Abstract
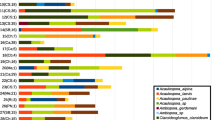
Solidago altissima is a dominant perennial of old field succession in North America with the ability to form dense, almost monospecific stands; the plant is also considered an aggressive invasive throughout Europe and Japan. S. altissima’s success is facilitated by large belowground rhizome systems that allow clonal spread and possible allelopathic suppression of competitors. A diversity of polar and nonpolar terpenes and polyacetylenes have been described from S. altissima rhizomes; however, there is little information documenting the concentration or distribution of these allelochemicals and how they relate to plant defense hypotheses. We thus developed a GC–MS method to simultaneously quantify rhizome terpenes and polyacetylenes that spanned a range of polarities and confirmed the presence of 19 terpenoids in addition to the polyacetylene dehydromatricaria ester (DME). We, (1) determined relative concentrations and variability of mono-, sesqui-, neutral diterpenes, diterpene acids and DME within a central NY population; (2) compared accumulation of these compounds in differently developed rhizome tissue; and (3) tested the alleopathic effect of DME at naturally occurring concentrations on germination and seedling growth of Asclepias syriaca, a common competitor of S. altissima. Overall, diterpene acids dominated the phytochemical profile of most genotypes (averaging 0.75% of fresh mass) with kolavenic acid being the single most abundant component. Monoterpene, sesquiterpene and acidic diterpene concentrations were all significantly greater in the actively elongating (current-year) rhizomes compared to established rhizomes and followed optimal defense theory predictions. Conversely, DME levels were lower in the current-year rhizomes than in established rhizomes. DME inhibited seed germination of A. syriaca in a dose-dependent manner. The tissue-specific accumulation of the different compounds may represent a biosynthetic and/or allocational strategy that limits the exposure of young-tissue to high concentrations of DME possibly limiting the cytotoxic effects of DME.



Similar content being viewed by others
References
Abhilasha D, Quintana N, Vivanco J, Joshi J (2008) Do allelopathic compounds in invasive Solidago canadensis restrain the native European flora? J Ecol 96:993–1001
Abrahamson WG, Anderson SS, McCrea KD (1991) Clonal integration: nutrient sharing between Sister Ramets of Solidago altissima (Compositae). Am J Bot 78(11):1508–1514
Angelo C, Pinto AC, Braga WF, Rezende CM, Garrido FMS, Veiga VF, Bergter L, Patitucci ML, Antunes OAC (2000) Separation of acid diterpenes of Copaifera cearensis Huber ex Ducke by flash chromatography using potassium hydroxide impregnated silica gel. J Braz Chem Soc 11(4):355–360
Anthonsen T, Henderson MS, Martin A, Murray RDH, McCrindle R, McMaster D (1973) Constituents of Solidago species. Part IV. Solidagoic acids A and B, diterpenoids from Solidago gigantea var. serotina. Can J Chem 51:1332–1345
Auld B, Morita H, Nishida T, Itol M, Michae P (2003) Shared exotica: plant invasions of Japan and south eastern Australia. Cunninghamia 8(1):147–152
Benevides PJC, Young MCM, Bolzani VD (2004) Biological activities of constituents from Psychotria spectabilis. Pharm Biol 42(8):565–569
Bohlmann F, Singh P, Singh RK, Joshi K, Jaicupovic J (1985) A diterpene with a new carbon skeleton from Solidago altissima. Phytochemistry 24(5):1114–1115
Cain ML (1990) Patterns of Solidago altissima ramet growth and mortality: the role of below-ground ramet connections. Oecologia 82(2):201–209
