Abstract
Objectives
To evaluate educational inequalities in diabetes mortality in Europe in the 2000s, and to assess whether these inequalities differ between genders.
Methods
Data were obtained from mortality registries covering 14 European countries. To determine educational inequalities in diabetes mortality, age-standardised mortality rates, mortality rate ratios, and slope and relative indices of inequality were calculated. To assess whether the association between education and diabetes mortality differs between genders, diabetes mortality was regressed on gender, educational rank and ‘gender × educational rank’.
Results
An inverse association between education and diabetes mortality exists in both genders across Europe. Absolute educational inequalities are generally larger among men than women; relative inequalities are generally more pronounced among women, the relative index of inequality being 2.8 (95 % CI 2.0–3.9) in men versus 4.8 (95 % CI 3.2–7.2) in women. Gender inequalities in diabetes mortality are more marked in the highest than the lowest educated.
Conclusions
Education and diabetes mortality are inversely related in Europe in the 2000s. This association differs by gender, indicating the need to take the socioeconomic and gender dimension into account when developing public health policies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
An individual’s disease risk cannot be separated from the population groups to which (s)he belongs (Elstad 2000). Important group-defining characteristics in this respect are socioeconomic position (SEP) and gender, with gender referring to the socially generated aspects of being a man/woman (Annandale 1998). Health and mortality differ systematically across SEP groups and gender. In contemporary western societies, the majority of health indicators are poorer among population groups with disadvantaged SEPs (Dalstra et al. 2008; Mackenbach et al. 2004). For gender, the picture is less univocal. Although women tend to be ill more often than men, they live longer. Despite biological factors playing a role, research has demonstrated that these differences are mainly socially generated (Case and Paxson 2005).
As for type 2 diabetes mellitus, there is an inverse gradient between SEP and both diabetes incidence and prevalence (Agardh et al. 2011; Espelt et al. 2008, 2013; Sacerdote et al. 2012), with some research reporting a rise in the magnitude of SEP inequalities in recent decades (Imkampe and Gulliford 2011; Smith 2007). The pattern is less clear for diabetes mortality, although most research does find an inverse association with SEP (Espelt et al. 2008; Forssas et al. 2003; Gnavi et al. 2004; Koskinen et al. 1996; Roper et al. 2001; Vandenheede et al. 2013). Furthermore, the majority of studies demonstrate a similar incidence and prevalence of type 2 diabetes mellitus in men and women (Gale and Gillespie 2001; Tang et al. 2003; Wild et al. 2004). This relative gender balance in incidence and prevalence does not hold for diabetes mortality. There is a clear preponderance of male diabetes deaths (Espelt et al. 2008; Romon et al. 2008).
Mounting evidence indicates that gender and SEP, as well as other social positions, do not operate in a vacuum, but often interact with each other (Macintyre and Hunt 1997; Sen et al. 2009). These interactive processes are not invariable, but vary within and between societies and over time (Macintyre and Hunt 1997), resulting in divergent implications for the design and development of public health policies (Sen et al. 2009). Although a burgeoning body of research shows that relative SEP inequalities in type 2 diabetes mellitus incidence, prevalence and mortality (Agardh et al. 2011; Dasgupta et al. 2010; Espelt et al. 2008, 2012, 2013; Robbins et al. 2001, 2005; Tang et al. 2003) are more pronounced in women relative to men, to the best of our knowledge, no study thus far has focused explicitly on the interaction between gender and SEP in relation to diabetes mortality. Assessing these associations may allow intricate insight into the mechanisms producing gender and SEP inequalities.
