Abstract
The reliance on visual cues can vary among ant species living in different habitats, and is mostly influenced by the level of ambient illumination and the morphological adaptations of ant species that can determine their period of daily activity. Thus, different illumination regimes might affect ant community composition in habitats with different ambient illumination intensities. Despite this, in the literature, information is scarce about the effects of ambient illumination on ant activity. In the present study, we investigated the effect of ambient illumination intensity on the activity of ant species and their community composition under herbaceous and woody plants in 24 localities from Kyiv and Kyiv region, Ukraine. Our results showed that the ambient illumination properties of the habitat type affect the presence of ant species and the activity of their workers. In open habitats, the maximum ambient illumination had a negative effect, whereas in closed, woody habitats the maximum ambient illumination and tree circumference generally had a positive effect on the number of non-dendrobiotic ant individuals found on individual plants. The dendrobiotic species were less affected by the maximum illumination intensity. Although, in closed habitats, the effect of the previous variables and their interaction had species-specific aspects. Based on the former, we can tell that more structured forest habitats can provide broader illumination intensity ranges, allowing more ant species to coexist. However, similar illumination intensity ranges can lead to overlaps in the activity of ant species under the same illumination conditions, especially in open habitats. The outcomes of these interactions are highly influenced by larger trees that besides lowering illumination intensities, can enhance ant worker activity and promote species coexistence.
Similar content being viewed by others
Introduction
The activity patterns of ants outside their nests are determined by a number of biotic (Czechowski and Markó 2006; Somogyi et al. 2017) and abiotic factors (Grześ et al. 2018; Bátori et al. 2019). For example, biotic factors include the composition of the vegetation (e.g., Grześ et al. 2018), but also the presence of carbohydrate and protein sources (e.g., Stukalyuk 2017a; Maák et al. 2021). Abiotic factors, on the other hand, can include the temperature and humidity of air and soil (Bátori et al. 2019; Juhász et al. 2020), or the ambient light regime (level of the illumination) (e.g., Stukalyuk 2017a). The combination of abiotic factors determines the microclimatic conditions that affect the activity patterns of ants during their outbound journeys. For ants, mostly for those living in deciduous forests, air temperature and soil moisture are of primary importance (Traniello 1989; Bátori et al. 2022), but the amount of precipitation and air humidity can also be decisive (Stukalyuk 2017a; Bátori et al. 2019).
Ambient illumination, the total luminous flux per unit area of a surface, is also one of the most important, but insufficiently studied factors that determine the activity of ants. Depending on the time of the year and the day, ambient illumination can show great fluctuations (Malet-Damour et al. 2019). Such changes in the ambient light intensity might influence the activity patterns of ants (Hodgson 1955; Narendra et al. 2010), but it can also influence the use of visual (Klotz and Reid 1993) and olfactory cues (Cammaerts and Rachidi 2009; Cammaerts 2012a, b). For example, in Lasius niger, it was found that with the decrease of ambient light levels, ants showed decreasing reliance on private visual information (e.g., memories), and a stronger reliance on social information (e.g., social signals like pheromone trails) due to lower navigational confidence (Jones et al. 2019). This can highly influence the tradeoffs between the expanses and gains of foraging, as low ambient light intensities can lead to lower foraging efficiency by causing a higher uncertainty and lower navigational success of workers (Klotz and Reid 1993; Narendra et al. 2013).
Ants can use a wide variety of visual orientation cues, for example, they can rely on the pattern of the polarized skylight (Wehner and Müller 2006), or visual landmark panorama (Akesson and Wehner 2002; Graham and Cheng 2009; Narendra et al. 2013). However, the reliance on visual cues can vary among species living in different habitats, having different activity periods (i.e., diurnal, nocturnal, or both) or foraging strategies (Klotz and Reid 1993; Wehner and Müller 2006; Cammaerts 2012a, b; Jones et al. 2019). For example, wood ants, when searching on the vertical axis of the vegetation, use simultaneously chemical cues and directional light, with a preference for the latter in conflicting information (Fourcassie and Beugnon 1988).
The ambient light intensity can be highly affected by the topography of the nest surroundings, like the composition of the vegetation that can cause a variable level of illumination (Tinya et al. 2009; Vele et al. 2009; Parui et al. 2015; Stukalyuk et al. 2020a). For instance, the vertical light attenuation can be more gradual and the horizontal light heterogeneity can be more pronounced in late succession forests (Matsuo et al. 2021, 2022), whereas the ambient illumination is higher in open areas with grassy vegetation (Endler 1993). Moreover, other characteristics (e.g., forest age, the presence of shrubs or clearings, canopy cover) of the vegetation can also strongly influence the levels of ambient illumination (Vele et al. 2009; Parui et al. 2015; Stukalyuk et al. 2020a; Matsuo et al. 2021, 2022). The differences in ambient illumination intensities can lead to different adaptations of different ant species. For example, the comparison of the compound eyes of three Camponotus (one exclusively nocturnal, one crepuscular and nocturnal, and one mainly diurnal) and one Cataglyphis species (strictly diurnal) of about the same body size showed that the eyes of the diurnal species contain more ommatidia and that ultrastructural changes occur in the ommatidia of the Camponotus, but not in the ommatidia of the Cataglyphis species (Menzi 1987). As a consequence, visual orientation becomes possible also in low levels of environmental light intensity in the crepuscular and nocturnal Camponotus species (Menzi 1987). Similarly, the diurnal Formica cunicularia workers have compound eyes with higher resolution, whereas the cathemeral Camponotus aethiops workers have eyes with lower resolution but higher sensitivity (Yilmaz et al. 2014). Thus, for each ant species, the level of ambient illumination and the related morphological adaptations influence their optimal period of daily activity (Menzi 1987; Yilmaz et al. 2014) that, besides other biotic and abiotic factors, might affect ant assemblage organizations in habitats with different ambient illumination intensities. Despite this, only one study addressed the possible preference for different ambient illumination levels by several Formica species (Vele et al. 2009).
