Abstract
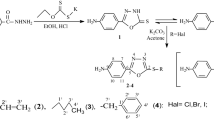
A series of some novel Ethyl 2-((1-hydroxynaphthalen-2-yl)methyleneamino)-5,6-dihydro-4H-cyclopenta[b]thiopehene-3-carboxylate, Ethyl 2-((1-hydroxynaphthalen-2-yl)methyleneamino)-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylate, Ethyl 2-((1-hydroxynaphtalen-2-yl)methyleneamino)-5,6,7,8-tetrahydro-4H-cyclohepta[b]thiophene-3-carboxylate and their Cr(III) and Zn(II) complexes have been synthesized. All of these substances have been examined for antibacterial activity against pathogenic strains Listeria monocytogenes 4b (ATCC-19115), Staphylococcus aureus (ATCC25923), Proteus OX2 Wrah (ETS.40-A-4), Escherichia coli (ATCC-1280), Salmonella typhi H (NCTC-901.8394), Pseudomonas putida sp., Brucella abortus (A.99, UK-1995) RSKK-03026. Sh. boydii type 11 (Pasteur51.6), Sh. boydii type 16 (cHe 67.11), Sh. boydii type 6 (RSKK-96043), and antifungal activity against Candida albicans (Y-1200-NIH, Tokyo). Some of the compounds exhibited activity comparable to ampicillin ofloxacin, nystatin, kanamycin, sulphamethoxazol, amoxycillin, and chloroamphenicol. Most of the studied compounds were found effective against bacteria studied and yeast.



Similar content being viewed by others
References
AbouEl-Enein SA, El-Saied FA, Kasher TI, El-Wardany AH (2007) Synthesis and characterization of iron(III), manganese(II), cobalt(II), nickel(II), copper(II) and zinc(II) complexes of salicylidene-N-anilinoacetohydrazone (H2L1) and 2-hydroxy-1-naphthylidene-N-anilinoacetohydrazone (H2L2). Spectrochim Acta A Mol Biomol Spectrosc 67:737–743. doi:10.1016/j.saa.2006.07.052
Angelici RJ (1990) Structural aspects of thiophene coordination in transition metal complexes. Coord Chem Rev 105:61–76. doi:10.1016/0010-8545(90)80018-O
Arslan S, Logoglu E, Öktemer A (2006) Antimicrobial activity studies on some piperidine and pyrrolidine substituted halogenobenzene derivatives. J Enzyme Inhib Med Chem 21:211–214. doi:10.1080/14756360600563063
Avaji PG, Patil SA, Badami S (2008) Transition metal complexes of 13- and 14-membered N2O2 macrocycles: synthesis and characterization. Trans Met Chem (Weinh) 33:275–283. doi:10.1007/s11243-007-9041-z
Basak S, Mondal A, Chopra D, Rajak KK (2007) Synthesis and structural characterisation of new Re(III) complexes using aldimines of α-amino acids as coligands. Polyhedron 26:3465–3470. doi:10.1016/j.poly.2007.03.036
Bermingham A, Derrick JP (2002) The folic acid biosynthesis pathway in bacteria evaluation of potential for antibacterial drug discovery. Bioessays 24:637–648. doi:10.1002/bies.10114
Burlov AS, Shepelenko EN, Vasil’chenko IS, Antsyshkina AS, Sadkov GG, Matuev PV, Nikolaevskii SA, Borodkin GS, Sergienko VS, Bren’ VA, Garnovskii AD (2006) Metal chelates with salicylidene-3-carboethoxy-4,5-dimethylthiophene derivatives as azomethine ligands of a new type. Russ J Coord Chem 32:879–884. doi:10.1134/S1070328406120049
Cotton FA, Wilkinson G (1972) Advanced inorganic chemistry, 3rd edn. Wiley, New York
Farkas E, Bátka D, Csóka H, Nagy NV (2007) Interaction of imidazole containing hydroxamic acids with Fe(III): hydroxamate versus imidazole coordination of the ligands Bioinorg Chem Appl 96536:1–8
Gewald K, Schinke E, Böttcher H (1966) Heterocyclen aus CH-aciden Nitrilen, VIII. 2-Amino-thiophene aus methylenaktiven Nitrilen. Carbonylverbindungen Schwefel Chemische Ber 99:94–100. doi:10.1002/cber.19660990116
Hallas G, Choi J-H (1999) Synthesis and properties of novel aziridinyl azo dyes from 2-aminothiophenes-Part 1: synthesis and spectral properties. Dyes Pigments 40:99–117. doi:10.1016/S0143-7208(98)00034-5
Halling SM, Peterson Burch BD, Bricker BJ, Zuerner RL, Qing Z, Li L, Kapur V, Alt DP, Olsen SC (2005) Completion of the genome sequence of Brucella abortus and comparison to the highly similar genomes of Brucella melitensis and Brucella suis. J Bacteriol 187:2715–2726. doi:10.1128/JB.187.8.2715-2726.2005
Herrera V, Muñoz B, Landaeta V, Canudas N (2007) Homogeneous hydrogenation of imines catalyzed by rhodium and iridium complexes. Kinetics and mechanism of the hydrogenation of N-(β-naphthyl methylene) aniline using [Ir(COD)(PPh3)2]PF6 as catalyst precursor. J Mol Cataly A 174:141–149
