Abstract
Objectives
This study assessed the awareness and use of traditional and novel tobacco products and dual use of cigarettes with electronic nicotine delivery systems (ENDS) among USA adults.
Methods
Data were obtained from the 2014 Tobacco Products and Risk Perceptions Survey of a probability sample of 5717 USA adults conducted June–November, 2014.
Results
Use of ENDS varied by demography and by cigarette and other tobacco use. Adults aged 25–34, non-heterosexual adults, and those reporting poorer health reported higher rates of current ENDS use. Current cigarette smokers had much greater odds of ENDS ever use than never smokers, with one-half of all cigarette smokers having used ENDS and 20.7 % currently using them. However, 22.0 % of current ENDS users were former cigarette smokers, and 10.0 % were never cigarette smokers.
Conclusions
Patterns of ENDS use are evolving rapidly and merit continued surveillance. Nearly 10 % of adult ENDS usage is among never smokers. The public health challenge is how to enhance the potential that ENDS can replace combusted tobacco products without expanding nicotine use among youth, long-term ex-smokers, and other vulnerable populations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The USA appears to be entering a period of dramatic transformation of the types of tobacco products commonly used in the USA, potentially as dramatic as when the “modern” cigarette replaced the cigar and chewing tobacco 100 years ago, and when the filtered and “low-tar” cigarette transformed the market 40–60 years ago (US Department of Health and Human Services 2014). The latter transformation was driven by the increased knowledge of the health risks of cigarettes (e.g., 1964 Surgeon General Report) but produced a shift in product that did not reduce population harm and increased individual risk for some tobacco-related diseases such as adenocarcinoma of the lung (US Department of Health and Human Services 2014). As societal knowledge about the harm caused by traditional cigarettes has continued to increase, the number and type of tobacco and nicotine products available to consumers have increased dramatically in recent years. The current array of novel and alternative products includes non-combustible, smokeless, spit-less tobacco products such as snus and dissolvables, which differ in form from conventional chewing tobacco (Bahreinifar et al. 2013; Seidenberg et al. 2012); little cigars and cigarillos (LCCs), which differ from their larger, conventional cigar counterparts; water pipes or hookahs; and an increasing variety of electronic vapor products (ENDS), known most commonly as e-cigarettes (Agaku and Alpert 2015; Agaku et al. 2014; McMillen et al. 2014). ENDS vary widely in appearance and functionality, but all are battery-powered and can deliver combinations of nicotine and other additives in an aerosol (Knorst et al. 2014; Orellana-Barrios et al. 2015).
Awareness of many of these novel and alternative products appears to be significant and rising steadily among adults in the USA (Choi and Forster 2013; King et al. 2015). Studies conducted on the prevalence of use for these products indicate some volatility in popularity of the newer smokeless products (Biener et al. 2014; Zhu et al. 2013) but substantial and rising prevalence rates for little cigars and cigarillos among certain subgroups (Cohn et al. 2015; Corey et al. 2014; Messer et al. 2015; Richardson et al. 2013), hookahs (Brockman et al. 2012; Villanti et al. 2015), and, especially, for ENDS (Agaku et al. 2014; Carroll Chapman and Wu 2014; King et al. 2015; McMillen et al. 2014; Pepper and Brewer 2014).
High rates of smoking-attributable disease and death, and the associated health and societal costs, have been predicted to persist for decades into the 21st century (US Department of Health and Human Services 2014). These disturbing predictions have prompted some to encourage this current pattern of tobacco product transformation, hoping that the novel products could develop into a much lower risk “disruptive technology” that replaces the most lethal forms of nicotine delivery: the cigarette and the other inhalable combusted products (Abrams 2014; Cobb and Abrams 2014; Fagerstrom and Bridgman 2014; West and Brown 2014). However, others have raised cautions about how these new products could produce harms to population health (Chapman 2015; Chen and Husten 2014; England et al. 2015; Grana et al. 2014; Schraufnagel et al. 2014).