In this study, we aimed to reveal the direct influence of the ambient illumination regime on the activity and structure of ant communities of different habitat types. Based on their main vegetation characteristic that influences the ambient illumination intensities, two habitat types were considered: open habitats with herbaceous plants, and closed habitats with woody vegetation. We hypothesized that the largest values of ambient illumination intensity will have opposite effects in the two habitat types (negative effect in open habitats, whereas positive in the closed habitats), and will also influence ant community composition. Moreover, we also hypothesized that maximum ambient illumination intensities will have a differential effect in different ant species leading to differences in the number of ant individuals, and in closed habitats, the tree circumference will modulate these effects. In dendrobiotic ant species, however, we hypothesized that the maximum ambient illumination intensity will have a lower effect than in the other species and that the tree circumference will pose a stronger influence.
Materials and methods
Study sites
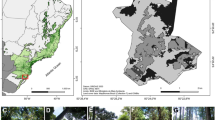
The study was carried out in June–August 2015–2018 in Kyiv (Ukraine) and its outskirts taking into account several habitat types (Fig. 1A, B).
The study sites in Kyiv and its outskirts (light gray—highly inhabited areas; dark gray—park and forest areas). Open habitat types with herbaceous vegetation are shown in white, whereas closed habitat types with woody vegetation are shown in gridded gray points. If in a study site both open and closed habitat types occurred, the points are filled half white and half gridded gray
Plants were surveyed in 24 sites, 20 of which were located within Kyiv, whereas four were in the outskirts of Kyiv. In total, 13,566 specimens of 32 species of herbaceous plants were examined in six sites in Kyiv and its outskirts. Based on the classification of the European Nature Information System (see EUNIS database) the studied sites for herbaceous plants belonged to six habitat type categories (Fig. 1): (a) E5.1—anthropogenic herb stands; (b) E5.21—xero-thermophile fringes; (c) FA.3—species-rich hedgerows of native species; (d) G1.D—fruit and nut tree orchards (partly open habitats); (e) G5.2—small broadleaved deciduous anthropogenic woodlands (partly open habitats); (f) I1.5—bare tilled, fallow or recently abandoned arable lands. Woody plant species were examined in 23 sites belonging to 10 different habitat types, during which in total 5301 specimens of 22 species were examined (Fig. 1). The investigated habitat types were: (a) E2.1—permanent mesotrophic pastures (open territories with groups of shrubs and seedlings of trees); (b) G1.A53—East-European linden forests; (c) G1.A162—mixed lime-oak-hornbeam forests; (d) G1.C2—exotic Quercus plantations; (e) G4.F—mixed forestry plantations; (f) G1.11—riverine Salix woodland; (g) G5.1—lines of trees; (h) G5.2—small deciduous anthropogenic woodlands; (i) I2—small cultivated areas of gardens and parks; (j) X11—large parks. The smaller number of sites in the open habitat types was due to the fact that to achieve evenness among the numbers in sampled herbaceous and woody plants, more herbaceous plants could be assessed in open areas compared to sites in closed habitat types where woody plants occupy a significant area. Therefore, to survey closed (woody) habitats a larger area had to be covered. If one study site involved more habitat types, the survey was conducted in every habitat type separately.
Sampling methods
Similarly to other studies (Zakharov 2015), the ant activity assessments were carried out during the same time period, i.e., from 10:00 to 13:00 h. Considering the bivalent effects of the change in ambient illumination (and temperature) on ant activity along a day, we considered ant activity only during this period, as a similar, but rather opposite pattern can be recorded during the afternoon. In general, during the day the sun shines at different intensities, but in total it gives the same amount of energy as if it shone for 5 h at maximum intensity. Thus, the indicators of total solar irradiation for Kyiv with a clear sky in June–August are in the range of 699–857 MJ/m2 (8570 Klux for June, 8290 Klux for July, and 6990 Klux for August) (Ribchenko and Revera 2012).
In every study site, all plants were considered along a 10-m transect with 5-m on each side (10-m width). Depending on the habitat size, there were 5–20 transects per study site. None of the transects crossed the boundaries between open and closed habitats. All ants climbing or descending on a plant were counted for 2 min in every plant individual. If there were only a few workers on a plant, they were counted visually. When visual counting was difficult due to a large number of ants moving along a trail, the exact number of ants was determined with the help of video recordings. In herbaceous plants, the whole plant was taken into account, whereas in woody plants, ants climbing the trunk at a height of 2 m were considered. In trees and shrubs, the measurements of the circumference of the tree trunks and shrubs were also carried out at breast height (1.6 m). We measured the circumference of the trunks as it is a more accurate representation of the size (age) of the trees since the diameter of the tree trunks does not have the correct geometric shape. In the dendrobiotic (build their nests in cavities under the trunks or roots, but also within the decaying parts of living trees and forage almost exclusively on trees; Czechowski et al. 2012) ant species (Lasius fuliginosus, L. brunneus, Dolichoderus quadripunctatus), if detected, the presence of trails and nests were separately indicated (the intensity of movement of ants along one line in both directions; more than 10 individuals per 1 min). The ant species identification was carried out according to Radchenko (2016), although some species could not be determined to species (only genera) level under field conditions; in such cases, the genera were used, but not mentioned separately hereafter.
The illumination level was measured using a CEM DT-1307 Solar Power Meter (Shenzen Everbest Machinery Industry, China) with sunlight measurements up to 1999 w/m2 or 634 BTU/(ft2 × h), resolution 1 W/m2 or 1 BTU/(ft2 × h), accuracy typically within ± 10 W/m2 [± 3 BTU/(ft2 × h)] or ± 5%, and the sampling time of approximately 0.25 s (see https://www.cem-instruments.com/en/product-id-1311). Under herbaceous plants, the illumination level was measured under their layer for 30 specimens belonging to the same species. Under the forest canopy, the illumination (in kLux) was measured for every ten woody plants of the same species with similar heights. In forests with several tree and shrub species, measurements were taken separately for each species. In every case, the minimum and maximum illumination levels were measured. During each illumination measurement, the observer was positioned in such a way that his shadow did not cover the view of the equipment. All measurements of illumination were carried out for 3 h (from 10:00 to 13:00) when the sun was at its zenith. The average illumination level was calculated for this time period and used for further analyses.