Jain M, Singh RV (2006) Synthesis, characterization and biotoxicity of NN donor sulphonamide imine silicon (IV) complexes. Bioinorg Chem Appl 2006:10. doi:10.1155/BCA/2006/13743
Joseph J, Mehta BH (2007) Synthesis, characterization, and thermal analysis of transition metal complexes of polydentate ONO donor Schiff base ligand. Russ J Coord Chem 53:124–129. doi:10.1134/S1070328407020091
Kurtoğlu M, İspir E, Kurtoğlu N, Serin S (2008) Novel vic-dioximes: synthesis, complexation with transition metal ions, spectral studies and biological activity. Dyes Pigments 77:75–80. doi:10.1016/j.dyepig.2007.03.010
Logoglu E, Arslan S, Öktemer A, Şakıyan I (2006) Biological activities of some natural compounds from Sideritis sipylea Boiss. Phytother Res 20:294–297. doi:10.1002/ptr.1855
Lippard SJ, Berg JM (1994) Principles of bioinorganic chemistry. University Science Books, Mill Valley, CA
Modi JD, Sabnis SS, Deliwala CV (1970) Potential anticancer agents. III. Schiff bases from benzaldehyde nitrogen mustards and aminophenylthiazoles. J Med Chem 13:935–941. doi:10.1021/jm00299a031
Mohanan K, Devi SN, Murukan B (2006) Complexes of copper(II) with 2-(N-salicylideneamino)-3-carboxyethyl-4,5,6,7-tetrahydrobenzo[b]thiophene containing different counter anions. Synt React Inorg Met Org Chem 36:441–449
Murukan B, Mohanan K (2007) Synthesis, characterization and antibacterial properties of some trivalent metal complexes with [(2-hydroxy-1-naphthaldehyde)-3-isatin]-bishydrazone. J Enzym Inhib Med Chem 22:65–70
Nakamoto K (1986) Infrared and Raman spectra of inorganic and coordination compounds, 4th edn. Wiley, New York
Nawwar GAM, Shafik NA (1995) Synthesis of 2-substituted benzothiazoles containing amino acid, imino or heteroaryl moieties with anticipated fungicidal activity. Collect Czech Chem Commun 60:2200–2208. doi:10.1135/cccc19952200
Peet NP, Sunder S, Barbuch RJ, Vinogradoff AP (1986) Mechanistic observations in the Gewald syntheses of 2-aminothiophenes. J Heterocycl Chem 23:129–134. doi:10.1002/jhet.5570230126
Sabnis RW, Rangnekar DW, Sonawane ND (1999) 2-Aminothiophenes by the Gewald reaction. J Heterocycl Chem 36:333–346. doi:10.1002/jhet.5570360203
Sari N, Gürkan P (2004) Some novel amino acid-Schiff bases and their complexes synthesis, characterization, solid state conductivity behaviors and potentiometric Studies. Z Naturforsch [B] 59b:692–697
Sari N, Gürkan P, Arslan S (2003) Synthesis, potentiometric and antimicrobial activity studies on 2-pyridinilidene-DL-amino acids and their complexes. Trans Met Chem (Weinh) 28:468–474. doi:10.1023/A:1023685719951
Sari N, Nartop D, Karcı F, Dişli A (2008) Novel hydrazone derivates and their tetracoordinated metal complexes. Asian J Chem 20:1975–1985
Silverstein RM, Bassler GC, Morrill TC (1981) Spectrophotometric identification of organic compounds, 4th edn. Wiley, New York
Singh K, Singh DP, Barwa MS, Tyagi P, Mırza Y (2006) Antibacterial Co(II), Ni(II), Cu(II) and Zn(II) complexes of Schiff bases derived from fluorobenzaldehyde and triazoles. J Enzyme Inhib Med Chem 21:557–562. doi:10.1080/14756360600642131
Takeuchi T, Böttcher A, Quezada CM, Meade TJ, Gray HB (1999) Inhibition of thermolysin and human α-thrombin by cobalt(III) Schiff base complexes. Bioorg Med Chem 5:815–819. doi:10.1016/S0968-0896(98)00272-7
Zhikuan L, Twieg RJ, Huang SD (2003) Copper-catalyzed amination of aromatic halides with 2-N,N-dimethylaminoethanol as solvent. Tetrahedron Lett 44:6289–6292. doi:10.1016/S0040-4039(03)01536-3
Acknowlegments
The authors thank the Gazi Üniversity Research Fund (Project number: 05/2009-20) for financial support and Elif Loğoğlu, Nazlıgül Tolu, and İsmail Kutlu for antibacterial studies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Altundas, A., Sarı, N., Colak, N. et al. Synthesis and biological activity of new cycloalkylthiophene-Schiff bases and their Cr(III) and Zn(II) complexes. Med Chem Res 19, 576–588 (2010). https://doi.org/10.1007/s00044-009-9214-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-009-9214-8