As of October, 2015, the Food and Drug Administration lacks the authority to regulate several novel tobacco products, notably ENDS marketed for non-therapeutic purposes (Chen and Husten 2014). Despite meeting the definition of a “tobacco product” under the Family Smoking Prevention and Tobacco Control Act (FSPTCA), the act only granted the FDA immediate authority over cigarettes, smokeless tobacco, and roll-your-own tobacco (Chen and Husten 2014). However, the FSPTCA allows the FDA to promulgate regulations extending regulatory authority over all other products meeting the definition of tobacco product (Lindblom 2015). The FDA needs current information on rates and trends of use of these products to guide and support its rule making and regulatory actions (or lack thereof).
In summary, the public health challenge is how to balance the public health messaging about these alternative and novel tobacco products and to enhance the potential that they can become a “disruptive technology” replacing the combusted tobacco products without expanding the patterns of nicotine use among youth and young adults, long-term ex-smokers, and other vulnerable populations. Information on how these alternative and novel tobacco products are being used is critically needed as the marketing and use of these products are rapidly increasing (Emery et al. 2014; Pepper et al. 2015). To address these needs, data from the 2014 Tobacco Products and Risk Perceptions Survey are presented here assessing the awareness and use of ENDS and other novel or alternative tobacco products among USA adults, demographic predictors of use, and dual use of combusted tobacco products and ENDS. We are not aware of any other published prevalence estimates of USA adult ENDS use from 2014. Together, these results will help inform public health and regulatory decisions about the rapidly evolving ENDS products.
Methods
Data source
We obtained data for this study from the 2014 Tobacco Products and Risk Perceptions Survey conducted by the Georgia State University Tobacco Center of Regulatory Science (TCORS). This survey is an annual, cross-sectional survey of a probability sample drawn from Gfk’s KnowledgePanel, a probability-based web panel designed to be representative of non-institutionalized USA adults. Only adults sampled via address-based sampling or random digit dialing (previous) are eligible to join KnowledgePanel. Recruited panelists without internet access are provided a computer with internet access. Information from the profile survey is used to calculate a panel demographic post-stratification weight to adjust for sources of sampling and non-sampling error, such as panel recruitment non-response and panel attrition. Data collection occurred June–November, 2014. Participants completed the main survey in 23 min (median) and received a cash-equivalent of $5 for their participation. This study was approved by the Georgia State University Institutional Review Board.
Sample
A probability sample of USA adults from KnowledgePanel and a representative oversample of pre-identified cigarette smokers were selected with probabilities proportional to size (PPS) after application of the panel demographic post-stratification weight. Overall, we invited 7991 KnowledgePanel members to participate in the survey: 7061 members for the general population sample, of which 74.3 % completed the screener survey and qualified for the main survey; and 930 members for the smoker augment sample, of which 697 completed the screener and 599 (74.9 %) qualified for the main survey by confirming their current smoking status. Of 5833 qualified completers, 116 cases were excluded due to refusing to answer more than one-half of the survey questions, yielding an analytic sample of 5717 cases. A final stage completion rate of 74.4 % and a qualification rate of 98.2 % were obtained. The average panel recruitment rate for this study, reported by GfK, was 13.7 % and the average profile rate was 65.3 %, for a cumulative response rate of 6.6 %. Following closure of the main survey field period, a study-specific post-stratification weight was computed using an iterative proportional fitting (raking) procedure to adjust for survey non-response as well as for oversampling of smokers. Demographic and geographic distributions from the most recent Current Population Survey (CPS) were employed as benchmarks for adjustment, and included gender, age, race/ethnicity, education, household income, census region, metropolitan area, and internet access.
Measures
Smoking status
Respondents that reported not having smoked at least 100 cigarettes in their lives were classified as never (established) smokers. Those respondents who reported smoking at least 100 cigarettes in their lives were asked, “Do you currently smoke cigarettes every day, some days, or not at all?” They were classified as current smokers if they reported currently smoking cigarettes “every day” or “some days” and as former smokers if they reported “not at all”. Recent former smokers were defined as former smokers who reported the last time they smoked a cigarette, even one or two puffs, was within the past 5 years, and non-recent former smokers were defined as former smokers who last smoked a cigarette more than 5 years ago.