Statistical analysis
We provided a complete description of the optimal intervals of ambient illumination levels (minimum, average, maximum, Table 2) for 11 ant species and species groups visiting herbaceous plants and for 20 ant species and species groups visiting woody plants in closed habitats. The data for herbaceous and woody plants were calculated separately since herbaceous plants growing in open areas are under more intense ambient illumination conditions than in forests. In our models, the logarithm of the maximum ambient illumination intensity was used, as perceptual responses of invertebrates to visual cues are on a log scale as suggested by Stevens’ power law (Stevens 1961).
Generalized Linear Mixed Models (Poisson error, maximum likelihood fit) were used to analyse the effects of the logarithm of maximum ambient illumination intensity (maximum ambient illumination hereafter) on the number of ant individuals (ant activity hereafter) found on herbaceous plants assessed from the different habitat types. In the model, the year, habitat types and plant species were included as nested random factors. Similar model construction was used to analyse the effect of maximum ambient illumination, tree circumference, and their interaction on the ant activity on the woody plants assessed from the different habitat types. Separate models were built for dendrobiotic and non-dendrobiotic ant species, as the presence of the dendrobiotic species on these plants is not exclusively linked with foraging. In the models, the maximum ambient illumination, tree and shrub circumference, and the interaction of the two variables were included as continuous variables, whereas the year, habitat types, and the sites of the observation were included as nested random factors. The same model constructions were used to test the effect of these variables on the activity of the most common ant species.
In all previous models, only those plant individuals were included on which at least one ant individual was found. Five plant individuals with ants (when being the single representatives of a separate species from a separate habitat type), whereas Leptothorax sp. with one individual were excluded from further analyses to avoid inflating factor levels. GLMMs were performed using glmer function of the ‘lme4’ package (Bates et al. 2015). In model over-dispersion, a negative binomial error term was applied. The p values for the final models were obtained by the function Anova from the ‘car’ package (Fox and Weisberg 2011). All statistical analyses were carried out in the R Statistical Environment (R Core Team 2019).
Results
Altogether, 50,370 ant workers belonging to 21 species were found on plants. On herbaceous plants from open habitats, 7400 ant workers of 11 species, whereas on woody plants from closed habitats 42,970 ant workers of 20 species (including dendrobiotic ones) were found (Table 1). In the study period, the most active ant species on herbs were F. cinerea (1375 workers), F. cunicularia (292), F. rufibarbis (128), and L. niger (3664) (Table S1). The most active ant species on woody plants were D. quadripunctatus (3799), F. cinerea (4622), F. polyctena (600), F. rufa (1212), L. brunneus (632), L. emarginatus (2744), L. fuliginosus (2240), L. niger (6144) and L. neglectus (1715) (Table 2). The remaining species had a smaller number of workers on the herbaceous and woody plants in the study period (Tables 1 and 2).
Ambient illumination and ant activity on herbaceous plants
In open habitats, from the 13,566 herbaceous plants assessed at least a single ant worker was found on 1344 plants (9.21%, Table 1). We found that the majority of ant species monopolized the plants they were visiting. In herbaceous plants, one ant species was present in 96.5% (1297 per total of 1344), two species were present in 3.35% (45 per 1344), whereas three species only in 0.15% (2 per 1344) of plants.
The ants found on herbaceous plants can be divided into two main groups. The first group included three eurybiontic ant species (F. cinerea, F. rufibarbis, and L. niger) that were found in most (3–5 types) habitats with a broad range of ambient illumination, but with a strong bias towards more illuminated habitats (Table 2). This also included two ant species (F. pratensis, F. cunicularia) that are typical for open habitats (meadows), but were found only in one habitat type assessed. Moreover, Formica pratensis was found visiting only the individuals of the Cirsium vulgare plant species (Table 1). The second group consisted of six ant species (Myrmica sp., Temnothorax sp., L. emarginatus, Leptothorax sp., F. fusca, D. quadripunctatus), which were more typical for forests, where the level of ambient illumination was lower than in open areas (Table 2).
In general, the maximum ambient illumination had a negative effect on the total number of ant individuals active on herbaceous plants (GLMM z = − 4.43, p < 0.001; Table 2; Fig. 2A).
The effect of maximum ambient illumination was also analysed separately in the most common ant species and species groups (F. cinerea, L. niger, Myrmica sp., F. rufibarbis) found on individual herbaceous plants (Table 1). In F. cinerea, the maximum ambient illumination had a strong negative effect on its activity on herbaceous plants (GLMM z = − 5.42, p < 0.001; Fig. 2B). In other more common species, however, the maximum illumination did not have any effect on the number of individuals (L. niger: z = − 0.99, p = 0.33, Fig. 2B; Myrmica sp.: z = − 1.18, p = 0.24; F. rufibarbis: z = 1.25, p = 0.21). The individuals of F. cunicularia (82.2%) and F. pratensis (100%) were mostly found under the same illumination maxima (7.6 kLux), thus their separate analyses were obsolete.
Ambient illumination and ant activity on woody plants
In woody plants, from the 5301 individual plants assessed at least a single ant worker was found on 2599 plants (49%, dendrobiotic included, Table 1). We found one plant visited by five ant species (0.04% per 2599 woody plants), nine by four ant species (0.35%), 51 by three ant species (1.96%), 367 by two species (14.12%), and 2171 by only one ant species (83.53%). The three dendrobiotic ant species visited altogether 514 woody plants with 13,000 workers (Table 3). On the other hand, non-dendrobiotic species visited 2272 woody plants with a total of 29,970 workers.
From the 20 ant species found on woody plants, three species belonged to the genus Camponotus (C. vagus, C. ligniperda, C. fallax), six to Formica (F. cinerea, F. cunicularia, F. fusca, F. polyctena, F. rufa, F. rufibarbis) and seven to Lasius. The Formica and Camponotus species can be either active at different times of the day (e.g., C. ligniperda), or not characteristic of forest habitats (e.g., F. cinerea, F. cunicularia, F. rufibarbis), or can be characterized by individual foraging (e.g., F. fusca, C. fallax). Most Lasius species had a high abundance, being present in 5–8 habitat types (Table S2). The most common were L. niger, L. fuliginosus, and L. emarginatus, but also species from other genera were quite common, like F. cinerea, Myrmica sp., and Temnothorax sp. (Table 2). On the other hand, some species were abundant in only one habitat type (red wood ants and L. neglectus; Table 2) visiting specific woody species. For example, Lasius neglectus was mainly visiting the individuals of Tilia cordata and Ulmus laevis (Table 1), whereas Formica polyctena only the individuals of Pinus sylvestris and F. rufa the individuals of Acer platanoides, Carpinus betulus, and Quercus robur (Table 1).
