Abstract
Background and purpose
The aim of this study was to evaluate an automatic multi-atlas-based segmentation method for generating prostate, peripheral (PZ), and transition zone (TZ) contours on MRIs with and without fat saturation (±FS), and compare MRIs from different vendor MRI systems.
Methods
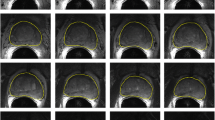
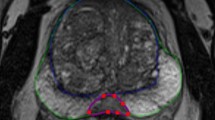
T2-weighted (T2) and fat-saturated (T2FS) MRIs were acquired on 3T GE (GE, Waukesha, WI, USA) and Siemens (Erlangen, Germany) systems. Manual prostate and PZ contours were used to create atlas libraries. As a test MRI is entered, the procedure for atlas segmentation automatically identifies the atlas subjects that best match the test subject, followed by a normalized intensity-based free-form deformable registration. The contours are transformed to the test subject, and Dice similarity coefficients (DSC) and Hausdorff distances between atlas-generated and manual contours were used to assess performance.
Results
Three atlases were generated based on GE_T2 (n = 30), GE_T2FS (n = 30), and Siem_T2FS (n = 31). When test images matched the contrast and vendor of the atlas, DSCs of 0.81 and 0.83 for T2 ± FS were obtained (baseline performance). Atlases performed with higher accuracy when segmenting (i) T2FS vs. T2 images, likely due to a superior contrast between prostate vs. surrounding tissue; (ii) prostate vs. zonal anatomy; (iii) in the mid-gland vs. base and apex. Atlases performance declined when tested with images with differing contrast and MRI vendor. Conversely, combined atlases showed similar performance to baseline.
Conclusion
The MRI atlas-based segmentation method achieved good results for prostate, PZ, and TZ compared to expert contoured volumes. Combined atlases performed similarly to matching atlas and scan type. The technique is fast, fully automatic, and implemented on commercially available clinical platform.
Zusammenfassung
Hintergrund und Zweck
Ziel der Studie war es, eine automatische multi-atlasbasierende Segmentierungsmethode zur Erzeugung von Prostata-, peripheren und Übergangszonenkonturen (PZ/TZ) auf Magnetresonanztomographie(MRT)-Bildern mit und ohne Fettsättigung (±FS) zu beurteilen und MRT-Systeme verschiedener Hersteller zu vergleichen.
Methoden
T2-gewichtete (T2) und fettgesättigte (T2FS) MRTs wurden auf 3T-Systemen von GE (GE, Waukesha, WI, USA) und Siemens (Erlangen, Deutschland) aufgenommen. Manuelle Prostata- und PZ-Konturen wurden verwendet, um Atlasbibliotheken zu erstellen. Nach dem Einlesen eines MRT-Testdatensatzes identifiziert das Verfahren zur Atlassegmentierung automatisch die Atlasobjekte, die am besten zum Testobjekt passen, gefolgt von einer normalisierten, intensitätsbasierenden, frei deformierbaren Registrierung. Die Konturen werden dem Testobjekt angepasst und die „Dice Similarity Coefficients“ (DSC) und der Hausdorff-Abstand zwischen atlasgenerierten und manuellen Konturen verwendet, um die Übereinstimmung zu beurteilen.
Ergebnisse
Drei Atlanten wurden basierend auf GE_T2 (n = 30), GE_T2FS (n = 30) und Siem_T2FS (n = 31) erstellt. Wenn die Testbilder mit dem gewählten Kontrast und Atlashersteller übereinstimmten, wurden DSC von 0,81 und 0,83 für T2 ± FS erzielt (Ausgangswert). Atlanten erreichten eine höhere Genauigkeit beim Segmentieren von: (i) T2FS-Bildern verglichen mit T2-Bildern, wahrscheinlich aufgrund des besseren Kontrasts zwischen Prostata und umgebendem Gewebe auf T2FS-Bildern; (ii) Prostata verglichen mit zonaler Anatomie; (iii) der Drüsenmitte verglichen mit Basis und Apex. Die Qualität der Atlanten ging zurück, wenn sie mit Bildern mit unterschiedlichem Kontrast und MRT-Gerät getestet wurden. Umgekehrt zeigten kombinierte Atlanten eine ähnliche Übereinstimmung und Qualität wie der Ausgangswert.
Schlussfolgerung
Die atlasbasierende MRT-Segmentierungsmethode erzielte gute Ergebnisse für Prostata, PZ und TZ im Vergleich zu konturierten Volumina. Kombinierte Atlanten erreichten eine ähnliche Übereinstimmung und Genauigkeit wie passender Atlas- und Scan-Typ. Die Technik ist schnell, vollautomatisch und auf einer kommerziell erhältlichen klinischen Plattform integriert.