Awareness and use of ENDS and other tobacco products
Awareness and use of ENDS and other tobacco products (namely, chewing tobacco, snuff, or dip; large, premium cigars; LCCs; snus; dissolvables; and hookahs) were assessed by asking respondents if they had heard of the product before taking the survey and, if so, whether they had ever tried the product, even just one time. Those respondents who indicated they have tried one or more of the products were asked whether they had used the products at least once during the past 30 days. Current users of these products were defined as those who had used the product at least once during the past 30 days. For some analyses, ever (non-current) users were defined as those who reported ever use of the product but no use in the past 30 days.
Prior to the questions assessing awareness and use of these products, respondents were shown descriptions and images of ENDS and LCCs. The description for ENDS used “e-cigarette” and referred broadly to electronic nicotine delivery systems. The description provided for LCCs characterized them as smaller than traditional cigars, usually brown, and listed several common brands.
Respondent characteristics
Demographic and other respondent characteristics data were obtained from profile surveys administered by GfK to KnowledgePanel panelists. Respondent characteristic included sex, age, race/ethnicity, educational attainment, annual household income, USA Census region, perceived health status, sexual orientation, and presence of a child in the home.
Statistical analysis
We used Stata/MP (v.13.1) to obtain design-based (weighted) point estimates and 95 % confidence intervals for awareness and use of ENDS, combustible, and non-combustible tobacco products. Associations among variables were tested using weighted logistic regression models and Rao–Scott χ 2 tests (Rao and Scott 1981). Prior to conducting these analyses, we assessed the extent and ignorability of missing data for ever use and past 30-day use questions for the tobacco products. Pearson Chi-square tests of the missing completely at random (MCAR) (Fuchs 1982) assumption were conducted using Mplus (v.7.3) and were non-significant (ps > 0.99). As an additional check, full information maximum likelihood estimates of the weighted proportions of using each product under the missing at random (MAR) assumption were compared to the corresponding MCAR estimates. Differences in estimates were less than 0.5 %. On the bases of these checks, respondents with missing data were excluded from further analyses under the supported assumption that missingness is ignorable and completely at random. (See supplemental document for an expanded summary of the missing data.)
Results
Table 1 reports awareness, ever use, and current use of ENDS and other novel and alternative tobacco products overall and by cigarette smoking status. Table 2 reports ever and current use of ENDS and current smoking by demography. Table 3 reports the adjusted associations between cigarette smoking status, other combustible tobacco use, and other non-combustible tobacco use with ENDS use. A detailed summary of the sample and reference population demographics and ENDS use by demography for emerging adults (18–24 years) and adults over 25 years old can be found in the supplemental document.
Awareness and use of electronic nicotine delivery systems (ENDS)
Overall, 91.9 % of USA adults had heard of ENDS. Our study estimated 14.9 % of USA adults have ever used ENDS and 4.9 % were current users. Use was substantially higher among current cigarette smokers compared to former and never smokers. Among current cigarette smokers, the odds of ever (OR = 21.4, 95 % CI = 16.5, 27.8) and current use (OR = 29.7, 95 % CI = 17.3, 51.0) of ENDS were substantially higher than for never smokers. Of those who have ever tried ENDS, 57.7 % (95 % CI = 53.7, 61.5) were current cigarette smokers, 25.3 % (95 % CI = 22.0, 28.9) were former smokers, and 17.0 % (95 % CI = 14.0, 20.6) were never smokers.
Ever and current use of ENDS is highest among young adults, particularly 25–34 year olds; those with some college education; those with less than a high school educational attainment; those with only fair or poor perceived health; and those with a non-heterosexual orientation. Young adults (25–34 years old) were more likely (OR = 1.7, 95 % CI = 1.0, 2.9) and those with a college degree were less likely (OR = 0.5, 95 % CI = 0.3, 0.7) to be current ENDS users. Adults with incomes between $40,000 and $60,000, $60,000 and $85,000, and greater than $100,000 had lower odds of current ENDS use (ORs = 0.3–0.6). Current ENDS use was less likely among those reporting good or better perceived physical health (OR = 0.5, 95 % CI = 0.3, 0.6), although this association was non-significant when controlling for cigarette smoking (AOR = 0.72, 95 % CI = 0.49, 1.05). Sex, race/ethnicity, presence of children in the home, and USA Census region were not associated with ENDS use in bivariate analyses. Sexual orientation was not associated with current ENDS use, though an association was observed for ever use. Non-heterosexual adults had higher odds (OR = 1.9, 95 % CI = 1.4, 2.6) of ever using ENDS. This association remained significant when controlling for smoking status (AOR = 1.8, 95 % CI = 1.2, 2.6).