Non-dendrobiotic ant activity on woody plants
In general, the maximum ambient illumination (GLMM z = 3.91, p < 0.001; Fig. 3A) and tree circumference (z = 11.98, p < 0.001; Fig. 3B) had a positive effect on the number of non-dendrobiotic ant individuals found on woody plants. However, the interaction of the two former variables did not have a significant effect on the number of non-dendrobiotic ant individuals found on woody plants (z = − 1.18, p = 0.24).
Additionally, we analysed separately the effect of the maximum ambient illumination, tree circumference and the interaction of the two variables on the activity of the most common ant species (F. cinerea, L. emarginatus, L. niger, L. platythorax) and species groups (Myrmica sp., Temnothorax sp.) found on individual woody plants (Tables 1 and 4). The variables had different effects on the different ant species (Table 4). The number of F. cinerea, Myrmica sp., and Temnothorax sp. individuals were affected positively by the maximum ambient illumination (Table 4), whereas the tree circumference had a positive effect in F. cinerea, L. emarginatus and L. niger, whereas a negative effect in Myrmica sp. (Table 4). The number of ant individuals active on the tree trunks was lower (F. cinerea, Temnothorax sp.) or larger (L. platythorax) on trees with larger circumferences exposed to larger maximum ambient illumination (Table 4).
Dendrobiotic ant activity on woody plants
In general, the dendrobiotic ant activity was influenced only by the tree circumference (GLMM z = 7.38, p < 0.001), whereas the maximum ambient illumination (z = 0.12, p = 0.9) and its interaction with tree circumference (z = 0.24, p = 0.81) did not have significant effects. Analysing also separately the effects of these variables on the dendrobiotic ant species, we found that the activity of the D. quadripunctatus was affected negatively by the maximum ambient illumination (GLMM z = − 2.28, p = 0.02), whereas its activity was larger on the tree trunks with larger circumference exposed to larger maximum ambient illumination (z = 4.19, p < 0.001). The tree circumference did not have a significant effect on the activity of this species (z = − 1.42, p = 0.15). On the other hand, the activity of L. fuliginosus was affected positively by the tree circumference (z = 5.29, p < 0.001), but not by the other two variables (log max. illumination: z = 0.6, p = 0.55; interaction: z = − 0.56, p = 0.58). The activity of L. brunneus was not affected by either of the variables (max. illumination: z = − 0.8, p = 0.42; tree circumference z = 1.2, p = 0.23; interaction: z = 0.043, p = 0.97).
Discussion
The characteristics of a habitat’s vegetation can highly affect its ambient illumination properties which influence the presence of ant species and the activity of their workers. The number of ant individuals was influenced by the maximum ambient illumination in both habitat type categories under study. In open habitats, the maximum ambient illumination had a negative effect on the number of ant individuals, whereas in closed habitats it had the opposite effect. Moreover, in these latter habitats, the reduced ambient illumination due to the larger tree circumference had mostly a negative effect on the number of non-dendrobiotic ant individuals. On the other hand, the dendrobiotic species were affected by the circumference of the woody species which mainly had a positive effect on the number of individuals of both dendrobiotic and non-dendrobiotic ant individuals found in woody habitats. However, the effects of the mid-day maximum ambient illumination and vegetation can be species-specific and altered also by other factors, like competition and food availability.
In both open and closed habitat types, the ambient illumination had an impact on the organization of ant communities. This can be due to the differential effects of ambient illumination levels on ant activity and foraging (Menzi 1987; Klotz and Reid 1993; Yilmaz et al. 2014; Symonowicz et al. 2015; Stukalyuk et al. 2020a). Illumination can determine the visual orientation of ants during foraging through the use of different visual signals (Gileva and Gilev 2019; Jones et al. 2019). For example, in Myrmica ants it was found that they are capable of recognizing objects with different shapes and colors (Cammaerts 2012a, b), and a decrease in light intensity can serve as a signal for the start of foraging activity (Narendra et al. 2010). Moreover, the level of preferred illumination can also determine the eye structure of ants being in close relationship with their reliance on visual cues (Yilmaz et al. 2014). However, in twilight or night (0–10 lx), when visual cues are less available, ants can switch to greater deposition of trace pheromones and use them as social cues (Beugnon and Fourcassie 1988; Jones et al. 2019). Other ants, like New World army ants, rely mostly on chemical cues also under conditions of proper illumination, although their activity is also determined by the level of illumination (Hölldobler and Wilson 1990). Thus, in open habitats, the ant activity was less variable due to the more even illumination. On the other hand, in closed habitats, the more heterogeneous ambient illumination conditions and less light available under the canopies can have a more differential effect on ant activity.
Forest vegetation provides a greater variety of ambient illumination regimes, depending on the density of the canopy and the age of the trees (Häusler et al. 2016). Thus, in forests, ant species with different light preferences can settle: from open areas with a high level of illumination (glades, edges) to completely shaded areas in the depths of the forest (Czechowski et al. 2012; Radchenko 2016; Seifert 2018). We have found that within forested habitats, besides maximum ambient illumination, the tree circumference also played an important role in the organization of ant communities, and had a positive effect on the number of ant individuals. Moreover, the larger shading caused by larger trunks had a negative effect on the number of ant individuals. Our data also showed that the vast majority of ant species visited trees with a trunk circumference of 1.26 m or larger which was especially true for dendrobiotic species. Their nests and trails are usually located on the largest trees (based on trunk circumference), although their trails can be present also on smaller trees, whereas the smallest trees are only visited by single foragers. For example, Hopkins and Thacker (1999) found a relationship between the occurrence of some aphid species and L. fuliginosus ants on trees, although this was not influenced by the diameters of trees. For other ant species, such as red wood ants, the largest trees (1.2 m in circumference) are the most visited because they are the main source of aphid honeydew (Gibb and Johansson 2010; Gibb et al. 2016). It was shown that the pine, oak, birch, hornbeam and spruce trees visited by F. rufa and F. polyctena had larger diameters compared to trees that were not visited (Sondej et al. 2020). Thus, trees with a larger trunk circumference can be more frequently visited by ants due to the presence of potential food sources. This is our case can be possible in Formica and Lasius species, like F. cinerea, L. emarginatus, L. fuliginosus, and L. niger.