One way to gain insight into the mechanisms producing inequalities is cross-country research. To the extent that gender and SEP differences in diabetes mortality vary between countries, country characteristics are likely to be important determinants of these inequalities. Alternatively, finding no variation between countries would point to the universal nature of the phenomenon. The lion’s share of studies, assessing the relationship between SEP and/or gender and diabetes mortality, includes only one country (e.g., Gnavi et al. 2004; Landman et al. 2013; Romon et al. 2008; Vandenheede et al. 2013). So far we know, there is only one study that does compare SEP inequalities in diabetes mortality across Europe (Espelt et al. 2008). Using data from the 1990s and the beginning of the 2000s, Espelt et al. (2008) found an inverse educational gradient in diabetes mortality across Europe in men as well as in women, with gradients being particularly pronounced in women. The first objective of this study is to corroborate these results using more recent data from the 2000s on the one hand, and including countries and regions for which data were previously unavailable on the other (Austria, England and Wales, the Basque County, Madrid, Tuscany and Hungary). The second objective is to assess whether there is an interaction between gender, SEP and diabetes mortality. Probing into these associations may deepen our understanding of the mechanisms producing gender and SEP inequalities and, ultimately, open up new paths for diabetes prevention and care.
Methods
Design and study population
Table 1 describes the characteristics of the mortality studies included in the analyses. Data were gathered as part of the EURO-GBD-SE project. Both longitudinal (Denmark, Finland, Norway, Sweden, England and Wales, Austria, Belgium, Switzerland, the Basque Country, Madrid, Turin and Tuscany) and cross-sectional studies (Barcelona, Estonia, Czech Republic, Hungary and Poland) were used for this project. All longitudinal mortality datasets and the data from Barcelona consisted of linkage of census or population register data with the mortality register. In some countries (regions) (Austria, Madrid, Barcelona and the Basque Country, respectively), linkage was less than 95 % complete, and a correction factor was applied to the number of deaths in these regions to account for this. All studies covered the entire national territory, except for the Spanish and Italian datasets, and included all subgroups of the population, except for the Swiss mortality dataset, which is limited to Swiss nationals. The number of diabetes deaths was derived from the respective mortality registers, whereas person-years at risk was based on census or population register information. Person-years at risk was calculated using the number of subjects alive at the beginning of the study period times the length of the study period in the unlinked cross-sectional studies, and using the sum of the number of subjects alive at the beginning and end of each follow-up year divided by 2 in most longitudinal and repeated cross-sectional studies. The study population consisted of 30- to 74-year-olds. All mortality data refer to the 2000s.
Variables
Mortality from diabetes mellitus was defined by the International Classification of Diseases (ICD). Most studies used the 10th revision of the ICD (codes E10–14). However, definition of diabetes mortality was based on ICD-9 (code 250) in the Turin and Tuscan mortality data, and in the Austrian mortality data for the year 2001. All studies applied the standard epidemiological practice of only using the underlying cause of death.
The variable education was used as an indicator of SEP. Education was categorised according to the International Standard Classification of Education (ISCED), version 1997: pre-primary, primary and lower secondary education (ISCED 0–2); upper secondary and post-secondary non-tertiary education (ISCED 3–4); and tertiary education (ISCED 5–6). The proportion of missing values on education ranged from 0 % for most datasets to 6 % for Switzerland. Gender was included in the analyses as a dummy variable. The variable age was included as a categorical variable (5-year age bands) in the direct standardisation analyses, and as a continuous variable (mid-age of the 5-year age groups) in the Poisson regression analyses. Sensitivity checks with age as a categorical variable in both types of analyses demonstrated the robustness of the results. In some of the longitudinal datasets (Denmark, Norway, Sweden, Belgium and Switzerland), information on age was available at baseline only; while, for the other longitudinal and all cross-sectional datasets, age at the time of death was recorded. In datasets with age at baseline, people were not allowed to move into the next age group as they grew older. Hence, mortality estimates in these populations were higher compared to the estimates that would be obtained if age at death was used. To ensure comparability between mortality estimates, an adjustment procedure was developed that corrects for the upward bias in datasets which use age at baseline. Since only mortality rates are biased, the adjustment method was only applied when calculating mortality rates and absolute differences. Please refer to Östergren et al. (2001) for a detailed description of the adjustment formula.
Data analysis
Cases with missing information on education were deleted from the analyses (complete-case analyses), and analyses were country and gender specific. To increase power, mortality data from Spain (Barcelona, the Basque Country and Madrid) and Italy (Turin and Tuscany) were grouped together.