Awareness and use of other tobacco products
In 2014, an estimated 16.6 % (95 % CI = 15.6, 17.6) of USA adults were current cigarette smokers and 27.6 % (95 % CI = 26.3, 28.9) were former cigarette smokers. We observed high awareness of other combustible tobacco products (Table 1), and nearly two-thirds (62.3 %, 95 % CI = 60.9, 63.8) of USA adults were ever users and 21.6 % (95 % CI = 20.4, 22.8) were current users of any combustible tobacco product. Use of these other combustible products varied by cigarette smoking status. Nearly one-third of all respondents reported ever using LCCs. Ever use of LCCs was more likely among current or former cigarette smokers, and current users were more likely to be current cigarette smokers. More than one in ten respondents has ever used hookahs, with use being higher among current and former cigarette smokers. Few adults reported current hookah use (1.2 %). Approximately one-quarter of respondents have ever smoked large cigars, with ever use being more common among former cigarette smokers. Current use of large cigars was reported by few adults (1.8 %). Whereas USA adults reported very high awareness of traditional smokeless tobacco (93.8 %), their awareness of snus and dissolvables was comparatively low (59.7 and 40.6 %, respectively). Nearly one-fifth of USA adults (17.9 %) have ever used traditional smokeless tobacco, and 2.1 % are current users. Snus use was less prevalent, with 4.0 % reporting ever use and only 0.3 % reporting current use. Dissolvables were the least prevalent non-combustible tobacco product examined in this study; only 1.1 % of USA adults reported ever using the product. Current use of dissolvables was too low to obtain reliable estimates with our sample. Non-combustible tobacco use was more prevalent among current and former cigarette smokers compared to never smokers. Current cigarette smokers (OR = 2.9, 95 % CI = 2.3, 3.5) and former smokers (OR = 3.2, 95 % CI = 2.7, 3.8) were more likely to ever use traditional smokeless tobacco than never smokers. Current smokeless tobacco use was greater among current cigarette smokers (OR = 2.5, 95 % CI = 1.5, 4.2) and former smokers (OR = 1.7, 95 % CI = 1.0, 2.9). Regarding snus, ever use was several times greater among current cigarette smokers (OR = 9.0, 95 % CI = 5.9, 13.8) and former smokers (OR = 3.9, 95 % CI = 2.5, 6.2) than never smokers. Similar associations with smoking status were observed for current use of snus use and ever use of dissolvables.
Associations between ENDS use and other tobacco use
Adjusting for other tobacco use, current smokers had nearly 18 times greater odds of ever use of ENDS and nearly 23 times the odds of current use of ENDS than did never smokers (Table 3, Model 1). Former smokers had twice the odds of ever ENDS use and 3.5 times the odds of current ENDS use as never smokers. These associations held and, in fact, were stronger, when statistically controlling for demographic and health status differences (Model 2). Other combustible and non-combustible use was also predictive of ENDS use. Ever use of any other combustible tobacco products (namely, LCCs, large cigars, and hookahs) was associated with 3.9 times greater odds of ever ENDS use than never use of these other combustible products. Ever users of non-combustible tobacco products (namely, smokeless tobacco, snus, and dissolvables) had 1.5 times greater odds of ever ENDS use than never users of these non-combustible tobacco products. Other combustible tobacco use was also predictive of current ENDS use. Adjusting for smoking status and non-combustible tobacco use, current users of non-combustible tobacco products had 4.4 times the odds and former use was associated with 1.8 times greater odds of current ENDS use than never users of these alternative combustible products. There was no statistically significant association between non-combustible tobacco product use and current ENDs use. These patterns of findings also remained and effect sizes undiminished after statistical adjustments were made for the demographic and health status covariates. After adjusting for other tobacco use and other covariates, sex, age, race/ethnicity, income, USA region, and perceived health status were significantly associated with ever use of ENDS and/or with current use of ENDS.
