Abstract
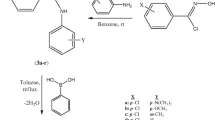
A series of 4-amino-3-hydroxy-naphthalene-1-sulfonic acid derivatives (1–18) was synthesized and tested in vitro for its anti-microbial potential. The results of anti-microbial studies indicated that derivatives having 3,4,5 trimethoxy (6) and 2,4 dichloro (17) groups on benzylidene amino portion were found to be most effective ones. The mt-QSAR model for anti-microbial activity revealed the importance of topological parameter, valence zero-order molecular connectivity index (0χv) in describing the anti-microbial activity of synthesized 4-amino-3-hydroxy-naphthalene-1-sulfonic acid derivatives.



Similar content being viewed by others
References
Abdel-Gawad SM, El-Gaby MSA, Heiba HI, Aly HM, Ghorab MM (2005) Synthesis and radiation stability of some new biologically active hydroquinoline and pyrimido[4,5-b]quinoline derivatives. J Chin Chem Soc 52:1227–1236
Amir M, Javed SA, Hassan MZ (2011) Synthesis and antimicrobial activity of pyrazolinones and pyrazoles having benzothiazole moiety. Med Chem Res. doi:10.1007/s00044-011-9642-0 (in press)
Bajaj S, Sambi SS, Madan AK (2005) Prediction of anti-inflammatory activity of N-arylanthranilic acids: computational approach using refined Zagreb indices. Croat Chem Acta 78(2):165–174
Balaban AT (1982) Highly discriminating distance based topological indices. Chem Phys Lett 89:399–404
Cappucino JG, Sherman N (1999) Microbiology—a laboratory manual. Addison Wesley, California, p 263
Chugh TD (2008) Emerging and re-emerging bacterial diseases in India. J Biosci 33(4):549–555
Cruz-Monteagudo M, Gonzalez-Diaz H, Aguero-Chapin G, Santana L, Borges F, Dominguez ER, Podda G, Uriarte E (2007) Computational chemistry development of a unified free energy Markov model for the distribution of 1300 chemicals to 38 different environmental or biological systems. J Comput Chem 28(11):1909–1923
Ebenezer GJ, Norman G, Joseph A, Daniel S, Job CK (2002) Drug resistant Mycobacterium leprae—results of mouse footpad studies from a laboratory in south India. Indian J Lepr 74:301–312
El-Saghier AMM, Naili MB, Rammash BK, Saleh NA, Kreddanc KM (2007) Synthesis and antibacterial activity of some new fused chromenes. ARKIVOK 13:83–91
Emami S, Falahati M, Banifafemi A, Shafiee A (2004) Stereoselective synthesis and antifungal activity of (Z)-trans-3-azolyl-2-methylchromanone oxime ethers. Bioorgan Med Chem 12:5881–5889
Furniss BS, Hannaford AJ, Smith PWG, Tatchell AR (1998) Vogel’s text book of practical organic chemistry. Addison Wesley Longman Inc., California, p 34
Furusjo E, Svenson A, Rahmberg M, Andersson M (2006) The importance of outlier detection and training set selection for reliable environmental QSAR predictions. Chemosphere 63:99–108
Golbraikh A, Tropsha A (2002) Beware of q 2! J Mol Graphics Model 20:269–276
Gonzalez-Diaz H, Prado–Prado FJ (2008) Unified QSAR and network-based computational chemistry approach to antimicrobials, part 1: multispecies activity models for antifungals. J Comput Chem 29(4):656–667
Gonzalez-Diaz H, Vilar S, Santana L, Uriarte E (2007) Medicinal chemistry and bioinformatics-current trends in drugs discovery with networks topological indices. Curr Top Med Chem 7(10):1015–1029
Gonzalez-Diaz H, Gonzalez-Diaz Y, Santana L, Ubeira FM, Uriarte E (2008) Networks and connectivity indices. Proteomics 8(4):750–778
