Abstract
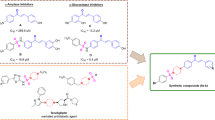
New series of barbiturates linked to aryl hydrazone derivatives 4a-n were designed and synthesized. Briefly, aniline derivatives in the presence of HBF4 and NaNO2 convert to aryl diazonium tetrafluoroborate which is further attacked to barbituric acid derivatives in water. Finally, with tautomerization, the desired products were achieved. Next, compounds were evaluated as possible urease inhibitors and all the synthesized compounds (IC50 = 8.43 ± 0.14–10.91 ± 0.42 μM) were more potent than standard inhibitors hydroxyurea (IC50 = 100.00 ± 0.15 μM) and thiourea (IC50 = 23 ± 1.7 μM) against urease. It was shown that 4-bromo substitution on the phenyl ring of barbiturate improved the inhibitory potency. Furthermore, based on the molecular dynamic studies, compound 4g depicted noticeable interaction with the urease active site and mobile flap residues through the barbituric acid moiety by coordinating toward the metal bi-nickel center and the essential residues at the active site flap-like Cys592, His593, His594, respectively. These interactions cause interfering catalytic activity of the active site and reduce the flexibility of the mobile flap at the entrance of the active site channel, which significantly decreases the ureolytic activity of urease.








Similar content being viewed by others
References
Blakeley RL, Zerner B. Jack bean urease: the first nickel enzyme. J Mol Catal. 1984;23:263–92.
Yang W, Feng Q, Peng Z, Wang G. An overview on the synthetic urease inhibitors with structure-activity relationship and molecular docking. Eur J Med Chem. 2022;234:114273.
Sadat-Ebrahimi SE, Bigdelou A, Sooreshjani RH, Montazer MN, Zomorodian K, Irajie C. et al. Novel phenylurea-pyridinium derivatives as potent urease inhibitors: Synthesis, in vitro, and in silico studies. J Mol Struct. 2022;1263:133078
Yang W, Feng Q, Peng Z, Wang G. An overview on the synthetic urease inhibitors with structure-activity relationship and molecular docking. Eur J Med Chem. 2022;234:114273.
Sohrabi M, Nazari Montazer M, Farid SM, Tanideh N, Dianatpour M, Moazzam A, et al. Design and synthesis of novel nitrothiazolacetamide conjugated to different thioquinazolinone derivatives as anti-urease agents. Sci Rep. 2022;12:1–14.
Jahantab MB, Safaripour AA, Hassanzadeh S, Yavari Barhaghtalab MJ. Demographic, chemical, and helicobacter pylori positivity assessment in different types of gallstones and the bile in a random sample of cholecystectomied iranian patients with cholelithiasis. Can J Gastroenterol Hepatol. 2021;2021:3351352.
Zahra U, Zaib S, Saeed A, Rehman MU, Shabir G, Alsaab HO, et al. New acetylphenol-based acyl thioureas broaden the scope of drug candidates for urease inhibition: synthesis, in vitro screening and in silico analysis. Int J Biol Macromol. 2022;198:157–67.
Sadat-Ebrahimi SE, Bigdelou A, Sooreshjani RH, Montazer MN, Zomorodian K, Irajie C, et al. Novel phenylurea-pyridinium derivatives as potent urease inhibitors: Synthesis, in vitro, and in silico studies. J Mol Struct. 2022;1263:133078.
Asadi M, Iraji A, Sherafati M, Nazari Montazer M, Ansari S, Mohammadi Khanaposhtani M, et al. Synthesis and in vitro urease inhibitory activity of 5-nitrofuran-2-yl-thiadiazole linked to different cyclohexyl-2-(phenylamino)acetamides, in silico and kinetic studies. Bioorgan Chem. 2022;120:105592.
Saeedian Moghadam E, Mohammed Al-Sadi A, Ghafarzadegan R, Talebi M, et al. Benzimidazole derivatives act as dual urease inhibitor and anti-helicobacter pylori agent; synthesis, bioactivity, and molecular docking study. Syn Commun. 2022;52:1–13.
Kafarski P, Talma M. Recent advances in design of new urease inhibitors: a review. J Adv Res. 2018;13:101–12.
Azizian H, Nabati F, Sharifi A, Siavoshi F, Mahdavi M, Amanlou M. Large-scale virtual screening for the identification of new Helicobacter pylori urease inhibitor scaffolds. J Mol Model. 2012;18:2917–27.