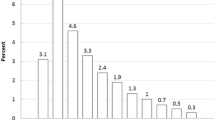
The findings from the logistic regression models are suggestive of high rates of dual use of ENDS and conventional cigarettes. Of those who were current ENDS users, 68.0 % were current cigarette smokers, 19.7 % were recent former smokers (quit within past 5 years), 2.3 % were non-recent former smokers (quit more than 5 years ago), and 10.0 % were never cigarette smokers (Fig. 1). Among current ENDS users, 21.0 % were current users of any alternative combustible tobacco product (namely, LCCs, large cigars, and/or hookah) and 75.1 % were current users of any combustible tobacco product (namely, LCCs, large cigars, hookah, and/or cigarettes).
Tobacco product use among current users of electronic nicotine delivery systems in the USA, 2014 (N = 316). Former cigarette smokers were classified as recent former smokers if they report last cigarette use within the past 5 years and as non-recent former smokers if they reported last cigarette use more than 5 years ago. The bars shaded in gray represent mutually exclusive and exhaustive groups and sum to 100 % of current ENDS users. Current users of any other combustible include those who reported past 30-day use of little cigars or cigarillos, large cigars, or hookah. Current users of any combustible include those who reported any combustible tobacco use in the past 30 days or current cigarette smoking someday or everyday. Error bars depict 95 % confidence intervals. RSE relative standard error. †30 % ≤ RSE ≤ 49 %
Discussion
The findings of this study provide current data about the awareness and use of ENDS and other novel tobacco products. Of particular interest, an estimated 1 in 20 USA adults, including 1 in 4 current cigarette smokers and 1 in 25 former smokers, were current ENDS users. These estimates are more than two times higher than 2012/2013 national estimates reported in a recently published study (King et al. 2015), although slightly lower than 2013 estimates from another study (McMillen et al. 2014). Of the alternative tobacco products assessed, ENDS had the highest prevalence of current use.
To a large degree, the observed patterns are being driven by market forces rather than by public health. The tobacco industry and the financial markets are noting that the tobacco product markets are entering a period of potentially dramatic product innovation and transformation (Herzog et al. 2015; Philip Morris International 2015). Published commentaries and editorials suggest that if public policy encourages these changes, the tobacco epidemic could be transformed and dramatically reduced (Abrams 2014; Cobb and Abrams 2014; Fagerstrom and Bridgman 2014; West and Brown 2014). The 2014 Surgeon General report acknowledged that additional “endgame strategies” could be needed to avert the projected and sustained pattern of smoking-attributable disease and premature death, but also noted, based upon the history of past tobacco industry driven product transformations, how the availability and promotion of these new nicotine delivery products could be projected either to reduce or increase population harm (US Department of Health and Human Services 2014). This potential impact that these products could have on population health was recently estimated in a dynamic population model (Vugrin et al. 2015). Recognizing that there was significant uncertainty in defining and selecting values for input parameters, the results showed how sensitive the model results were to (1) tobacco-related health risks of the various new and traditional products, (2) the rates of initiation of traditional and new products, and (3) the rates of switching and complete cessation, versus (4) sustained dual use of the novel products along with traditional cigarettes (Vugrin et al. 2015).
The results from this 2014 Tobacco Products and Risk Perceptions Survey and future annual surveys will provide nationally representative parameter estimates to inform such modeling of population harms. Specifically, the current data provide details about the manner in which ENDS are being combined (i.e., “dual use”) not only with cigarettes but with other combusted tobacco products. These high rates of dual use merit close monitoring to see if they could evolve into cessation of combusted tobacco product use or sustained dual use that delays individuals who would have quit tobacco products completely from actually quitting (Chen and Husten 2014). Second, the fact that almost one-third of the current ENDS users were ex-smokers and never smokers also merits careful monitoring. Of the 22.4 % of current ENDS users who were former cigarette smokers, about one in ten were long-term (more than 5 years) ex-smokers. This pattern raises concerns that the current marketing and promotion of ENDS are contributing to increased nicotine use and renormalization of tobacco use. Future surveys need to monitor these patterns to see if ex-smokers using ENDS are using them to prevent relapse or if their re-initiation of nicotine use could lead to relapsing back to combusted tobacco (McMillen et al. 2014).
