Abstract
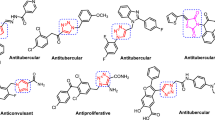
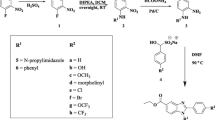
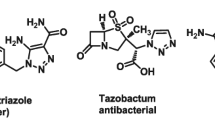
Herein, we describe synthesis and antitubercular evaluation of novel-substituted 1,2,3-triazolylmethyl carbazoles rationally designed by incorporating two pharmacophoric heterocycles, viz. carbazole and triazole in one molecular frame. The new compounds 4a–z and 4aa–ac were obtained in very good yields using copper-catalysed Huisgen [3 + 2] cycloaddition reaction of 9-propynyl-9H-carbazole (2) with various aryl/alkyl azides 3a–z and 3aa–ac. Evaluation of all 29 new analogues for in vitro antimycobacterial activity against Mycobacterium tuberculosis H37Rv (ATCC 27294) resulted two compounds 4x and 4z (MIC: 6.25 μg/mL) as most promising antitubercular agents with low cytotoxicity.






Similar content being viewed by others
References
Abubakar I, Zignol M, Falzon D, Raviglione M, Ditiu L, Masham S, Adetifa I, Ford N, Cox H, Lawn SD, Marais BJ, McHugh TD, Mwaba P, Bates M, Lipman M, Zijenah L, Logan S, McNerney R, Zumla A, Sarda K, Nahid P, Hoelscher M, Pletschette M, Memish ZA, Kim P, Hafner R, Cole S, Migliori GB, Maeurer M, Schito M, Zumla A (2013) Drug-resistant tuberculosis: time for visionary political leadership. Lancet Infect Dis 13:529–539
Addla D, Jallapally A, Gurram D, Yogeeswari P, Sriram D, Kantevari S (2014) Rational design, synthesis and antitubercular evaluation of novel 2-(trifluoromethyl)phenothiazine-[1,2,3]triazole hybrids. Bioorg Med Chem Lett 24:233–236
Agalave SG, Maujan SR, Pore VS (2011) Click chemistry: 1,2,3-triazoles as pharmacophores. Chem Asian J 6:2696–2718
Berg R, Straub BF (2013) Advancements in the mechanistic understanding of the copper-catalyzed azide–alkyne cycloaddition. Beilstein J Org Chem 9:2715–2750
Boechat N, Ferreira VF, Ferreira SB, de Lourdes G, Ferreira M, de da Silva CF, Bastos MM, Dos S, Costa M, Lourenço MC, Pinto AC, Krettli AU, Aguiar AC, Teixeira BM, da Silva NV, Martins PR, Bezerra FA, Camilo AL, da Silva GP, Costa CC (2011) Novel 1,2,3-triazole derivatives for use against Mycobacterium tuberculosis H37Rv (ATCC 27294) strain. J Med Chem 54:5988–5999
Choi TA, Czerwonka R, Fröhner W, Krahl MP, Reddy KR, Franzblau SG, Knölker HJ (2006) Synthesis and activity of carbazole derivatives against Mycobacterium tuberculosis. Chem Med Chem 1:812–815
Daley CL, Caminero JA (2013) Management of multidrug resistant tuberculosis. Semin Respir Crit Care Med 34:44–59
Dash M (2013) Drug resistant tuberculosis: a diagnostic challenge. Postgrad Med 59:196–202
García A, Bocanegra-García V, Palma-Nicolás JP, Rivera G (2012) Recent advances in antitubercular natural products. Eur J Med Chem 49:1–23
Ghodbane R, Drancourt MR (2013) Non-human sources of Mycobacterium tuberculosis. Tuberculosis 93:589–595
Gonzaga DT, da Rocha DR, de da Silva CF, Ferreira VF (2013) Recent advances in the synthesis of new antimycobacterial agents based on the 1H–1,2,3-triazoles. Curr Top Med Chem 13:2850–2865
Hai-Bin Z, Guan-Sai L, Zhu-Jun Y (2007) Short and efficient total synthesis of luotonin A and 22-hydroxyacuminatine using a common cascade strategy. J Org Chem 72:6270–6272
Isaakidis P, Rangan S, Pradhan A, Ladomirska J, Reid T, Kielmann K (2013) ‘I cry every day’: experiences of patients co-infected with HIV and multidrug-resistant tuberculosis. Trop Med Int Health 18:1128–1133
Kantevari S, Yempala T, Surineni G, Sridhar B, Yogeeswari P, Sriram D (2011) Synthesis and antitubercular evaluation of novel dibenzo[b, d]furan and 9-methyl-9H-carbazole derived hexahydro-2H-pyrano[3,2-c] quinolines via povarov reaction. Eur J Med Chem 46:4827–4833
Kharaneko OI, Shishkina SV, Shishkin OV, Kibalnyi AV, Dulenko VI (2004) Concerting the product of [2 + 2] cyclodimerization of 9-allenyl carbazole. Chem Heterocycl Comp 40:1427–1434
Knölker HJ, Reddy KR (2008) Chemistry and biology of carbazole alkaloids. Alkaloids Chem Biol 65:1–410
Kolb HC, Sharpless KB (2003) The growing impact of click chemistry on drug discovery. Drug Discov Today 8:1128–1137
Kumar A, Kumar AM, Gupta D, Kanchar A, Mohammed S, Srinath S, Tripathy S, Rajasekaran S, Chan PL, Swaminathan S, Dewan PK (2012) Global guidelines for treatment of tuberculosis among persons living with HIV: unresolved issues. Int J Tuberc Lung Dis 16:573–578
