACO.0000000000000262
Weiler N, Eberle B, Heinrichs W (1999) The laryngeal mask airway: routine, risk or rescue? Intensive Care Med 25(7):761–762
Further Reading
Conlon NP, Sullivan RP, Herbison PG, Zacharias M, Buggy DJ (2007) The effect of leaving dentures in place on bag-mask ventilation at induction of general anesthesia. Anesth Analg 105(2):370–373. https://doi.org/10.1213/01.ane.0000267257.45752.31
Gasteiger L, Oswald E, Keplinger M, Putzer G, Luger M, Neururer S et al (2020) A randomised trial comparing the Ambu® Aura‑i TM and the Ambu® Aura Gain TM laryngeal mask as conduit for tracheal intubation in children. Minerva Anestesiol. https://doi.org/10.23736/S0375-9393.20.14422-5
Moser B, Audige L, Keller C, Brimacombe J, Gasteiger L, Bruppacher HR (2017) Flexible bronchoscopic intubation through the AuraGainTM laryngeal mask versus a slit Guedel tube: a non-inferiority randomized-controlled trial. Can J Anaesth 64(11):1119–1128
Wong DT, Ooi A, Singh KP et al (2018) Comparison of oropharyngeal leak pressure between the Ambu® AuraGain™ and the LMA® Supreme™ supraglottic airways: a randomized-controlled trial. Can J Anaesth 65(7):797–805. https://doi.org/10.1007/s12630-018-1120-4
Funding
This project was supported only by departmental resources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
L. Gasteiger, H. Tauber, C. Velik-Salchner, M. Thoma, R. Fantin, V. Pustilnik, S. Neururer, C. Keller and B. Moser declare that they have no competing interests.
Ethical standards
This report describes human research. IRB contact information: Ethikkommission der Medizinischen Universität Innsbruck—Austria, Christoph-Probst Platz 1, Innrain 52, 6020 Innsbruck mail: i‑master@i-med.ac.at, https://www.i-med.ac.at/ethikkommission/index. This study was conducted with written informed consent from the study subjects. The study was conducted according to the Helsinki Declaration. This report describes a prospective randomized clinical trial. The author states that the report includes every item in the CONSORT checklist for a prospective randomised clinical trial. This was not an observational clinical study. This manuscript was screened for plagiarism using Plagiarism Checker.
Rights and permissions
About this article
Cite this article
Gasteiger, L., Tauber, H., Velik-Salchner, C. et al. Guided vs. non-guided insertion of Ambu AuraGain™ in edentulous patients. Anaesthesist 70, 761–767 (2021). https://doi.org/10.1007/s00101-021-00914-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00101-021-00914-x