Similar content being viewed by others
References
Barentsz JO, Richenberg J, Clements R et al (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22:746–757
Weinreb JC, Barentsz JO, Choyke PL et al (2016) PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol 69:16–40
Stoyanova R, Takhar M, Tschudi Y et al (2016) Prostate cancer radiomics and the promise of radiogenomics. Transl Cancer Res 5:432–447
Rastinehad AR, Turkbey B, Salami SS et al (2014) Improving detection of clinically significant prostate cancer: magnetic resonance imaging/transrectal ultrasound fusion guided prostate biopsy. J Urol 191:1749–1754
Wysock JS, Rosenkrantz AB, Huang WC et al (2014) A prospective, blinded comparison of magnetic resonance (MR) imaging-ultrasound fusion and visual estimation in the performance of MR-targeted prostate biopsy: the PROFUS trial. Eur Urol 66:343–351
Haffner MC, Mosbruger T, Esopi DM et al (2013) Tracking the clonal origin of lethal prostate cancer. J Clin Invest 123:4918–4922
Lips IM, van der Heide UA, Haustermans K et al (2011) Single blind randomized phase III trial to investigate the benefit of a focal lesion ablative microboost in prostate cancer (FLAME-trial): study protocol for a randomized controlled trial. Trials 12:255
Bauman G, Haider M, Van der Heide UA, Menard C (2013) Boosting imaging defined dominant prostatic tumors: a systematic review. Radiother Oncol 107:274–281
Hocht S, Aebersold DM, Albrecht C et al (2017) Hypofractionated radiotherapy for localized prostate cancer. Strahlenther Onkol 193:1–12
Pathmanathan AU, van As NJ, Kerkmeijer LGW et al (2018) Magnetic resonance imaging-guided adaptive radiation therapy: a “game changer” for prostate treatment? Int J Radiat Oncol Biol Phys 100:361–373
Hild S, Graeff C, Rucinski A et al (2016) Scanned ion beam therapy for prostate carcinoma: comparison of single plan treatment and daily plan-adapted treatment. Strahlenther Onkol 192:118–126
Litjens G, Toth R, van de Ven W et al (2014) Evaluation of prostate segmentation algorithms for MRI: the PROMISE12 challenge. Med Image Anal 18:359–373
Ou YDJ, Erus G, Davatzikos C (2012) Multi_atlas segmentation of the prostate; a zooming process with robust registration and atlas selection. MICCAI grand challenge: prostate MR image segmentation 2012. https://promise12.grand-challenge.org/. Accessed: 1 Oct 2012
Ou Y, Sotiras A, Paragios N, Davatzikos CDRAMMS (2011) Deformable registration via attribute matching and mutual-saliency weighting. Med Image Anal 15:622–639
Klein S, van der Heide UA, Lips IM, van Vulpen M, Staring M, Pluim JPW (2008) Automatic segmentation of the prostate in 3D MR images by atlas matching using localized mutual information. Med Phys 35:1407–1417
Hegde JV, Mulkern RV, Panych LP et al (2013) Multiparametric MRI of prostate cancer: an update on state-of-the-art techniques and their performance in detecting and localizing prostate cancer. J Magn Reson Imaging 37:1035–1054
Bhavsar A, Verma S (2014) Anatomic imaging of the prostate. Biomed Res Int 2014:728539
Xie Q, Ruan D (2014) Low-complexity atlas-based prostate segmentation by combining global, regional, and local metrics. Med Phys 41:41909
Cheng R, Turkbey B, Gandler W et al (2014) Atlas based AAM and SVM model for fully automatic MRI prostate segmentation. Conf Proc Ieee Eng Med Biol Soc 2014:2881–2885
Korsager AS, Fortunati V, van der Lijn F et al (2015) The use of atlas registration and graph cuts for prostate segmentation in magnetic resonance images. Med Phys 42:1614–1624
Chilali O, Puech P, Lakroum S, Diaf M, Mordon S, Betrouni N (2016) Gland and zonal segmentation of prostate on T2W MR images. J Digit Imaging 29:730–736. https://doi.org/10.1007/s10278-016-9890-0
Wu K, Garnier C, Alirezaie J, Dillenseger JL (2014) Adaptation and evaluation of the multiple organs OSD for T2 MRI prostate segmentation. Conf Proc Ieee Eng Med Biol Soc 2014:4687–4690
Makni N, Iancu A, Colot O, Puech P, Mordon S, Betrouni N (2011) Zonal segmentation of prostate using multispectral magnetic resonance images. Med Phys 38:6093–6105
Litjens G, Debats O, van de Ven W, Karssemeijer N, Huisman H (2012) A pattern recognition approach to zonal segmentation of the prostate on MRI. Med Image Comput Comput Assist Interv 15:413–420
Maan B, van der Heijden F, Futterer JJ (2012) A new prostate segmentation approach using multispectral Magnetic Resonance Imaging and a statistical pattern classifier. Proc SPIE Int Soc Opt Eng 8314. https://doi.org/10.1117/12.911194
Chowdhury N, Toth R, Chappelow J et al (2012) Concurrent segmentation of the prostate on MRI and CT via linked statistical shape models for radiotherapy planning. Med Phys 39:2214–2228
Toth R, Madabhushi A (2012) Multifeature landmark-free active appearance models: application to prostate MRI segmentation. IEEE Trans Med Imaging 31:1638–1650