Lawn SD, Mwaba P, Bates M, Piatek A, Alexander H, Marais BJ, Cuevas LE, McHugh TD, Zijenah L, Kapata N, Abubakar I, McNerney R, Hoelscher M, Memish ZA, Migliori GB, Kim P, Maeurer M, Schito M, Zumla A (2013) Advances in tuberculosis diagnostics: the Xpert MTB/RIF assay and future prospects for a point-of-care test. Lancet Infect Dis 13:349–361
Liu X, Chen C, He W, Huang P, Liu M, Wang Q, Guo H, Bolla K, Lu Y, Song F, Dai H, Liu M, Zhang L (2012) Exploring anti-TB leads from natural products library originated from marine microbes and medicinal plants. Antonie Van Leeuwenhoek 102:447–461
Ma C, Case RJ, Wang Y, Zhang HJ, Tan GT, Van Hung N, Cuong NM, Franzblau SG, Soejarto DD, Fong HH, Pauli GF (2005) Anti-tuberculosis constituents from the stem bark of Micromelum hirsutum. Planta Med 71:261–267
Nathan C (2014) Drug-resistant tuberculosis: a new shot on goal. Nat Med 20:121–123
Ngo LT, Okogun JI, Folk WR (2013) 21st Century natural product research and drug development and traditional medicines. Nat Prod Rep 30:584–592
Okunade AL, Elvin-Lewis MP, Lewis WH (2004) Natural antimycobacterial metabolites: current status. Phyto chemistry 65:1017–1032
Patpi SR, Pulipati L, Yogeeswari P, Sriram D, Jain N, Sridhar B, Murthy R, Anjana Devi T, Kalivendi SV, Kantevari S (2012) Design, synthesis and structure–activity correlations of novel dibenzo[b, d]furan, dibenzo[b, d]thiophene and N-methyl carbazole clubbed 1,2,3-triazoles as potent inhibitors of Mycobacterium tuberculosis. J Med Chem 55:3911–3922
Putapatri SR, Kanwal A, Banerjee SK, Kantevari S (2014) Synthesis of novel l-rhamnose derived acyclic C-nucleosides with substituted 1,2,3-triazole core as potent sodium-glucose co-transporter (SGLT) inhibitor. Bioorg Med Chem Lett 24:1528–1531
Salomon CE, Schmidt LE (2012) Natural products as leads for tuberculosis drug development. Curr Top Med Chem 12:735–765
Schmidt AW, Reddy KR, Knölker HJ (2012) Occurrence, biogenesis, and synthesis of biologically active carbazole alkaloids. Chem Rev 112:3193–3228
Swaminathan S, Narendran G, Venkatesan P, Iliayas S, Santhanakrishnan R, Menon PA, Padmapriyadarsini C, Ramachandran R, Chinnaiyan P, Suhadev M, Sakthivel R, Narayanan PR (2010) Efficacy of a 6 month versus 9 month intermittent treatment regimen in HIV-infected patients with tuberculosis: a randomized clinical trial. Am J Respir Crit Care Med 181:743–751
Thirumurugan P, Matosiuk D, Jozwiak K (2013) Click chemistry for drug development and diverse chemical-biology applications. Chem Rev 113:4905–4979
Vainio MJ, Kogej T, Raubacher F, Sadowski J (2013) Scaffold hopping by fragment replacement. J Chem Inf Mod 53:1825–1835
Yempala T, Sridevi JP, Yogeeswari P, Sriram D, Kantevari S (2013) Design, synthesis and antitubercular evaluation of novel 2-substituted-3H-benzofuro benzofurans via palladium-copper catalysed Sonagashira coupling reaction. Bioorg Med Chem Lett 23:5393–5936
Yempala T, Sridevi JP, Yogeeswari P, Sriram D, Kantevari S (2014) Rational design and synthesis of novel dibenzo[b, d]furan-1,2,3-triazole conjugates as potent inhibitors of Mycobacterium tuberculosis. Eur J Med Chem 71:160–167
Zhou B, He Y, Zhang X, Xu J, Luo Y, Wang Y, Franzblau SG, Yang Z, Chan RJ, Liu Y, Zheng J, Zhang ZY (2010) Targeting mycobacterium protein tyrosine phosphatase B for antituberculosis agents. Proc Natl Aca Sci 107:4573–4578
Zumla A, George A, Sharma V, Herbert N, Baroness MI (2013a) WHO’s 2013 global report on tuberculosis: successes, threats, and opportunities. Lancet 382:1765–1767
Zumla A, Raviglione M, Hafner R, von Reyn CF (2013b) Tuberculosis. New Engl J Med 368:745–755
Acknowledgments
Authors are thankful to Dr. Lakshmi Kantham, Director and Dr. V. Jayathirtha Rao, Head, Crop Protection Chemicals Division, CSIR-IICT, Hyderabad, INDIA for their continuous encouragement, support and financial assistance through CSIR-12th FYP Projects (ORIGIN, CSC0108; DENOVA, CSC0205 and INTELCOAT, CSC0114). GS is thankful to UGC for Granting SRF.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Surineni, G., Yogeeswari, P., Sriram, D. et al. Rational design, synthesis and evaluation of novel-substituted 1,2,3-triazolylmethyl carbazoles as potent inhibitors of Mycobacterium tuberculosis . Med Chem Res 24, 1298–1309 (2015). https://doi.org/10.1007/s00044-014-1210-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-1210-y