Abstract
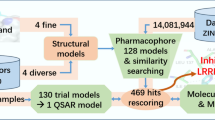
A mutation in the gene, encoding leucine rich repeat kinase 2 (LRRK2), is a genetic cause of Parkinson’s disease (PD). LRRK2 is a dimeric multidomain protein, largely regulates guanosine triphosphate (GTP). G2019S and I2020T, the mutation encodes in the kinase domain of LRRK2 increase the GTPase activity, are the important regulators in pathogenesis of PD. To design potent LRRK2 inhibitors, pharmacophore modeling approach was employed with a wide chemical diversity of compound’s database. The best hypothesis consists of hydrogen-bond acceptor and donor as well as hydrophobic aliphatic and ring aromatic features. The model was validated by the test and decoy sets followed by Fischer’s randomization test. The validated model was used to screen the database of compounds, which were designed through de novo approach. Homology model of the kinase domain of LRRK2 was built initially using the crystal structure of Janus kinase 3. The designed molecules were further screened for ADMET properties, and ligand–receptor interaction of top hits was analyzed by molecular docking studies to explore potent LRRK2 inhibitors.





Similar content being viewed by others
References
Anand VS, Reichling LJ, Lipinski K, Stochaj W, Duan W, Kelleher K, Pungaliya P, Brown EL, Reinhart PH, Somberg R (2009) Investigation of leucine rich repeat kinase 2. FEBS J 276(2):466–478. doi:10.1111/j.1742-4658.2008.06789.x
Baker-Glenn C, Burdick DJ, Chambers M, Chan BK, Chen H, Estrada A, Gunzner JL, Shore D, Sweeney ZK, Wang S (2011a) Aminopyrimidine derivatives as LRRK2 modulators. WO Patent App. PCT/EP2011/059009
Baker-Glenn C, Burdick DJ, Chambers M, Chan BK, Chen H, Estrada A, Sweeney ZK (2011b) Pyrazole aminopyrimidine derivatives as LRRK2 modulators. WO Patent App. PCT/EP2011/069696
Cereto-Massagué A, Guasch L, Valls C, Mulero M, Pujadas G, Garcia-Vallvé S (2012) DecoyFinder: an easy-to-use python GUI application for building target-specific decoy sets. Bioinformatics 28(12):1661–1662. doi:10.1093/bioinformatics/bts249
Chan B, Estrada A, Sweeney Z, Mciver EG (2011) Pyrazolopyridines as inhibitors of the kinase LRRK2. WO Patent App. PCT/GB2011/050937
Chan BK, Estrada AA, Chen H, Atherall J, Baker-Glenn C, Beresford A, Burdick DJ, Chambers M, Dominguez SL, Drummond J (2012) Discovery of a highly selective, brain-penetrant aminopyrazole LRRK2 inhibitor. ACS Med Chem Lett 4(1):85–90. doi:10.1021/ml3003007
Chen H, Chan BK, Drummond J, Estrada AA, Gunzner Toste J, Liu X, Liu Y, Moffat J, Shore D, Sweeney ZK (2012) Discovery of selective LRRK2 inhibitors guided by computational analysis and molecular modeling. J Med Chem 55(11):5536–5545. doi:10.1021/jm300452p
Choi HG, Zhang J, Deng X, Hatcher JM, Patricelli MP, Zhao Z, Alessi DR, Gray NS (2012) Brain penetrant LRRK2 inhibitor. ACS Med Chem Lett 3(8):658–662. doi:10.1021/ml300123a
Chrencik JE, Patny A, Leung IK, Korniski B, Emmons TL, Hall T, Weinberg RA, Gormley JA, Williams JM, Day JE (2010) Structural and thermodynamic characterization of the TYK2 and JAK3 kinase domains in complex with CP-690550 and CMP-6. J Mol Biol 400(3):413–433. doi:10.1016/j.jmb.2010.05.020
Cole C, Barber JD, Barton GJ (2008) The Jpred 3 secondary structure prediction server. Nucleic Acids Res 36(suppl 2):W197–W201. doi:10.1093/nar/gkn238