This survey also shows that little cigars and cigarillos (LCC) had the highest overall prevalence of ever use among the alternative tobacco products, although this ranking of prevalence did not hold across smoking status subpopulations. Among smokeless products, chewing tobacco/snuff/dip tobacco was most common and dissolvables the least common in ever and current use. While the risk of tobacco-related diseases is known to vary across these products (US Department of Health and Human Services 2014), the risks of cigars, particularly the little cigars/cigarillos that are used and inhaled like cigarettes may have risks as high or exceeding the risks from smoking cigarettes (Chang et al. 2015). Thus, the data on the patterns of the use of these individual novel or alternative tobacco products or their use in combination with other combustible tobacco products can guide what additional evaluations of tobacco-related risk and risk perceptions are most needed.
Limitations
This study has several limitations. First, the use of the internet panel may raise concerns about sample representativeness, especially if the panel has been used in prior tobacco research. Second, the data are based upon self-report, and biochemical verification of cigarette smoking and use of other products could not be conducted. While the validity of self-reported cigarette smoking has been confirmed (Caraballo et al. 2004; US Department of Health and Human Services 2014), the accuracy of self-report of other products, particularly the novel products, has not been evaluated and remains uncertain. Third, due to the rapidly changing nature of ENDS products being marketed and used, how accurately the questionnaire descriptions and terminology are assessing actual patterns of use is continuing to be evaluated.
Conclusions
The results from this survey in conjunctions with other published data highlight that the patterns of trial and use of ENDS are evolving rapidly and merit continued surveillance and study. Use of ENDS is more common among current and former cigarette smokers, with patterns of use of little cigars/cigarillos, hookah, large cigars, smokeless tobacco, and ENDS varying significantly by sex, age, race/ethnicity, education, income, perceived health status, and sexual orientation. While use of ENDS primarily is combined with cigarette smoking, more than one-tenth of ENDS usage is among never cigarette smokers. Additionally, the patterns of higher rates of dual use of ENDS and combustible tobacco products among the less educated, lower income, and those with poorer perceived health are of concern. If these rates of dual use in these vulnerable populations do not contribute to increased smoking cessation then, when combined with the continuing high rates of use of all combusted tobacco products, these data raise significant public health concerns that the growing prevalence of ENDS use could contribute to increased population harm. Thus, this survey provides the FDA with timely population-level data that may help inform FDA policy development. Once FDA authority is properly extended, this survey provides the agency with evidence to inform product standards for novel nicotine products appropriate for the protection of public health, a standard specifically focused on evaluating product risk and benefit at a population level (Chen and Husten 2014; Husten and Deyton 2013).
References
Abrams DB (2014) Promise and peril of e-cigarettes: can disruptive technology make cigarettes obsolete? JAMA 311:135–136
Agaku IT, Alpert HR (2015) Trends in annual sales and current use of cigarettes, cigars, roll-your-own tobacco, pipes, and smokeless tobacco among US adults, 2002-2012. Tob Control. doi:10.1136/tobaccocontrol-2014-052125
Agaku IT, King BA, Husten CG, Bunnell R, Ambrose BK, Hu SS, Holder-Hayes E, Day HR, Centers for Disease Control and Prevention (2014) Tobacco product use among adults–United States, 2012-2013. Morb Mortal Wkly Rep 63:542–547
Bahreinifar S, Sheon NM, Ling PM (2013) Is snus the same as dip? Smokers’ perceptions of new smokeless tobacco advertising. Tob Control 22:84–90. doi:10.1136/tobaccocontrol-2011-050022