First, to quantify the burden of diabetes mortality in each educational group, age-standardised mortality rates (ASMRs) and their 95 % confidence intervals (CIs) were computed, directly standardised to the 1976 European Standard Population.
Then, using age-adjusted Poisson regression models with person-years as the offset, both absolute and relative educational differences in diabetes mortality were estimated, allowing for a comprehensive picture of inequalities, with absolute inequalities being of more public health interest and relative inequalities being of more analytic interest. To measure absolute educational differences in diabetes mortality, the slope index of inequality (SII) was calculated. Relative educational differences were estimated through two different measures: the relative index of inequality (RII) and mortality rate ratios (MRRs). SII and RII represent the difference between the predicted diabetes mortality rates at the lower versus the higher end of the educational distribution. To estimate RII, a country- and gender-specific educational rank variable, ranging from 1 to 0 (lowest to highest end of the educational distribution), was calculated based on the ISCED-categorised education variable, and included in the Poisson models. SII was calculated using the following formula: \({{\left[ { 2\times {\text{ASMR}} \times \left( {{\text{RII}} - 1} \right)} \right]} \mathord{\left/ {\vphantom {{\left[ { 2\times {\text{ASMR}} \times \left( {{\text{RII}} - 1} \right)} \right]} {\left( {{\text{RII}} + 1} \right)}}} \right. \kern-0pt} {\left( {{\text{RII}} + 1} \right)}}\). As SII and RII account for differences in the distribution of education between countries and genders, they can be used for comparative purposes, but only on the condition of linearity between diabetes mortality rates and the educational rank variable (Wagstaff et al. 1991). Hence, in case of non-linearity, SIIs and RIIs are not presented. The MRRs represent the differences between the predicted diabetes mortality rates by education, using ISCED 5–6 as the reference group.
Next to these gender-specific analyses, age-adjusted Poisson regression models were fitted to test for interaction between gender, education and diabetes mortality. In addition to this, a pooled dataset, consisting of the data for all countries, was constructed to estimate the combined effect of education for all countries under study. Weights were assigned, so that the separate countries carried equal weight in the combined results.
Results
Tables 2 and 3 present an overview of ASMRs per country by education for men and women, respectively. The general picture is one of the higher burden of diabetes mortality among men. With the exception of England and Wales and Estonia, ASMRs are higher in men than women. Among men, ASMRs (per 100,000 person-years) are between 10 and 15 in most countries; whereas, among women, ASMRs are generally between 5 and 10. All countries combined 14 diabetes deaths per 100,000 person-years occur in men, whereas the number of diabetes deaths in women is 9 per 100,000. The burden of diabetes differs also by education: diabetes mortality is higher among the lower educated in both men and women. Notably, the burden of diabetes mortality is particularly small among higher educated women, with the country-combined ASMR for higher educated women (ISCED levels 5–6) being 4.2 (95 % CI 2.7–5.8) per 100,000.
There is an inverse association between education and diabetes mortality in all countries in both genders. In most countries, this association takes on the form of a gradient, as is the case in, for example, Danish men and Belgian women. In Danish men, ASMRs are 32.2 (95 % CI 30.1–34.3), 21.9 (95 % CI 20.1–23.6) and 12.7 (95 % CI 10.6–14.7) among the lowest (ISCED 0–2), mid- (ISCED 3–4), and highest educated (ISCED 5–6), respectively (Table 2). MRRs among Belgian women are 4.0 (95 % CI 2.6–6.2) for the lowest educated and 1.8 (95 % CI 1.2–2.9) for the mid-educated, relative to the highest educated women (Table 3). In other countries, such as England and Wales, there is an inverse association between education and diabetes mortality, where the ISCED 0–2 group has the highest mortality, but the relationship is not graded.