Hansch C, Fujita T (1964) p-σ-π Analysis. A method for the correlation of biological activity and chemical structure. J Am Chem Soc 86:1616–1626
Hansch C, Leo A, Unger SH, Kim KH, Nikaitani D, Lien EJ (1973) “Aromatic” substituent constants for structure-activity correlations. J Med Chem 16(11):1207–1216
Hussain Z, Khan KM, Perveen S, Nawaz Y, Bukhari IH (2011) Antifungal activity of the pyrolyzate of glucose, sucrose and starch in comparison to paper pyrolyzate. J Chem Soc Pak 33(5):694–697
Hyperchem 6.0 (1993) Hypercube Inc., Florida
Ibrahim MA, Sharif SAI, El-Tajory AN, Elamari AA (2011) Synthesis and antibacterial activities of some schiff bases. E-J Chem 8(1):212–216
Jha TK (2006) Drug unresponsiveness and combination therapy for Kala Azar. Indian J Med Res 123:389–398
Judge V, Narasimhan B, Ahuja M, Sriram D, Yogeeswari P, Clercq ED, Pannecouque C, Balzarini J (2011a) Synthesis, antimycobacterial, antiviral, antimicrobial activity and QSAR studies of isonicotinic acid-1-(substituted phenyl)-ethylidene/cycloheptylidene hydrazides. Med Chem Res. doi:10.1007/s00044-011-9705-2 (in press)
Judge V, Narasimhan B, Ahuja M, Sriram D, Yogeeswari P, Clercq ED, Pannecouque C, Balzarini J (2011b) Isonicotinic acid hydrazide derivatives: synthesis, antimicrobial activity and QSAR studies. Med Chem Res. doi:10.1007/s00044-011-9662-9 (in press)
Khan KM, Mughala UR, Khana S, Perveen S, Choudhary MI (2009) Synthesis and antibacterial and antifungal activity of 5-substituted imidazolones. Lett Drug Des Discov 6:69–77
Kier LB, Hall LH (1999) In: Devillers J, Balaban AT (eds) Topological indices and related descriptors in QSAR and QSPR. Gordon and Breach Sci. Pub, Amsterdam, pp 455–489
Kumar A, Narasimhan B, Kumar D (2007) Synthesis, antimicrobial, and QSAR studies of substituted benzamides. Bioorgan Med Chem 15:4113–4124
Kumar D, Judge V, Narang R, Sangwan S, Cercq ED, Balzarini J, Narasimhan B (2010a) Benzylidene/2-chlorobenzylidene hydrazides: synthesis, antimicrobial activity, QSAR studies and antiviral evaluation. Eur J of Med Chem 45:2806–2816
Kumar P, Narasimhan B, Yogeswari P, Sriram D (2010b) Synthesis and antitubercular activities of substituted benzoic acid N’-(substituted benzylidene/furan-2-ylmethylene)-N-(pyridine-3-carbonyl)-hydrazides. Eur J of Med Chem 45:6085–6089
Kumar D, Narang A, Judge V, Kumar D, Narasimhan B (2011) Antimicrobial evaluation of 4-methylsulfanyl benzylidene/3-hydroxy benzylidene hydrazides and QSAR studies. Med Chem Res. doi:10.1007/s00044-010-9543-7 (in press)
Laxmi SV, Rajitha B (2011) Synthesis and antimicrobial activity of newer indole semicarbazones. Med Chem Res. doi:10.1007/s00044-010-9502-3 (in press)
Mahiwal K, Kumar P, Narasimhan B (2011) Synthesis, antimicrobial evaluation, ot-QSAR and mt-QSAR studies of 2-amino benzoic acid derivatives. Med Chem Res. doi:10.1007/s00044-010-9537-5 (in press)
Marzio WD, Saenz ME (2004) Quantitative structure–activity relationship for aromatic hydrocarbons on freshwater fish. Ecotoxicol Environ Saf 59:256–262
Metwally KA, Abdel-Aziz LM, Lashine EM, Husseiny MI, Badawy RH (2006) Hydrazones of 2-aryl-quinoline-4-carboxylic acid hydrazides: synthesis and preliminary evaluation as antimicrobial agents. Bioorgan Med Chem 14:8675–8682