Tian Z, Liu L, Fei B (2015) A fully automatic multi-atlas based segmentation method for prostate MR images. Proc Spie Int Soc Opt Eng 9413. https://doi.org/10.1117/12.2082229
Rothke M, Blondin D, Schlemmer HP, Franiel T (2013) PI-RADS classification: structured reporting for MRI of the prostate. Fortschr Röntgenstr 185:253–261
Rosenkrantz AB, Taneja SS (2014) Radiologist, be aware: ten pitfalls that confound the interpretation of multiparametric prostate MRI. Ajr Am J Roentgenol 202:109–120
Dice LR (1945) Measures of the amount of ecologic association between species. Ecology 26:297–302
Hausdorff F (1914) Grundzüge der Mengenlehre. Veit, Leipzig
Zou KH, Warfield SK, Bharatha A et al (2004) Statistical validation of image segmentation quality based on a spatial overlap index. Acad Radiol 11:178–189
Huttenlocher DP, Klanderman GA, Rucklidge WJ (1993) Comparing images using the Hausdorff distance. Ieee T Pattern Anal 15:850–863
Piper J, Nelson A, Harper J (2013) Deformable image registration in MIM Maestro™. Evaluation and description. MIMsoftware, Ohio, p 5
Johnson PB, Padgett KR, Chen KL, Dogan N (2016) Evaluation of the tool “Reg Refine” for user-guided deformable image registration. J Appl Clin Med Phys 17:158–170
Warfield SK, Zou KH, Wells WM (2004) Simultaneous truth and performance level estimation (STAPLE): an algorithm for the validation of image segmentation. IEEE Trans Med Imaging 23:903–921
Hoeks CMA, Barentsz JO, Hambrock T et al (2011) Prostate cancer: multiparametric MR imaging for detection, localization, and staging. Radiology 261:46–66
McGurk RJ, Bowsher J, Lee JA, Das SK (2013) Combining multiple FDG-PET radiotherapy target segmentation methods to reduce the effect of variable performance of individual segmentation methods. Med Phys 40:42501
Rohlfing T, Brandt R, Menzel R, Maurer CR Jr. (2004) Evaluation of atlas selection strategies for atlas-based image segmentation with application to confocal microscopy images of bee brains. Neuroimage 21:1428–1442
Fiorino C, Reni M, Bolognesi A, Cattaneo GM, Calandrino R (1998) Intra- and inter-observer variability in contouring prostate and seminal vesicles: implications for conformal treatment planning. Radiother Oncol 47:285–292
Choi HJ, Kim YS, Lee SH et al (2011) Inter- and intra-observer variability in contouring of the prostate gland on planning computed tomography and cone beam computed tomography. Acta Oncol 50:539–546
Lutgendorf-Caucig C, Fotina I, Stock M, Potter R, Goldner G, Georg D (2011) Feasibility of CBCT-based target and normal structure delineation in prostate cancer radiotherapy: multi-observer and image multi-modality study. Radiother Oncol 98:154–161
Villeirs GM, Van Vaerenbergh K, Vakaet L et al (2005) Interobserver delineation variation using CT versus combined CT + MRI in intensity-modulated radiotherapy for prostate cancer. Strahlenther Onkol 181:424–430
Acknowledgements
This work was supported by National Cancer Institute (R01CA189295 and R01CA190105 to A.P.). We would also like to thank Randi Steinhagen and Dr. rer. nat. Dipl.-Phys. Ellen Ackerstaff for translating our abstract into German.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. Swallen, S. Pirozzi, J. Piper, and A. Nelson were employed by MIM Software Inc. during the duration of this investigation. Additionally, J. Piper and A. Nelson have an ownership interest in MIM Software Inc. K.R. Padgett, F.M. Chinea, M.C. Abramowitz, A. Pollack, and R. Stoyanova declare that they have no competing interests.
Caption Electronic Supplementary Material
Supplementary Table S1
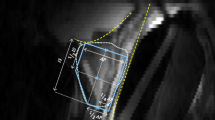
: Patient Clinical Characteristics. Supplementary Table S2: Comparison of manually drown contours on GE_T2 and GE_T2FS sequences. In the second part of the table, the reproducibility of the prostate contour in three sections of the prostate is examined. Supplementary Table S3: Comparison of manually drown contours on GE_T2FS by two expert radiation oncologists (inter-reader comparison). Supplementary Table S4: Summary of contrast comparisons. The ratio of the signal intensity of the inner rind and the outer rind is presented along with the ratio of the signal intensity between the peripheral and transition zones (PZ/TZ). Supplementary Figure S1: (top) The inner and outer rinds that were used for contrast comparisons between T2 and T2FS acquisitions; (bottom) Contrast comparisons between manually drawn TZ and PZ contours for the T2 and T2FS scans from the same patient
Rights and permissions
About this article
Cite this article
Padgett, K.R., Swallen, A., Pirozzi, S. et al. Towards a universal MRI atlas of the prostate and prostate zones. Strahlenther Onkol 195, 121–130 (2019). https://doi.org/10.1007/s00066-018-1348-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-018-1348-5