The interaction between gender and education
With the exception of Hungary and Poland, absolute educational inequalities are generally larger in men compared to women. For example, the SII among Norwegian men amounts to 14.4 per 100,000 person-years, while the SII among Norwegian women is 7.1 per 100,000 (Tables 2, 3). Overall, the SII is 13.0 in men and 12.1 in women. Absolute inequalities are slightly higher in men relative to women. On the other hand, relative educational inequalities are generally larger in women. For example, the RII among Hungarian women is 8.0 (95 % CI 4.6–16.1) versus 3.7 (95 % CI 3.1–4.5) among Hungarian men. For all countries combined, the estimated mortality risk in the lowest educated men is almost 3 times as high as in the highest educated men; while for women, this figure is nearly 5.
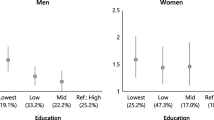
Table 4 focuses specifically on gender differences in educational inequalities in diabetes mortality. It presents the results from the age-adjusted Poisson regression models, which test for the interaction between gender, educational rank and diabetes mortality. Table 4 shows, on the one hand, that relative educational inequalities are more marked among women than men; on the other, that gender inequalities in diabetes mortality are more pronounced among the higher educated than the lower educated. This pattern is remarkably similar across countries. All countries combined, the burden of diabetes mortality in the highest educated women is 0.4 times that of the highest educated men, while the burden of diabetes mortality in the lowest educated women is 0.8 times (0.4 × 2.1) the burden in the lowest educated men.
Discussion
Summary of findings
This study indicates that education is inversely related to mortality from diabetes mellitus across Europe in both men and women. Absolute educational inequalities are generally larger in men than in women, reflecting men’s higher diabetes mortality. Relative educational inequalities in diabetes mortality are more marked among women than men. There is an interaction effect between education, gender and diabetes mortality in most European countries, with the burden of diabetes mortality being particularly small among the highest educated women. All countries combined, diabetes mortality among the highest educated women is 0.4 times that of the highest educated men, while diabetes mortality among the lowest educated women is 0.8 times that of the lowest educated men.
Methodological considerations
Data were derived from both longitudinal and cross-sectional mortality studies. All longitudinal data and data from the repeated cross-sectional study were linked. Yet, all datasets, from Eastern European and Baltic countries, used an unlinked study design. Unlinked data are more prone to numerator–denominator bias, since misclassification between the numerator (number of deaths, derived from mortality register) and the denominator (number of person-years, derived from census/population register) of the rates might occur. However, as educational and gender patterns in diabetes mortality do not differ markedly between linked and unlinked studies, the effect of numerator–denominator bias on the magnitude of inequalities is expected to be rather small.
Another methodological consideration relates to the analysis of cause-specific mortality data at a European level. Comparing cause-specific mortality between European countries inevitably raises questions concerning the comparability of cause-specific death registration. There may be incongruities between European countries in death certificate models; nature and amount of information entered; application of the rules for selection of underlying, intermediary and associated causes of death and other coding practices; ICD-revision used; implementation of automated coding systems (Meslé 2002). These discrepancies make it difficult to compare the absolute burden of cause-specific mortality between countries. When it comes to diabetes, some discrepancies may be rather minor. For example, a bridge-coding study, calculating cause-specific mortality estimates using both ICD-9 and ICD-10 indicated an excellent comparability for diabetes. There was a less than 1 % increase in diabetes deaths using ICD-10 instead of ICD-9 (Anderson et al. 2001). Additional cross-country comparison difficulties are related to ambiguities in the role of diabetes as a cause of death. Since diabetes is often considered part of a complex clinical picture, it is mostly registered as one of the contributing causes, not as the underlying cause of death. To the extent that the use of diabetes as an underlying/contributing cause differs between countries, cross-country comparisons of diabetes mortality are difficult to interpret (Jougla et al. 1992).
As in most other studies on diabetes mortality (Espelt et al. 2008; Gnavi et al. 2004; Roper et al. 2001), we were unable to distinguish between mortality from type 1 and mortality from type 2 diabetes mellitus. The bias introduced by this merging together is considered to be minimal, since approximately 90 % of all people with diabetes do have type 2 diabetes (Dawson 2009). The frequent registration of diabetes mellitus as contributing instead of underlying cause leads to a severe underestimation of the burden of diabetes mortality—by 150–400 % depending on the study population—because of the standard epidemiological practice of using the underlying cause of death only (Romon et al. 2008; Vandenheede et al. 2011).