Besides the former, the effects of the vegetation characteristics can also interact with the effects of microclimatic and physical characteristics of a habitat that in their turn can determine the distribution and abundance of ant species (Boomsma and De Vries 1980; Doncaster 1981; Gallé 1991; Jayatilaka et al. 2011; Bátori et al. 2019, 2022). For example, the environmental variables inside the canopy can be considerably different from those outside the canopy (Kitaya et al. 1998). On the other hand, the maximum ambient insolation can also influence the temperature of a surface, although this can be determined by many factors, like the cooling mechanism of the substrate (Li et al. 2015; Li et al. 2018 for a review). In general, the insolation and air and ground temperatures are always highly correlated, but they are not equivalent if used to explain features of ant biology (Elmes and Wardlaw 1982). The increased temperature can also highly influence the microclimate (e.g., air humidity) and as a result, ant activity (Jayatilaka et al. 2011). Moreover, the higher surface temperature can also accelerate pheromone degradation (van Oudenhove et al. 2011) and increase the running speed of ants (Ślipiński and Cerdá 2022). These factors can determine community organization depending also on the site characteristics and thermal tolerance of species (van Oudenhove et al. 2011; Stuble et al. 2013; Bátori et al. 2019, 2022).
Generally, the information in the literature about the effect of ambient light levels on ants is usually indirect. In one of our previous works, we found that the highest number of workers is found in habitats with natural illumination levels up to 5 kLux (Stukalyuk et al. 2020a). Open and closed habitats (forests) can be very different in terms of ant species diversity, which is generally higher in forests compared to open habitats (Andersen 2019), although tree monocultures can have a lower variability of species diversity compared to areas with natural vegetation (Cerdá et al. 2009).
Our results mainly concern ant species with daytime activity and showed that different species of ants prefer different ambient illumination regimes at which foraging takes place. In open habitats, these intervals can lead to overlaps in the activity of species under the same illumination conditions. For example, that of F. cinerea with those of L. niger can lead to competition between them, especially in open habitats. Both species are dominant in most biotopes of Kyiv, being mostly present in meadows, pastures, alleys of trees, and parks (Stukalyuk et al. 2019; Stukalyuk et al. 2020a). In our measurements, F. cinerea had one of the widest ranges of maximum ambient illumination being between 0.9–35.0 kLux in open and up to 67.0 kLux in woody habitats. Despite being a thermotolerant species (Ślipiński and Cerdá 2022), the maximum ambient illumination had a strong negative effect on its activity on plants in open, and a strong positive effect in closed habitats. Similar to the former, Formica rufibarbis is also able to inhabit biotopes with a maximum illumination level comparable to the preferences of F. cinerea in open spaces (15.1 kLux). The rest of the species visiting herbaceous plants had a smaller amplitude of the illumination level optima, as well as lower average values (L. emarginatus, L. platythorax, Temnothorax sp., Myrmica sp.). This is due to the fact that these species are typical for forest communities (Radchenko 2016), where the ambient illumination regime is lower. Myrmica sp. in open spaces occurred typically in less illuminated areas than L. niger or F. cinerea, thus Myrmica sp. can avoid these two species. Moreover, in summer, the activity peak of some species, like the one belonging to the genus Myrmica, is mainly at dusk and at night which can lead to a low activity in the studied period (see Czechowski et al. 2012; Radchenko 2016; Seifert 2018), an effect that should be taken into account when considering our results. The nocturnal activity of Myrmica is usually caused by the presence of competing species that forage mainly during the day, but it can also be associated with high temperatures during the hot summer months (Vepsäläinen and Savolainen 1990).
Contrary to open habitats, closed forest habitats are more structured, thus they can provide broader ambient illumination intensity ranges, allowing for more ant species to coexist. Therefore, forests are represented by a set of areas with different ambient illumination regimes, each being inhabited by an ant species with a peculiar preference. Naturally, the minimum and maximum ambient illumination values for different ant species will overlap, but the optimal ambient illumination values for most ant species are different, indicating their differentiation in preference. If we arrange forest ant species that visited woody plants in a growing order according to the preferred average ambient light intensity, we get the following hierarchy: F. rufa (1.1 kLux), L. emarginatus and L. fuliginosus (2.3–2.4 kLux), F. polyctena (2.9 kLux), L. platythorax, but also L. brunneus and D. quadripunctatus (3.2—3.6 kLux), F. cinerea (3.9 kLux), L. niger (5.5 kLux), and C. fallax (6.6 kLux). However, dominant species, like L. emarginatus and L. fuliginosus, living in the same ambient illumination conditions are competing with each other (Stukalyuk 2017b).
According to our data, it turns out that L. emarginatus and L. fuliginosus (2 kLux) live in low-illuminated areas within a forest, L. platythorax, L. brunneus, and D. quadripunctatus (3 kLux) live in middle-illuminated areas, whereas the most illuminated places in the habitats with woody vegetation are occupied by L. niger, C. fallax, and red wood ants. The widest ambient illumination amplitudes were found for C. fallax, F. fusca, and L. brunneus that occurred in forest patches, whereas the red wood ants included in this study lived in most shaded areas of deciduous forests, although their optimal ambient illumination intensities found in earlier studies were 7.7 kLux for F. rufa and 5.0 kLux for F. polyctena (Stukalyuk et al. 2020b, 2021). In red wood ants, it is also known that in 90% of cases their nests are located in a strip no further than 10–15 m from the edge or clearing (Seifert 2018). Other dominant ant species appear in forests in a sporadic manner or are confined to the forest edges (Radchenko et al. 2019). Lasius brunneus, L. platythorax, and C. fallax, which are usually subdominant, live in more illuminated forest areas than those preferred by L. emarginatus and L. fuliginosus, and their average optima did not overlap. The subordinate ant species, like Myrmica sp. and Temnothorax sp., occurred in forested areas with a very narrow range of ambient illumination intensities (1.8–1.9 kLux), whereas F. fusca preferred more illuminated places (3.3 kLux), as found also by Vele et al. (2009). Such differences can be also due to the organizational effects of this dominant species on the subordinate ant activity (Savolainen and Vepsäläinen 1988; Maák et al. 2020).