Biener L, Roman AM, Mc Inerney SA, Bolcic-Jankovic D, Hatsukami DK, Loukas A, O’Connor RJ, Romito L (2014) Snus use and rejection in the USA. Tob Control. doi:10.1136/tobaccocontrol-2013-051342
Brockman LN, Pumper MA, Christakis DA, Moreno MA (2012) Hookah’s new popularity among US college students: a pilot study of the characteristics of hookah smokers and their Facebook displays. BMJ Open 2:e001709. doi:10.1136/bmjopen-2012-001709
Caraballo RS, Giovino GA, Pechacek TF (2004) Self-reported cigarette smoking vs. serum cotinine among US adolescents. Nicotine Tob Res 6:19–25. doi:10.1080/14622200310001656821
Carroll Chapman SL, Wu LT (2014) E-cigarette prevalence and correlates of use among adolescents versus adults: a review and comparison. J Psychiatr Res 54:43–54. doi:10.1016/j.jpsychires.2014.03.005
Chang CM, Corey CG, Rostron BL, Apelberg BJ (2015) Systematic review of cigar smoking and all cause and smoking related mortality. BMC Public Health 15:390. doi:10.1186/s12889-015-1617-5
Chapman S (2015) The future of electronic cigarette growth depends on youth uptake. Med J Aust 202:467–468
Chen I-L, Husten CG (2014) Introduction to tobacco control supplement. Tob Control 23(Suppl 2):ii1–ii3. doi:10.1136/tobaccocontrol-2013-051504
Choi K, Forster J (2013) Awareness, perceptions and use of snus among young adults from the upper Midwest region of the USA. Tob Control 22:412–417. doi:10.1136/tobaccocontrol-2011-050383
Cobb NK, Abrams DB (2014) The FDA, e-cigarettes, and the demise of combusted tobacco. N Engl J Med 371:1469–1471. doi:10.1056/NEJMp1408448
Cohn A, Cobb CO, Niaura RS, Richardson A (2015) The other combustible products: prevalence and correlates of little cigar/cigarillo use among cigarette smokers. Nicotine Tob Res. doi:10.1093/ntr/ntv022
Corey CG, King BA, Coleman BN, Delnevo CD, Husten CG, Ambrose BK, Apelberg BJ, Centers for Disease Control and Prevention (2014) Little filtered cigar, cigarillo, and premium cigar smoking among adults–United States, 2012-2013. Morb Mortal Wkly Rep 63:650–654
Emery SL, Vera L, Huang J, Szczypka G (2014) Wanna know about vaping? Patterns of message exposure, seeking and sharing information about e-cigarettes across media platforms. Tob Control 23(Suppl 3):iii17–iii25. doi:10.1136/tobaccocontrol-2014-051648
England LJ, Bunnell R, Pechacek TF, Tong VT, McAfee T (2015) Nicotine and the developing human: A neglected element in the electronic cigarette debate. Am J Prev Med In press. doi:10.1016/j.amepre.2015.01.015
Fagerstrom KO, Bridgman K (2014) Tobacco harm reduction: the need for new products that can compete with cigarettes. Addict Behav 39:507–511. doi:10.1016/j.addbeh.2013.11.002
Fuchs C (1982) Maximum likelihood estimation and model selection in contingency tables with missing data. J Am Stat Assoc 77:270–278
Grana RA, Benowitz N, Glantz SA (2014) E-cigarettes: a scientific review. Circ J Am Heart Assoc 129:1972–1986. doi:10.1161/CIRCULATIONAHA.114.007667
Herzog B, Gerberi J, Scott A (2015) Tobacco Talk: independent vapor Mfr. survey. Wells Fargo Securities, LLC, Equity Research Department, New York
Husten CG, Deyton LR (2013) Understanding the Tobacco Control Act: efforts by the US Food and Drug Administration to make tobacco-related morbidity and mortality part of the USA’s past, not its future. Lancet 381:1570–1580. doi:10.1016/S0140-6736(13)60735-7
King BA, Patel R, Nguyen KH, Dube SR (2015) Trends in awareness and use of electronic cigarettes among US adults, 2010-2013. Nicotine Tob Res 17:219–227. doi:10.1093/ntr/ntu191
Knorst MM, Benedetto IG, Hoffmeister MC, Gazzana MB (2014) The electronic cigarette: the new cigarette of the 21st century? J Bras Pneumol 40:564–572
Lindblom EN (2015) Effectively regulating e-cigarettes and their advertising—and the first amendment. Food Drug Law J 70:57–94
McMillen RC, Gottlieb MA, Shaefer RM, Winickoff JP, Klein JD (2014) Trends in electronic cigarette use among US adults: use is increasing in both smokers and nonsmokers. Nicotine Tob Res. doi:10.1093/ntr/ntu213