On the condition that registration practices are the same across educational groups and genders within countries, relative differences are not affected. Therefore, we decided not to emphasise the differences between countries in ASMRs or SIIs, but instead to focus on the general pattern and on the differences between countries in RIIs. Relative differences in diabetes mortality as found in our analysis echo findings from incidence and prevalence data (Agardh et al. 2011; Espelt et al. 2008, 2013; Sacerdote et al. 2012). The inverse graded association between education and diabetes mortality reflects the educational pattern of diabetes incidence and prevalence (Agardh et al. 2011; Espelt et al. 2008, 2013; Sacerdote et al. 2012), indicating that differential diabetes mortality by education is not solely due to differential registration practices, but mainly reflects actual differences in diabetes mortality.
Several advantages and disadvantages are associated with the use of education as a measure of SEP. Main advantages are: (1) its universality, it is applicable to people regardless of age and working circumstances; (2) its comparability; (3) the relative ease with which it can be measured; and (4) its relative insensitivity to health-related selection, and thus to reverse causality (Galobardes et al. 2007). Despite its general applicability, education lacks a universal meaning, since its implications are related to gender, ethnicity, birth cohort, and social class, amongst other things. Other disadvantages are mainly associated with the fact that education captures only a part of SEP, i.e. knowledge-related skills and assets rather than material circumstances (Galobardes et al. 2007). Yet, as both education and diabetes mortality are strongly lifestyle related, education may be a particularly relevant indicator for studying SEP inequalities in diabetes mortality. Furthermore, despite being attained rather early in life, education has been shown to remain as a key indicator of SEP through the life course.
Theoretical considerations
Educational inequalities in diabetes mortality
In line with the most previous studies (Espelt et al. 2008; Tang et al. 2003; Vandenheede et al. 2013), a strong inverse association between education and diabetes mortality was found among both men and women. Our results are similar to the ones of Espelt et al. (2008), indicating that the educational patterning of diabetes mortality found in the 1990s largely persists in the 2000s. If there is any change between the two periods, it is an increase in the magnitude of the association among men. Other studies also report a rise in the magnitude of SEP inequalities in diabetes in recent decades (Imkampe and Gulliford 2011; Smith 2007). We also observed an inverse association between education and diabetes mortality in countries and regions for which data were previously unavailable.
Several explanations for SEP inequalities in diabetes mortality have been put forward. Analogous to explanations for educational differences in diabetes incidence and prevalence, lifestyle, and more specifically obesity, is considered to be a key intermediary factor (Espelt et al. 2013; Vandenheede et al. 2013). Furthermore, factors related to diabetes progression, such as access to and quality of diabetes care, metabolic control and complications, could also be important determinants (Bachmann et al. 2003). Since diabetes management is becoming ever more technical and demanding, and since higher educated groups are better equipped to take full advantage of these developments than lower educated groups, educational inequalities in diabetes management and mortality are likely to widen (Phelan et al. 2010; Smith 2007). Another explanation for the rather strong association of education with diabetes mortality could be related to attitudes towards health. While higher-SEP people are inclined to think about their future health risk, to consider themselves as having control over their health, and to be aware of the influence of lifestyle on health, lower-SEP people are less prone to do so (Wardle and Steptoe 2003). These SEP differences in lifestyle, diabetes progression factors and attitudes cannot be separated from the differences in life opportunities and material circumstances between SEP groups, which may themselves arise because of the macro socioeconomic and political context (CSDH 2010).
The enduring and consistent relationship between education and diabetes mortality in most European countries, despite huge variation in macro-economic and political characteristics, points to the persistent nature of health inequalities (Phelan et al. 2010). Furthermore, it suggests that it is difficult to breach these inequality patterns and develop policies that give equal chances to all. Despite educational patterns in diabetes mortality being similar across Europe, inequalities are of different magnitudes in different countries. Hence, it would be of importance for future research to include country variables in order to determine how country characteristics affect educational inequalities in diabetes mortality. Doing so, we can gain further insight into inequality-generating mechanisms and fine-tune policies to reduce these inequalities.