Capek M (1971) The possibility of biological control of imported weeds of the genus Solidago in Europe. Acta Instituti Forestalis Zvolenensis 1971:429–441
Curtis JD, Lersten NR (1990) Oil reservoirs in stem, rhizome and root of Solidago canadensis (Asteraceae, tribe Astereae). Nord J Bot 10(4):443–449
Etterson JR, Delf DE, Craig TP, Ando Y, Ohgushi T (2008) Parallel patterns of clinal variation in Solidago altissima in its native range in central USA and its invasive range in Japan. Botany 86:91–97
Haig T (2008) Allelochemicals in plants. In: Zeng RS, Azim U, Mallik AU, Luo SM (eds) Allelopathy in sustainable agriculture and forestry. Springer, Berlin, pp 63–104
Hartnett DC, Bazazz FA (1984) Physiological integration among intraclonal ramets in Solidago canadensis. Ecology 64(4):779–788
Henderson MS, McCrindle R, McMaster D (1973) Constituents of Solidago species part V. Non-acidic diterpenoids form Solidago gigantea var. serotina. Can J Chem 51:1346–1358
Hull-Sanders HM, Clare R, Johnson RH, Meyer GA (2007) Evaluation of the evolution of increased competitive ability (EICA) hypothesis: loss of defense against generalist but not specialist herbivores. J Chem Ecol 33:781–799
Johnson RH, Hull-Sanders HM, Meyer GA (2007) Comparison of foliar terpenes between native and invasive Solidago gigantea. Biochem Syst Ecol 35(12):821–830
Kagan J, Kolyvas CP, Lam J (1980) The ovicidal activity of cis-dehydromatricaria ester: time-dependance of its enhancement by UV light. Experientia 40:1396–1397
Kalemba D, Marschall H, Bradesi P (2001) Constituents of the essential oil of Solidago gigantea Ait (Giant goldenrod). Flav Fragr J 16:19–26
Kobayashi A, Morimoto S, Shibata Y, Yamashita K, Numata M (1980) C10-polyacetylenes as allelopathic substances in dominants in early stages of secondary succession. J Chem Ecol 6(1):119–131
Krügel T, Lim M, Gase K, Halitschke R, Baldwin IT (2002) Agrobacterium-mediated transformation of Nicotiana attenuata, a model ecological expression system. Chemoecology 12:177–183
Kuhnel E, Laffan DDP, Lloyd-Jones GC, del Campo TM, Shepperson IR, Slaughter JL (2007) Mechanism of methyl esterification of carboxylic acids by trimethylsilyldiazomethane. Angew Chem Int Ed 46:7075–7078
Lewinsohn E, Savage TJ, Gijzen M, Croteau R (1993) Simultaneous analysis of monoterpenes and diterpenoids of conifer oleoresin. Phytochem Meth 4:220–225
Liu J, Liang SC, Liu FH, Wang RQ, Dong M (2005) Invasive alien plant species in China: regional distribution patterns. Div Dist 11:341–347
Lu T, Menelaou MA, Vargas D, Fronczek FR, Fisher NH (1993) Polyacetylenes and diterpenes from Solidago canadensis. Phytochemistry 32(6):1483–1488
Maddox GD, Root R (1990) Structure of the encounter between goldenrod (Solidago altissima) and its diverse insect fauna. Ecology 71(6):2115–2124
Maddox GD, Cook RE, Wimberger PH, Gardescu S (1989) Clone structure in four Solidago altissima (Asteraceae) populations: rhizome connections within genotypes. Am J Bot 76:318–326
Manabe S, Nishino C (1986) Stereochemistry of cis-clerodane diterpenes. Tetrahedron 42(13):3461–3470
Matsunaga H, Katano M, Tasaki M, Yamamoto H, Mori M, Takata K (1990) Inhibitory effect of cis-dehydromatricaria ester isolated from Solidago altissima on the growth of mammalian cells. Chem Pharm Bull 38(12):3483–3484
McLachlan D, Arason T, Lam J (1984) Structure–function relationships in the phototoxicity of acetylenes from the Asteraceae. Biochem Syst Ecol 14(1):17–23
Merritt AT, Ley SV (1992) Clerodane diterpenoids. Nat Prod Rep 9:243–286
Meyer AH, Schmid B (1999a) Experimental demography of the old-field perennial Solidago altissima: the dynamics of the shoot population. J Ecol 87(1):17–27