Messer K, White MM, Strong DR, Wang B, Shi Y, Conway KP, Pierce JP (2015) Trends in use of little cigars or cigarillos and cigarettes among US smokers, 2002–2011. Nicotine Tob Res 17:515–523. doi:10.1093/ntr/ntu179
Orellana-Barrios MA, Payne D, Mulkey Z, Nugent K (2015) Electronic cigarettes-a narrative review for clinicians. Am J Med. doi:10.1016/j.amjmed.2015.01.033
Pepper JK, Brewer NT (2014) Electronic nicotine delivery system (electronic cigarette) awareness, use, reactions and beliefs: a systematic review. Tob Control 23:375–384. doi:10.1136/tobaccocontrol-2013-051122
Pepper JK, Emery SL, Ribisl KM, Rini CM, Brewer NT (2015) How risky is it to use e-cigarettes? Smokers’ beliefs about their health risks from using novel and traditional tobacco products. J Behav Med 38:318–326. doi:10.1007/s10865-014-9605-2
Philip Morris International (2015) Annual meeting of shareholds. http://investors.pmi.com/phoenix.zhtml?c=146476&p=irol-eventDetails&EventId=5125408. Accessed May 6 2015
Rao JN, Scott AJ (1981) The analysis of categorical data from complex sample surveys: Chi squared tests for goodness of fit and independence in two-way tables. J Am Stat Assoc 76:221–230
Richardson A, Rath J, Ganz O, Xiao H, Vallone D (2013) Primary and dual users of little cigars/cigarillos and large cigars: demographic and tobacco use profiles. Nicotine Tob Res 15:1729–1736. doi:10.1093/ntr/ntt053
Schraufnagel DE, Blasi F, Drummond MB, Lam DC, Latif E, Rosen MJ, Sansores R, Van Zyl-Smit R, Forum of International Respiratory Societies (2014) Electronic cigarettes. A position statement of the forum of international respiratory societies. Am J Respir Crit Care Med 190:611–618. doi:10.1164/rccm.201407-1198PP
Seidenberg AB, Rees VW, Connolly GN (2012) R. J. Reynolds goes international with new dissolvable tobacco products. Tob Control 21:368–369. doi:10.1136/tobaccocontrol-2011-050116
US Department of Health and Human Services (2014) The health consequences of smoking–50 years of progress: a report of the Surgeon General. US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, Atlanta, GA
Villanti AC, Cobb CO, Cohn AM, Williams VF, Rath JM (2015) Correlates of Hookah use and predictors of Hookah trial in US young adults. Am J Prev Med. doi:10.1016/j.amepre.2015.01.010
Vugrin ED, Rostron BL, Verzi SJ, Brodsky NS, Brown TJ, Choiniere CJ, Coleman BN, Paredes A, Apelberg BJ (2015) Modeling the potential effects of new tobacco products and policies: a dynamic population model for multiple product use and harm. PLoS One 10:e0121008. doi:10.1371/journal.pone.0121008
West R, Brown J (2014) Electronic cigarettes: fact and faction. Br J Gen Pract 64:442–443. doi:10.3399/bjgp14X681253
Zhu SH, Gamst A, Lee M, Cummins S, Yin L, Zoref L (2013) The use and perception of electronic cigarettes and snus among the US population. PLoS One 8:e79332. doi:10.1371/journal.pone.0079332
Acknowledgments
The authors acknowledge Meghan Nix for technical assistance in preparation of this manuscript. Research reported in this publication was supported by Grant Number P50DA036128 from the National Institutes of Health, National Institute of Drug Abuse (NIH/NIDA) and Food and Drug Administration, Center for Tobacco Products (FDA CTP). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH or the Food and Drug Administration. Drs. Pechacek and Eriksen receive research funding support from Pfizer, Inc. (“Diffusion of Tobacco Control Fundamentals to Other Large Chinese Cities” Michael Eriksen, Principal Investigator). No financial disclosures were reported by the other authors of this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no other potential conflicts of interest.
Additional information
This article is part of the special issue “Electronic Cigarettes and Public Health”.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Weaver, S.R., Majeed, B.A., Pechacek, T.F. et al. Use of electronic nicotine delivery systems and other tobacco products among USA adults, 2014: results from a national survey. Int J Public Health 61, 177–188 (2016). https://doi.org/10.1007/s00038-015-0761-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00038-015-0761-0