A consistent gender pattern
We observed a higher burden of diabetes mortality among men relative to women, supporting previous evidence (Espelt et al. 2008). A possible explanation for this observation is differences in diabetes progression, as diabetes incidence and prevalence are highly comparable between men and women (Gale and Gillespie 2001; Wild et al. 2004). Previous studies have demonstrated a differential pattern of complications between men and women (Abbate et al. 2012), differences in diabetes management and in use of health care (Krämer et al. 2012). Another possible explanation is that many deaths from diabetes involve cardiovascular complications. Since the background risk of mortality from cardiovascular disease is higher among men than women, mortality risks of people with diabetes may be higher among men than women.
A larger SEP gradient in diabetes incidence (Espelt et al. 2013) prevalence (Espelt et al. 2008, 2013) and mortality (Espelt et al. 2008) among women relative to men has been established in previous research. However, to our knowledge, none of these studies has examined the interaction between gender, SEP and diabetes mortality explicitly. The interaction can be examined from different perspectives: relative educational differences are larger among women than men, and gender differences are more pronounced among the highest versus the lowest educated. The larger relative inequalities among women compared to men may be related to the smaller overall diabetes mortality levels among women, since there is an empirical relationship between relative mortality inequalities and the overall mortality level. The lower the burden of mortality is, the higher relative differences generally are (Houweling et al. 2007). Furthermore, larger educational differences in diabetes mortality among women mirror larger educational inequalities in some highly prevalent chronic diseases such as cardiovascular disease (Dalstra et al. 2008), and have been explained by the more pronounced gradients in obesity and related lifestyle factors among women relative to men (Espelt et al. 2013). Among higher-SEP women, prevalence of obesity, one of the major risk factors of diabetes, is rather low. One of the main explanations given in the literature is that higher-SEP women apply stricter behavioural norms and thinness ideals than both men and lower-SEP women (Roskam et al. 2010). The very small burden of diabetes mortality among the highest educated women mirrors the obesity findings. Hence, the explanation may be rather similar as well. Highly educated women are a very health-conscious group. They are more committed to their health than both lower educated women and men, and engage more in preventive efforts (Annandale 1998). The very small burden of diabetes mortality among this group of women ultimately shows that diabetes mortality is highly preventable, and, hence, an unnecessary cause of death.
Conclusions and recommendations
There is a need for studies probing into the mechanisms behind the interaction between gender, education and diabetes mortality, and for research into the persistence of health inequalities over time and in different settings. The low burden of diabetes mortality among higher educated women suggests large possibilities for intervention (e.g., by prevention and treatment of obesity). The relatively high burden of diabetes mortality among higher educated as well as lower educated men indicates that there are multiple barriers in men to effectively engage in diabetes management and care. Future policies should aim at reducing these barriers, while simultaneously improving diabetes care for all. One possible track for both the improvement of diabetes care and the reduction of inequalities in diabetes care may be close cooperation between general practitioners and endocrinologists (“shared care”) (Gnavi et al. 2009).