On the other hand, open habitat specialists also occurred in habitats with woody vegetation. For example, Formica cinerea, a species inhabiting sandy areas on the southern sides of woodlands (Czechowski et al. 2012), from where it visits the nearby trees, or Lasius niger, a species present mostly in parks and squares than in forested areas (Radchenko et al. 2019). This species can also enter forested habitats only from open spaces where it lives (Radchenko 2016). Moreover, also the colonies of the dendrobiotic D. quadripunctatus are usually small in habitats with closed woody vegetation, but it can form supercolonies in gardens (Stukalyuk 2018).
Conclusion
The vegetation characteristics of a habitat can highly affect its ambient illumination characteristics that influence the ant community composition and activity patterns. In ants, different species, but also different aged workers of a species can have different illumination preferences that are shown by their trait adaptations linked with their use of cues during foraging. As such, different conditions of ambient illumination can determine the exploitation efficiency of food sources and colony fitness. Our results showed that in some ant species, the amplitude of the optimum ambient illumination is rather narrow, while other species of ants demonstrate wider adaptation to different ambient illumination conditions. Moreover, submissive ant species can be characterized by a preference for lighting modes that do not coincide with the optima of dominants, whereas subdominants tend to be active in a wider range of ambient illumination conditions. Thus, habitat heterogeneity can promote ant species coexistence also through various ambient illumination intensities, a factor that is mainly indirectly assessed but should get a stronger emphasis in relevant ecological studies.
Data availability
The data underlying the results presented in the paper are available in Dryad Digital Repository: https://doi.org/10.5061/dryad.w9ghx3frb.
References
Akesson S, Wehner R (2002) Visual navigation in desert ants Cataglyphis fortis: are snapshots coupled to a celestial system of reference? J Exp Biol 205:1971–1978
Andersen AN (2019) Responses of ant communities to disturbance: five principles for understanding the disturbance dynamics of a globally dominant faunal group. J Anim Ecol 88:350–362. https://doi.org/10.1111/1365-2656.12907
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bátori Z, Vojtkó A, Maák IE et al (2019) Karst dolines provide diverse microhabitats for different functional groups in multiple phyla. Sci Rep 9:7176. https://doi.org/10.1038/s41598-019-43603-x
Bátori Z, Gallé R, Gallé-Szpisjak N, Császár P, Nagy DD, Lőrinczi G, Torma A, Cs T, Maák IE, Frei K, Hábenczyus AA, Hornung E (2022) Topographic depressions provide potential microrefugia for ground-dwelling arthropods. Elementa 10:00084
Boomsma JJ, De Vries A (1980) Ant species distribution in a sandy coastal plain. Ecol Entomol 5:189–204
Cammaerts M-C (2012a) Navigation system of the ant Myrmica rubra (Hymenoptera: Formicidae). Myrmecol News 16:111–121
Cammaerts MC (2012b) The visual perception of the ant Myrmica ruginodis (Hymenoptera: Formicidae). Biologia 67:1165–1174. https://doi.org/10.2478/s11756-012-0112-z
Cammaerts M-C, Rachidi Z (2009) Olfactive conditioning and use of visual and odorous elements for movement in the ant Myrmica sabuleti (Hymenoptera, Formicidae). Myrmecol News 12:117–127
Cerdá X, Palacios R, Retana J (2009) Ant community structure in citrus orchards in the mediterranean basin: impoverishment as a consequence of habitat homogeneity. Environ Entomol 38:317–324. https://doi.org/10.1603/022.038.0203
Czechowski W, Markó B (2006) Uncomfortable protection: Formica polyctena FÖRST. shelters Formica fusca L. from Formica sanguinea LATR. (Hymenoptera: Formicidae). Annal Zool 56:539–548
Czechowski W, Radchenko A, Czechowska W, Vepsäläinen K (2012) The ants (Hymenoptera, Formicidae) of Poland with reference to the myrmecofauna of Europe. Museum and Institute of Zoology, Polish Academy of Sciences, Warsaw
Doncaster CP (1981) The spatial distribution of ants’ nests on Ramsey Island, South Wales. J Anim Ecol 50:195–218
Elmes GW, Wardlaw JC (1982) A population study of the ants Myrmica sabuleti and Myrmica scabrinodis, living at two sites in the South of England. I. A comparison of colony populations. J Anim Ecol 51:651–664
Endler JA (1993) The color of light in forests and its implications. Ecol Monog 63:2–27. https://doi.org/10.2307/2937121
Fourcassie V, Beugnon G (1988) How do red wood ants orient when foraging in a three dimensional system? I Laboratory experiments. Insect Soc 35:92–105. https://doi.org/10.1007/BF02224141
Fox J, Weisberg S (2011) An R companion to applied regression, 2nd edn. Sage, Thousand Oaks
Gallé L (1991) Structure and succession of ant assemblages in a north European sand dune area. Holarc Ecol 14:31–37
Gibb H, Johansson T (2010) Forest succession and harvesting of hemipteran honeydew by boreal ants. Ann Zool Fenn 47:99–110. https://doi.org/10.5735/086.047.0203
Gibb H, Andersson J, Johansson T (2016) Foraging loads of red wood ants: Formica aquilonia (Hymenoptera: Formicidae) in relation to tree characteristics and stand age. PeerJ 4:e2049. https://doi.org/10.7717/peerj.2049