Mohanty S, Patel DK, Pati SS, Mishra SK (2006) Adjuvant therapy in cerebral malaria. Indian J Med Res 124:245–260
Narang R, Narasimhan B, Sharma S, Sriram D, Yogeeswari P, Clercq ED, Pannecouque C, Balzarini J (2011a) Synthesis, antimycobacterial, antiviral, antimicrobial activity and QSAR studies of nicotinic acid benzylidene hydrazide derivatives. Med Chem Res. doi:10.1007/s00044-011-9664-7 (in press)
Narang R, Narasimhan B, Sharma S, Sriram D, Yogeeswari P, Clercq ED, Pannecouque C, Balzarini J (2011b) Nicotinic acid benzylidene/phenyl-ethylidene hydrazides: synthesis and antimicrobial evaluation and QSAR studies. Lett Drug Des Discov 8:733–749
Narasimhan B, Judge V, Narang R, Ohlan S, Ohlan R (2007) Quantitative structure–activity relationship studies for prediction of antimicrobial activity of synthesized 2,4-hexadienoic acid derivatives. Bioorg Med Chem Lett 17:5836–5845
Perveen S, Fatima N, Khan KM, Khan A, Ali M, Choudhary MI (2010) Synthesis of carbamate derivatives of biological interest. J Chem Soc Pak 32(3):338–343
Pharmacopoeia of India (2007) Controller of Publications, Ministry of Health Department, Govt. of India, New Delhi, vol. I: 37
Rajasekaran A (2007) Synthesis, anti-nociceptive, anti-inflammatory and anti-epileptic evaluation of some novel indeno[1,2-b]quinoxalin-11-ylidenamines. Iranian J Pharm Sci 3(4):251–262
Rajasekaran A, Rajamanickram V, Darlinquine S (2011) Synthesis, anti-inflammatory and analgesic evaluation of thioxoquinazolinone derivatives. Yakugaku Zasshi 131(7):1079–1084
Randic M (1975) On characterization of molecular branching. J Am Chem Soc 97:6609–6615
Randic M (1993) Comparative regression analysis: regression based on a single descriptor. Croat Chem Acta 66:289–312
Ray K, Bala M, Gupta SM, Khunger N, Puri P, Muralidhar S, Kumar J (2006) Changing trends in sexually transmitted infections at a regional STD centre in north India. Indian J Med Res 124:559–568
Rodriguez-Arguelles MC, Lopez-Silva EC, Sanmartin J, Pelagatti P, Zani F (2005) Copper complexes of imidazole-2-, pyrrole-2- and indol-3-carbaldehyde thiosemicarbazones: inhibitory activity against fungi and bacteria. J Inorg Biochem 99:2231–2239
Rosu T, Negoiu M, Pasculescu S, Pahontu E, Poirier D, Gulea A (2010) Metal-based biologically active agents: synthesis, characterization, antibacterial and antileukemia activity evaluation of Cu(II), V(IV) and Ni(II) complexes with antipyrine-derived compounds. Eur J Med Chem 45:774–789
Sharma NC, Mandal PK, Dhillon R, Jain M (2007) Changing profile of Vibrio cholerae 01, 0139 in Delhi and periphery (2003–2005). Indian J Med Res 125:633–640
Singhal S, Purnapatre KP, Kalia V, Dube S, Nair D, Deb M, Agarwal P, Gupta S (2007) Ciprofloxacin-resistant Neisseria meningitidis, Delhi, India. Emerg Infect Dis 13:1614–1616
SPSS for Windows (1999) version 10.05, SPSS Inc., Bangalore
TSAR 3D Version 3.3 (2000) Oxford Molecular Limited, Oxford
Wiener HJ (1947) Structural determination of paraffin boiling points. J Am Chem Soc 69:17–20
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, R., Kumar, P., Kumar, M. et al. Synthesis, anti-microbial evaluation, and QSAR studies of 4-amino-3-hydroxy-naphthalene-1-sulfonic acid derivatives. Med Chem Res 21, 4301–4310 (2012). https://doi.org/10.1007/s00044-011-9954-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-011-9954-0