Biglar M, Mirzazadeh R, Asadi M, Sepehri S, Valizadeh Y, Sarrafi Y, et al. Novel N, N-dimethylbarbituric-pyridinium derivatives as potent urease inhibitors: synthesis, in vitro, and in silico studies. Bioorgan Chem. 2020;95:103529.
Kazemzadeh H, Hamidian E, Hosseini FS, Abdi M, Niasari Naslaji F, Talebi M, et al. Isoindolin-1-ones fused to barbiturates: from design and molecular docking to synthesis and urease inhibitory evaluation. ACS Omega. 2022;7:19401
Pedrood K, Azizian H, Montazer MN, Mohammadi‐Khanaposhtani M, Asgari MS, Asadi M, et al. Arylmethylene hydrazine derivatives containing 1,3-dimethylbarbituric moiety as novel urease inhibitors. Sci Rep. 2021;11:10607.
Asgari MS, Azizian H, Nazari Montazer M, Mohammadi‐Khanaposhtani M, Asadi M, Sepehri S, et al. New 1, 2, 3‐triazole–(thio) barbituric acid hybrids as urease inhibitors: design, synthesis, in vitro urease inhibition, docking study, and molecular dynamic simulation. Arch Pharm. 2020;353:2000023.
Arqué X, Romero-Rivera A, Feixas F, Patiño T, Osuna S, Sánchez S. Intrinsic enzymatic properties modulate the self-propulsion of micromotors. Nat Commun. 2019;10:1–12.
Azizian H, Mousavi Z, Faraji H, Tajik M, Bagherzadeh K, Bayat P, et al. Arylhydrazone derivatives of naproxen as new analgesic and anti-inflammatory agents: Design, synthesis and molecular docking studies. J Mol Graph Model. 2016;67:127–36.
Svane S, Sigurdarson JJ, Finkenwirth F, Eitinger T, Karring H. Inhibition of urease activity by different compounds provides insight into the modulation and association of bacterial nickel import and ureolysis. Sci Rep. 2020;10:1–14.
Pedrood K, Azizian H, Montazer MN, Mohammadi‐Khanaposhtani M, Asgari MS, Asadi M, et al. Arylmethylene hydrazine derivatives containing 1, 3-dimethylbarbituric moiety as novel urease inhibitors. Sci Rep. 2021;11:1–16.
Sedaghati S, Azizian H, Montazer MN, Mohammadi-Khanaposhtani M, Asadi M, Moradkhani F, et al. Novel (thio) barbituric-phenoxy-N-phenylacetamide derivatives as potent urease inhibitors: synthesis, in vitro urease inhibition, and in silico evaluations. Struct Chem. 2021;32:37–48.
Asgari MS, Azizian H, Nazari Montazer M, Mohammadi-Khanaposhtani M, Asadi M, Sepehri S. New 1,2,3-triazole-(thio)barbituric acid hybrids as urease inhibitors: design, synthesis, in vitro urease inhibition, docking study, and molecular dynamic simulation. Arch Pharm. 2020;353:e2000023.
Sohrabi M, Nazari Montazer M, Farid SM, Tanideh N, Dianatpour M, Moazzam A, et al. Design and synthesis of novel nitrothiazolacetamide conjugated to different thioquinazolinone derivatives as anti-urease agents. Sci Rep. 2022;12:2003.
Azizian H, Esmailnejad A, Fathi Vavsari V, Mahernia S, Amanlou M, Balalaie S. Pantoprazole derivatives: synthesis, urease inhibition assay and in silico molecular modeling studies. ChemistrySelect. 2020;5:4580–7.
Pedrood K, Azizian H, Montazer MN, Moazzam A, Asadi M, Montazeri H, et al. Design and synthesis of new N-thioacylated ciprofloxacin derivatives as urease inhibitors with potential antibacterial activity. Sci Rep. 2022;12:13827.
Tahmasebi B, Iraji A, Sherafati M, Moazzam A, Akhlagh SA, Adib M, et al. Structure-based drug discovery and antimicrobial activity of ciprofloxacin-grafted Ugi adducts. J Biomol Struct Dyn. 2022;10:1–10.
Iraji A, Yazdanpanah S, Alizadeh F, Mirzamohammadi S, Ghasemi Y, Pakshir K, et al. Screening the antifungal activities of monoterpenes and their isomers against Candida species. J Appl Microbiol. 2020;129:1541–51.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mollazadeh, M., Azizian, H., Fakhrioliaei, A. et al. Different barbiturate derivatives linked to aryl hydrazone moieties as urease inhibitors; design, synthesis, urease inhibitory evaluations, and molecular dynamic simulations. Med Chem Res 32, 930–943 (2023). https://doi.org/10.1007/s00044-023-03050-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-023-03050-w