Colovos C, Yeates TO (1993) Verification of protein structures: patterns of nonbonded atomic interactions. Protein Sci 2(9):1511–1519. doi:10.1002/pro.5560020916
Cookson MR (2010) The role of leucine-rich repeat kinase 2 (LRRK2) in Parkinson’s disease. Nat Rev Neurosci 11(12):791–797. doi:10.1038/nrn2935
Dachsel JC, Farrer MJ (2010) LRRK2 and Parkinson disease. Arch Neurol 67(5):542. doi:10.1001/archneurol.2010.79
Deng X, Dzamko N, Prescott A, Davies P, Liu Q, Yang Q, Lee JD, Patricelli MP, Nomanbhoy TK, Alessi DR (2011) Characterization of a selective inhibitor of the Parkinson’s disease kinase LRRK2. Nat Chem Biol 7(4):203–205. doi:10.1038/nchembio.538
Deng X, Choi HG, Buhrlage SJ, Gray NS (2012) Leucine-rich repeat kinase 2 inhibitors: a patent review (2006–2011). Expert Opin Ther Pat 22(12):1415–1426. doi:10.1517/13543776.2012.729041
Dexter DT, Jenner P (2013) Parkinson’s disease: from pathology to molecular disease mechanisms. Free Radic Biol Med. 62:132–144. doi:10.1016/j.freeradbiomed.2013.01.018
Dhoke GV, Gangwal RP, Sangamwar AT (2012) A combined ligand and structure based approach to design potent PPAR-alpha agonists. J Mol Struct 1028:22–30. doi:10.1016/j.molstruc.2012.06.032
Discovery studio 2.5 (2009) Accelrys Inc., San Diego
Enslein K, Gombar VK, Blake BW (1994) Use of SAR in computer-assisted prediction of carcinogenicity and mutagenicity of chemicals by the TOPKAT program. Mutat Res 305(1):47–61. doi:10.1016/0027-5107(94)90125-2
Estrada AA, Liu X, Baker-Glenn C, Beresford A, Burdick DJ, Chambers M, Chan BK, Chen H, Ding X, DiPasquale AG (2012) Discovery of highly potent, selective, and brain-penetrable leucine-rich repeat kinase 2 (LRRK2) small molecule inhibitors. J Med Chem 55(22):9416–9433. doi:10.1021/jm301020q
Eswar N, Eramian D, Webb B, Shen MY, Sali A (2008) Protein structure modeling with MODELLER. Structural Proteomics. Springer, London, pp 145–159. doi:10.1007/978-1-60327-058-8_8
Gandhi PN, Chen SG, Wilson-Delfosse AL (2009) Leucine rich repeat kinase 2 (LRRK2): a key player in the pathogenesis of Parkinson’s disease. J Neurosci Res 87(6):1283–1295. doi:10.1002/jnr.21949
Gilsbach BK, Ho FY, Vetter IR, van Haastert PJ, Wittinghofer A, Kortholt A (2012) Roco kinase structures give insights into the mechanism of Parkinson disease-related leucine-rich-repeat kinase 2 mutations. Proc Natl Acad Sci USA 109(26):10322–10327. doi:10.1073/pnas.1203223109
Glide 5.5 (2009) Schrödinger. LLC, New York
Gloeckner CJ, Kinkl N, Schumacher A, Braun RJ, O’Neill E, Meitinger T, Kolch W, Prokisch H, Ueffing M (2006) The Parkinson disease causing LRRK2 mutation I2020T is associated with increased kinase activity. Hum Mol Genet 15(2):223–232. doi:10.1093/hmg/ddi439
Greggio E, Cookson MR (2009) Leucine-rich repeat kinase 2 mutations and Parkinson’s disease: three questions. ASN Neuro 1(1):13–24. doi:10.1042/AN20090007
Humphrey W, Dalke A, Schulten K (1996) VMD: visual molecular dynamics. J Mol Graph 14(1):33–38. doi:10.1016/0263-7855(96)00018-5
Kare P, Bhat J, Sobhia ME (2013) Structure-based design and analysis of MAO-B inhibitors for Parkinson’s disease: using in silico approaches. Mol Divers 17(1):111–122. doi:10.1007/s11030-012-9420-z