Gileva OB, Gilev AV (2019) Development of behavioral skills in Myrmica ants (Hymenoptera, Formicidae) through varied nest illumination. Entmol Rev 99:137–142. https://doi.org/10.1134/S0013873819020015
Graham P, Cheng K (2009) Ants use the panoramic skyline as a visual cue during navigation. Curr Biol 19:R935–R937. https://doi.org/10.1016/j.cub.2009.08.015
Grześ IM, Ślipiński P, Babik H et al (2018) Colony size and brood investment of Myrmica rubra ant colonies in habitats invaded by goldenrods. Insect Soc 65:275–280. https://doi.org/10.1007/s00040-018-0612-0
Häusler M, Silva JMN, Cerasoli S, López-Saldaña G, Pereira JMC (2016) Modelling spectral reflectance of open cork oak woodland: a simulation analysis of the effects of vegetation structure and background. Int J Remote Sens 37:492–515. https://doi.org/10.1080/01431161.2015.1134847
Hodgson ES (1955) An ecological study of the behavior of the leaf-cutting ant Atta cephalotes. Ecology 36:293–304. https://doi.org/10.2307/1933235
Hölldobler B, Wilson EO (1990) The Ants. The Belknap Press of Harvard University, Cambridge
Hopkins GW, Thacker JI (1999) Ants and habitat specificity in aphids. J Insect Conserv 3:25–31. https://doi.org/10.1023/A:1009626405307
Jayatilaka P, Narendra A, Reid SF, Cooper P, Zeil J (2011) Different effects of temperature on foraging activity schedules in sympatric Myrmecia ants. J Exp Biol 214:2730–2738
Jones S, Czaczkes TJ, Gallager AJ, Bacon JP (2019) Copy when uncertain: Lower light levels result in higher trail pheromone deposition and stronger reliance on pheromone trails in the ant Lasius niger. Anim Behav 156:87–95. https://doi.org/10.1101/473579
Juhász O, Bátori Z, Trigos-Peral G, Lőrinczi G, Módra G, Bóni I, Kiss PJ, Aguilon DJ, Tenyér A, Maák I (2020) Large- and small-scale environmental factors drive distributions of ant mound size across a latitudinal gradient. Insects 11:350. https://doi.org/10.3390/insects11060350
Kitaya Y, Shibuya T, Kozai T, Kubota C (1998) Effects of light intensity and air velocity on air temperature, water vapor pressure, and CO2 concentration inside a plant canopy under an artificial lighting condition. Life Support Biosph Sci Int J Earth Space 5:199–203
Klotz JH, Reid BL (1993) Nocturnal orientation in the black carpenter ant Camponotus pennsylvanicus (DeGeer) (Hymenoptera: Formicidae). Ins Soc 40:95–106. https://doi.org/10.1007/BF01338835
Li G, Pei G, Ji L, Yang M, Su Y, Xu N (2015) Numerical and experimental study on a PV/T system with static miniature solar concentrator. Sol Energy 120:565–574. https://doi.org/10.1016/j.solener.2015.07.046
Li G, Xuan Q, Pei G, Su Y, Ji J (2018) Effect of non-uniform illumination and temperature distribution on concentrating solar cell—a review. Energy 144:1119–1136. https://doi.org/10.1016/j.energy.2017.12.067
Maák I, Zs C, Erős K, Zs P, Markó B (2020) Living on the edge: changes in the foraging strategy of a territorial ant species occurring with a rival supercolony—a case study. J Insect Behav 33:59–68. https://doi.org/10.1007/s10905-020-09745-x
Maák IE, Sondej I, Juhász O, Trigos-Peral G, Tenyér A, Camera J, Czechowski W (2021) Unexpected distribution of subordinates around nests of the wood ants. Acta Oecol 110:103709. https://doi.org/10.1016/j.actao.2021.103709
Malet-Damour B, Bigot D, Guichard S, Boyer H (2019) Photometrical analysis of mirrored light pipe: From state-of-the-art on experimental results (1990–2019) to the proposition of new experimental observations in high solar potential climates. Sol Energy 193:637–653. https://doi.org/10.1016/j.solener.2019.09.082ff.ffhal-02368566ff
Matsuo T, Martínez-Ramos M, Bongers F, van der Sande MT, Poorter L (2021) Forest structure drives changes in light heterogeneity during tropical secondary forest succession. J Ecol 109:2871–2884
Matsuo T, Hiura T, Onoda Y (2022) Vertical and horizontal light heterogeneity along gradients of secondary succession in cool- and warm-temperate forests. J Veg Sci 33:e13135. https://doi.org/10.1111/jvs.13135
Menzi U (1987) Visual adaptation in nocturnal and diurnal ants. J Comp Physiol 160:11–21. https://doi.org/10.1007/BF00613437
Narendra A, Reid SF, Hemmi JM (2010) The twilight zone: ambient light levels trigger activity in primitive ants. Proc R Soc B 277:1531–1538. https://doi.org/10.1098/rspb.2009.2324
Narendra A, Reid SF, Raderschall CA (2013) Navigational efficiency of nocturnal Myrmecia ants suffers at low light levels. PLoS ONE 8:e58801. https://doi.org/10.1371/journal.pone.0058801
Parui A, Chatterjee S, Basu P (2015) Habitat characteristics shaping ant species assemblages in a mixed deciduous forest in Eastern India. J Tropic Ecol 31:267–280. https://doi.org/10.1017/S0266467415000036
Radchenko AG (2016) Ants (Hymenoptera, Formicidae) of Ukraine. Institute of Zoology, Kiev (in Russian)
Radchenko AG, Stukalyuk SV, Netsvetov MV (2019) Ants (Hymenoptera, Formicidae) of Kyiv. Entomol Rev 99:753–773. https://doi.org/10.1134/S0013873819060058
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ribchenko LS, Revera TO (2012) Solar radiation in Kyiv and Odessa for a clear sky. Sci Pract Ukr Sci Pres Hydrometeorol Inst 261:72–86
Savolainen R, Vepsäläinen K (1988) A competition hierarchy among boreal ants: impact on resource partitioning and community structure. Oikos 51:135–155. https://doi.org/10.2307/3565636
Seifert B (2018) The ants of Central and North Europe. Lutra Verlags und Vertriebsgesellschaft, Tauer