References
Abbate R, Mannucci E, Cioni G, Fatini C, Marcucci R (2012) Diabetes and sex: from pathophysiology to personalized medicine. Intern Emerg Med Suppl 7:S215–S219
Agardh E, Allebeck P, Hallqvist J, Moradi T, Sidorchuk A (2011) Type 2 diabetes incidence and socio-economic position: a systematic review and meta-analysis. Int J Epidemiol 40:804–818
Anderson RN, Miniño AM, Hoyert DL, Rosenberg HM (2001) Comparability of cause of death between ICD-9 and ICD-10: preliminary estimates. Natl Vital Stat Rep 49:1–28
Annandale E (1998) Part II: contemporary health inequalities. In: Annandale E (ed) The sociology of health and illness: a critical introduction. Blackwell Publishers, Oxford, pp 87–192
Bachmann MO, Eachus J, Hopper CD et al (2003) Socio-economic inequalities in diabetes complications, control, attitudes and health service use: a cross-sectional study. Diabet Med 20:921–929
Case A, Paxson C (2005) Sex differences in morbidity and mortality. Demography 42:189–214
Commission on Social Determinants of Health (CSDH) (2010) A conceptual framework for action on the social determinants of health. WHO, Geneva
Dalstra JAA, Kunst AE, Borrell C et al (2008) Socioeconomic differences in the prevalence of common chronic diseases: an overview of eight European countries. Int J Epidemiol 34:316–326
Dasgupta K, Khan S, Ross NA (2010) Type 2 diabetes in Canada: concentration of risk among most disadvantaged men but inverse social gradient across groups in women. Diabet Med 27:522–531
Dawson LJ (2009) Diabetes mellitus. In: Copstead LEC, Banasik JL (eds) Pathophysiology. Saunders, Saint Louis, pp 942–968
Elstad JI (2000) Social inequalities in health and their explanations. NOVA, Oslo
Espelt A, Borrell C, Roskam AJ et al (2008) Socioeconomic inequalities in diabetes mellitus across Europe at the beginning of the 21st century. Diabetologia 51:1971–1979
Espelt A, Kunst AE, Palència L, Gnavi R, Borrell C (2012) Twenty years of socio-economic inequalities in type 2 diabetes mellitus prevalence in Spain, 1987–2006. Eur J Public Health 22:765–771
Espelt A, Borrell C, Palència L et al (2013) Socioeconomic inequalities in the incidence and prevalence of type 2 diabetes mellitus in Europe. Gac Sanit 27:494–501
Forssas E, Keskimäki I, Reunanen A, Koskinen S (2003) Widening socioeconomic mortality disparity among diabetic people in Finland. Eur J Public Health 13:38–43
Gale EA, Gillespie KM (2001) Diabetes and gender. Diabetologia 44:3–15
Galobardes B, Lynch J, Davey Smith G (2007) Measuring socioeconomic position in health research. Br Med Bull 81–82:21–37
Gnavi R, Petrelli A, Demaria M, Spadea T, Carta Q, Costa G (2004) Mortality and educational level among diabetic and non-diabetic population in the Turin longitudinal study: a 9-year follow-up. Int J Epidemiol 33:864–871
Gnavi R, Picariello R, Karaghiosoff L, Costa G, Giorda C (2009) Determinants of quality in diabetes care process: the population-based Torino study. Diabetes Care 32:1980–1985
Houweling T, Kunst AE, Huisman M, Mackenbach JP (2007) Using relative and absolute measures for monitoring health inequalities: experiences from cross-national analyses on maternal and child health. Int J Equity Health 6:15. doi:10.1186/1475-9276-6-15
Imkampe AK, Gulliford MC (2011) Increasing socio-economic inequality in type 2 diabetes prevalence—repeated cross-sectional surveys in England 1994–2006. Eur J Public Health 21:484–490
Jougla E, Papoz L, Balkau B et al (1992) Death certificate coding practices related to diabetes in European countries—the ‘EURODIAB Subarea C’ Study. Int J Epidemiol 21:343–351
Koskinen SV, Martelin TP, Valkonen T (1996) Socioeconomic differences in mortality among diabetic people in Finland: 5 year follow up. BMJ 313:975–978
Krämer H, Rüter G, Schöttker B et al (2012) Gender differences in healthcare utilization of patients with diabetes. Am J Manag Care 18:362–369
Landman GW, Kleefstra N, van Hateren KJ, Bilo HJ, Groenier KH (2013) Educational disparities in mortality among patients with type 2 diabetes in The Netherlands (ZODIAC-23). Neth J Med 71:76–79
Macintyre S, Hunt K (1997) Socio-economic position, gender and health: how do they interact? J Health Psychol 2:315–334