Kristam R, Gillet VJ, Lewis RA, Thorner D (2005) Comparison of conformational analysis techniques to generate pharmacophore hypotheses using catalyst. J Chem Inf Model 45(2):461–476. doi:10.1021/ci049731z
Larkin M, Blackshields G, Brown N, Chenna R, McGettigan P, McWilliam H, Valentin F, Wallace I, Wilm A, Lopez R (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23(21):2947–2948. doi:10.1093/bioinformatics/btm404
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystallogr 26(2):283–291. doi:10.1107/S0021889892009944
Lee BD, Dawson VL, Dawson TM (2012) Leucine-rich repeat kinase 2 (LRRK2) as a potential therapeutic target in Parkinson’s disease. Trends Pharmacol Sci 33(7):365–373. doi:10.1016/j.tips.2012.04.001
Lewis PA (2009) The function of ROCO proteins in health and disease. Biol Cell 101(3):183–191. doi:10.1042/BC20080053
LigPrep 2.3 (2009) Schrödinger. LLC, New York
Protein Preparation Wizard (2009) Schrödinger. LLC, New York
PyMOL 1.3 (2010) Schrödinger. LLC, New York
QikProp 3.2 (2009) Schrödinger. LLC, New York
Ramachandran G, Ramakrishnan C, Sasisekharan V (1963) Stereochemistry of polypeptide chain configurations. J Mol Biol 7:95. doi:10.1016/S0022-2836(63)80023-6
Reith AD, Bamborough P, Jandu K, Andreotti D, Mensah L, Dossang P, Choi HG, Deng X, Zhang J, Alessi DR (2012) GSK2578215A; a potent and highly selective 2-arylmethyloxy-5-substituent-N-arylbenzamide LRRK2 kinase inhibitor. Bioorg Med Chem Lett 22(17):5625–5629. doi:10.1016/j.bmcl.2012.06.104
Rudenko IN, Chia R, Cookson MR (2012) Is inhibition of kinase activity the only therapeutic strategy for LRRK2-associated Parkinson’s disease? BMC Med 10(1):20. doi:10.1186/1741-7015-10-20
Singh R, Balupuri A, Sobhia ME (2013a) Development of 3D-pharmacophore model followed by successive virtual screening, molecular docking and ADME studies for the design of potent CCR2 antagonists for inflammation-driven diseases. Mol Simul 39(1):49–58. doi:10.1080/08927022.2012.701743
Singh U, Gangwal RP, Prajapati R, Dhoke GV, Sangamwar AT (2013b) 3D QSAR pharmacophore-based virtual screening and molecular docking studies to identify novel matrix metalloproteinase 12 inhibitors. Mol Simul 39(5):385–396. doi:10.1080/08927022.2012.731506
Smellie A, Teig SL, Towbin P (1995) Poling: promoting conformational variation. J Comput Chem 16(2):171–187. doi:10.1002/jcc.540160205
SYBYL 7.1 (2005) Tripose Inc., St. Louis
Tsika E, Moore DJ (2012) Mechanisms of LRRK2-mediated neurodegeneration. Curr Neurol Neurosci Rep 12(3):251–260. doi:10.1007/s11910-012-0265-8
Yuan Y, Pei J, Lai L (2011) LigBuilder 2: a practical de novo drug design approach. J Chem Inf Model 51(5):1083–1091. doi:10.1021/ci100350u
Zhang J, Deng X, Choi HG, Alessi DR, Gray NS (2012) Characterization of TAE684 as a potent LRRK2 kinase inhibitor. Bioorg Med Chem Lett 22(5):1864–1869. doi:10.1016/j.bmcl.2012.01.084
Acknowledgments
The authors would like to thank the University Grant Commission (UGC), New Delhi, India for financial assistance of the project.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bhayye, S.S., Roy, K. & Saha, A. Exploring structural requirement, pharmacophore modeling, and de novo design of LRRK2 inhibitors using homology modeling approach. Med Chem Res 23, 3705–3713 (2014). https://doi.org/10.1007/s00044-014-0955-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-0955-7