Ślipiński P, Cerdá X (2022) Higher soil temperatures cause faster running and more efficient homing in the temperate thermophilous ant Formica cinerea (Hymenoptera: Formicidae). Myrmecol News 32:149–158. https://doi.org/10.25849/myrmecol.news_032:149
Somogyi AÁ, Lőrinczi G, Kovács J, Maák IE (2017) Structure of ant assemblages in planted poplar (Populus alba) forests and the effect of the common milkweed (Asclepias syriaca). Acta Zool Acad Sci Hung 63:443–457
Sondej I, Domisch T, Finér L, Czechowski W (2020) Wood ants prefer conifers to broadleaved trees in mixed temperate forests. Agric Forest Entomol 23:287–296. https://doi.org/10.1111/afe.12431
Stevens SS (1961) To honor Fechner and repeal his law. Science 133:80–86
Stuble KL, Pelini ShL, Diamond SE, Fowler DA, Dunn RR, Sanders NJ (2013) Foraging by forest ants under experimental climatic warming: a test at two sites. Ecol Evol 3:2045–7758. https://doi.org/10.1002/ece3.473
Stukalyuk SV (2017a) The effect of climatic factors on the activity of ants (Hymenoptera, Formicidae) in broadleaf forests of Kiev. Entomol Rev 97:1234–1243. https://doi.org/10.1134/S0013873817090056
Stukalyuk SV (2017b) Interactions of three dominant ant species, Lasius emarginatus (Ol.), Formica rufa L., and Lasius fuliginosus (Latr.) (Hymenoptera, Formicidae) among themselves and with subordinate species in broadleaf forests. Entomol Rev 97:747–767. https://doi.org/10.1134/S0013873817060057
Stukalyuk SV (2018) Supercolony of Dolichoderus quadripunctatus Linnaeus (Hymenoptera, Formicidae). Biol Bull Rev 8:449–462. https://doi.org/10.1134/S2079086418050092
Stukalyuk SV, Zhuravlev VV, Netsvetov MV, Kozyr MS (2019) Effect of invasive species of herbaceous plants and associated aphids (Hemiptera, Sternorrhyncha: Aphididae) on the structure of ant assemblages (Hymenoptera, Formicidae). Entomol Rev 99:711–732. https://doi.org/10.1134/S0013873819060022
Stukalyuk SV, Kozyr MS, Netsvetov MV, Zhuravlev VV (2020a) Effect of the invasive phanerophytes and aphids on them on the ant (Hymenoptera, Formicidae) assemblages. Halteres 11:56–89. https://doi.org/10.5281/zenodo.4192900
Stukalyuk SV, Radchenko YN, Netsvetov MV, Gilev AA (2020b) Effect of mound size on intranest thermoregulation in the red wood ants Formica rufa and F. polyctena (Hymenoptera, Formicidae). Turk J Zool 44:266–280. https://doi.org/10.3906/zoo-1912-26
Stukalyuk S, Gilev A, Antonov I, Netsvetov M (2021) Size of nest complexes, the size of anthills, and infrastructure development in 4 species of red wood ants (Formica rufa, F. polyctena, F. aquilonia, F. lugubris) (Hymenoptera; Formicidae). Turk J Zool 45:464–478. https://doi.org/10.3906/zoo-2105-39
Symonowicz B, Kieruzel M, Szczuka A, Korczynska J, Wnuk A et al (2015) Behavioral reversion and dark–light choice behavior in workers of the red wood ant Formica polyctena. J Insect Behav 28:245–256. https://doi.org/10.1007/s10905-015-9496-2
Tinya F, Márialigeti S, Király I, Németh B, Ódor P (2009) The effect of light conditions on herbs, bryophytes and seedlings of temperate mixed forests in Őrség, Western Hungary. Plant Ecol 204:69–81. https://doi.org/10.1007/s11258-008-9566-z
Traniello JF (1989) Foraging strategies in ants. Ann Rev Entomol 34:191–210. https://doi.org/10.1146/annurev.en.34.010189.001203
van Oudenhove L, Billoir E, Boulay R, Bernstein C, Cerdá X (2011) Temperature limits trail following behaviour through pheromone decay in ants. Naturwissenschaften 98:1009–1017. https://doi.org/10.1007/s00114-011-0852-6
Vele A, Holusa J, Frouz J (2009) Ecological requirements of some species of the genus Formica (Hymenoptera, Formicidae) in spruce forests. J Forest Sci 55:32–40. https://doi.org/10.17221/56/2008-JFS
Vepsäläinen K, Savolainen R (1990) The effect of interference by Formicine ants on the foraging of Myrmica. J Anim Ecol 59:643–654
Wehner R, Müller M (2006) The significance of direct sunlight and polarized skylight in the ant’s celestial system of navigation. PNAS 103:12575–12579
Yilmaz A, Aksoy V, Camlitepe Y, Giurfa M (2014) Eye structure, activity rhythms, and visually-driven behavior are tuned to visual niche in ants. Front Behav Neurosci 8:205. https://doi.org/10.3389/fnbeh.2014.00205
Zakharov AA (2015) Ants of forest communities, their life cycle and role in forests. KMK Scientific Press, Moscow (in Russian)
Acknowledgements
The research leading to this publication has received funding from “The support of the priority research areas development of Ukraine, KPKVK 6541230” (for SS). IM was also supported by the János Bolyai Research Scholarship of the Hungarian Academy of Sciences (BO/00708/22) and the New National Excellence Program of the Ministry for Culture and Innovation (ÚNKP-22-5 -SZTE-581). The publication of the paper was supported by the University of Szeged Open Access Fund, Grant number 6591.
Funding
Open access funding provided by University of Szeged.
Author information
Authors and Affiliations
Contributions
SS designed the study and collected the data. IEM analysed the data and prepared the figures. SS and IEM wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stukalyuk, S., Maák, I.E. The influence of illumination regimes on the structure of ant (Hymenoptera, Formicidae) community composition in urban habitats. Insect. Soc. 70, 423–437 (2023). https://doi.org/10.1007/s00040-023-00935-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-023-00935-2