Mackenbach JP, Huisman M, Andersen O et al (2004) Inequalities in lung cancer mortality by the educational level in 10 European populations. Eur J Cancer 40:126–135
Meslé F (2002) The registration of causes of death: problems of comparability. In: Wunsch G, Mouchart M, Duchêne J (eds) The life table. Modelling survival and death. Kluwer Academic Publishers, Dordrecht, pp 171–190
Östergren O, Menvielle G, Lundberg O (2001) Adjustment method to ensure comparability between populations reporting mortality data in different formats in the EURO-GBD-SE project. Working document. http://www.euro-gbd-se.eu/fileadmin/euro-gbd-se/public-files/Working%20document%20on%20the%20correction%20factor.pdf. Accessed 23 July 2014
Phelan JC, Link BG, Tehranifar P (2010) Social conditions as fundamental causes of health inequalities: theory, evidence, and policy implications. J Health Soc Behav Suppl 51:S28–S40
Robbins JM, Vaccarino V, Heping Z, Kasl SV (2001) Socioeconomic status and type 2 diabetes in African American and non-hispanic white women and men: evidence from the Third National Health and Nutrition Examination Survey. Am J Publ Health 91:76–83
Robbins JM, Vaccarino V, Zhang H, Kasl SV (2005) Socioeconomic status and diagnosed diabetes incidence. Diabetes Res Clin Pract 68:230–236
Romon I, Jougla E, Balkau B, Fagot-Campagna A (2008) The burden of diabetes-related mortality in France in 2002: an analysis using both underlying and multiple causes of death. Eur J Epidemiol 23:327–334
Roper NA, Bilous RW, Kelly WF, Unwin NC, Connolly VM (2001) Excess mortality in a population with diabetes and the impact of material deprivation: longitudinal, population based study. BMJ 322:1389–1393
Roskam AJ, Kunst AE, Van Oyen H et al (2010) Comparative appraisal of educational inequalities in overweight and obesity among adults in 19 European countries. Int J Epidemiol 39:392–404
Sacerdote C, Ricceri F, Rolandsson O et al (2012) Lower educational level is a predictor of incident type 2 diabetes in European countries: the EPIC-InterAct study. Int J Epidemiol 41:1162–1173
Sen G, Iyer A, Mukherjee C (2009) A methodology to analyse the intersections of social inequalities in health. J Hum Dev Capab 10:397–415
Smith J (2007) Diabetes and the rise of the SES health gradient. Working Paper 12905. National Bureau of Economic Research, Cambridge
Tang M, Chen Y, Krewski D (2003) Gender-related differences in the association between socioeconomic status and self-reported diabetes. Int J Epidemiol 32:381–385
Vandenheede H, Lammens L, Deboosere P, Gadeyne S, De Spiegelaere M (2011) Ethnic differences in diabetes-related mortality in the Brussels-Capital Region (2001–05): the role of socioeconomic position. Int J Public Health 56:533–539
Vandenheede H, Vanroelen C, Gadeyne S, De Grande H, Deboosere P (2013) Household-based socioeconomic position and diabetes-related mortality among married and cohabiting persons: findings from a census-based cohort study (Flanders, 2001–2010). J Epidemiol Community Health 67:765–771
Wagstaff A, Paci P, van Doorslaer E (1991) On the measurement of inequalities in health. Soc Sci Med 33:545–557
Wardle J, Steptoe A (2003) Socioeconomic differences in attitudes and beliefs about healthy lifestyles. J Epidemiol Community Health 57:440–443
Wild S, Roglic G, Green A, Sicree R, King H (2004) Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 27:1047–1053
Acknowledgments
This work was funded by the Public Health Programme of the European Union, Grant Agreement 20081309.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
For the EURO-GBD-SE consortium (including Annibale Biggeri, Santiago Esnaola, Suzanne Fry, Katalin Kovács, Johannes Klotz, Anita Lange, Olle Lundberg, Olof Östergen, Enrique Regidor, Bjørn Heine Strand, Bogdan Wojtyniak).
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Vandenheede, H., Deboosere, P., Espelt, A. et al. Educational inequalities in diabetes mortality across Europe in the 2000s: the interaction with gender. Int J Public Health 60, 401–410 (2015). https://doi.org/10.1007/s00038-015-0669-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00038-015-0669-8