Meyer AH, Schmid B (1999b) Seed dynamics and seedling establishment in the invading perennial Solidago altissima under different experimental treatments. J Ecol 87(1):28–41
Meyer G, Clare R, Weber E (2005) An experimental test of the evolution of increased competitive ability hypothesis in goldenrod, Solidago gigantea. Oecologia 144:299–307
Morrow PA, Olfelt JP (2003) Phoenix clones: recovery after long-term defoliation induced dormancy. Ecol Lett 6:119–125
Preus LE, Morrow PA (1999) Direct and indirect effects of two herbivore species on resource allocation in their shared host plant: the rhizome galler Eurosta comma, the folivore Trirhabda canadensis and Solidago missouriensis. Oecologia 119:219–226
Rhoades DF (1979) Evolution of plant defense against herbivores. In: Rosenthal GA, Janzen DH (eds) Herbivores: their interaction with secondary metabolites. Academic Press, New York, pp 1–55
Root RB, Cappuccino N (1992) Patterns in population change and the organization of the insect community associated with goldenrod. Ecol Monogr 62:393–420
Rostas M, Eggert K (2008) Ontogenetic and spatio-temporal patterns of induced volatiles in Glycine max in the light of the optimal defense hypothesis. Chemoecology 18:29–38
Sawabe A, Minimoto K, Ouchi S, Okamoto K (1999) Effects of acetylenes and terpenoids from Solidago altissima L. on seed germination. J Jpn Oil Chem Soc 48(2):139–142
Sawabe A, Minimoto K, Minematsu T, Morita M, Ouchi S, Okamoto T (2000) Characterization of acetylenes and terpenoids isolated from Solidago altísima L. Bull Inst Compr Agr Sci Kinki Univ 8:81–88
Schmelz EA, Engelberth J, Alborn HT, O’Donnell P, Sammons M, Toshima H, Tumlinson JH (2003) Simultaneous analysis of phytohormones, phytotoxins, and volatile organic compounds in plants. Proc Natl Acad Sci USA 100(18):10552–10557
Semple JC, Cook RE (2006) Solidago. In: Flora North America Editorial Committee (ed) Flora of North America. Oxford University Press, Oxford, pp 107–166
Tori M, Katto A, Sono M (1999) Nine new clerodane diterpenoids from rhizomes of Solidago altissima. Phytochemistry 52:487–493
Tsao R, Eto M (1996) Light-activated plant growth inhibitory activity of cis-dehydromatricaria ester, rose bengal and fluoren-9-one on lettuce (Lactuca sativa L.). Chemosphere 32(7):1307–1317
Weber E (2001) Current and potential ranges of three exotic goldenrods (Solidago) in Europe. Conserv Biol 15:122–128
Werner PA, Bradbury IK, Gross RS (1980) The biology of Canadian weeds. 45. Solidago canadensis L. Can J Plant Sci 60:1393–1409
Zhang Q, Yao LJ, Yang RY, Yang XY, Tang JJ, Chen X (2007) Potential allelopathic effects of an invasive species Solidago canadensis on the mycorrhizae of native plant species. Allelopathy J 20(1):71–78
Acknowledgments
This study was funded by a USDA Federal Formula Fund grant (NYC-183420), Cornell University and a Medaille College sabbatical grant. The authors thank Richard Root for fruitful Solidago discussions and technical assistance, Anarag Agrawal for providing A. syriaca seeds and Paul Feeny for the generous use of laboratory facilities. The authors also thank two anonymous reviewers and the editorial staff for substantial improvements to the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Johnson, R.H., Halitschke, R. & Kessler, A. Simultaneous analysis of tissue- and genotype-specific variation in Solidago altissima (Asteraceae) rhizome terpenoids, and the polyacetylene dehydromatricaria ester. Chemoecology 20, 255–264 (2010). https://doi.org/10.1007/s00049-010-0055-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-010-0055-1