Abstract
In the Bale Mountains, the ericaceous belt ranges between 3200 and 3800 m asl. Studies indicate an expansion on the Sanetti Plateau at the end of the Late Glacial and during the early Holocene. Currently, only patches of Erica growing between boulders are found on the Plateau, while most of the landscape above 3800 m asl is covered by afro-alpine plants. Driving factors for Erica patches above the upper ericaceous ecotone is a matter of debate. This study evaluates site variables and biogeochemical properties of soils under Erica patches and nearby Erica-free control to understand the environmental conditions responsible for the patchy occurrence of Erica on the Sanetti Plateau. Except for the boulder richness, Erica and control plots have comparable topography, soil texture, and electrical conductivity. However, soils below Erica patches have higher total organic carbon, nitrogen, carbon-to-nitrogen ratios, and black carbon contents than the control plots indicating fresh organic matter input and availability of combustible fuel. This implies that Erica did not fully cover the control plots in former times. Carbon and nitrogen stocks were slightly higher in control plots due to the lower stone contents of the profiles. In addition, soils of the Erica plots showed more positive δ13C values than the control soils, possibly attributed to water stress. In general, the relief and soil conditions of control plots may support the growth of Erica. However, Erica growing between boulders seems to benefit from the favorable microclimate and physical protection against grazing and fire.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Defining the tree line ecotone of high-elevation vegetation is often problematic and debatable as it is difficult to unambiguously identify and quantify the factor that plays the most significant role (Körner 2007; Jacob et al. 2015). Such difficulties have been observed in the East African Mountains, which are vulnerable to climate change and anthropogenic impacts (Wesche et al. 2000; Jacob et al. 2015). Erica, also known as heathers or heath, is one of the most widely distributed plant genera in the Ericaceae family (Oliver 1989; Kron et al. 2002). Its geographical distribution covers Europe, the Middle East, South America, and Africa (Mcguire and Kron 2011). Erica species (Erica trimera and Erica arborea) dominate and characterize most high-elevation mountains in eastern Africa, forming the upper tree line forest (Hedberg 1951; Wesche et al. 2000; Fetene et al. 2006). Ericaceous vegetation is widespread in northern (Siemen Mountains) and southern Ethiopia (Bale Mountains, Mt. Chillalo, Mt. Kaka, Galama Mountains, and Arsi highlands) (Hedberg 1951; Miehe and Miehe 1994). In the Bale Mountains, the ericaceous belt covers an area of 90,000 ha and spans between 3200 and 3800 m asl (Miehe and Miehe 1994). According to Fetene et al. (2006), the ericaceous vegetation is grouped into three elevational subzones. The lower subzone is characterized by the Erica-dominated Hagenia-Hypericum forest, which spans between 3000 and 3400 m asl. The central part covers the elevation between 3400 and 3600 m asl; dominated by Erica trimera, Hypericum revolutum, and Alchemilla abyssinica. In the upper subzone between 3600 and 4200 m asl, the Ericaceous vegetation has a patchy appearance, especially at the Sanetti Plateau between 3800 and 4200 m asl. Such patchy appearance of Erica vegetation is also reported from other high-elevation African mountains such as Mt. Kilimanjaro, Mt. Elgon, Mt. Kenya, and the Rwenzori Mountains (Hedberg 1951; Beck et al. 1983; Wesche et al. 2000) and the Andes in South America (Kessler 2000). The presence of these Erica patches, mainly growing between big boulders, makes the demarcation of the upper limit of Erica difficult (Friis 1986; Miehe and Miehe 1994). The Sanetti Plateau is one of the largest afro-alpine areas in Africa (Hillman 1988; Groos et al. 2021). The area is mainly covered by afro-alpine plant species such as Helichrysum, Alchemilla, Lobelia, and grasses (Friis 1986; Miehe and Miehe 1994). However, big boulders, frequently present on slopes of the Plateau, serve as "refugee camps" for Erica above the upper ericaceous ecotone at ca. 3800 m (Miehe and Miehe 1994).
Climatic stress, fire, and overgrazing are postulated as potential drivers for tree line changes in the ericaceous vegetation of the Bale Mountains (Miehe and Miehe 1994; Wesche et al. 2000; Fagúndez 2013; Jacob et al. 2015; Johansson et al. 2018; Kidane et al. 2022). Similarly, the presence of Erica patches in the highest elevation east African mountains is explained by the impact of human-induced fire and climate change (Kessler 2000; Hemp and Beck 2001; Hemp 2005). For instance, Chala et al. (2017) suggested the downward shift of the tree line by 1000 m and the corresponding expansion of the afro-alpine habitat during the Last Glacial Maximum (LGM) in eastern Africa. Furthermore, paleoenvironmental studies in the Bale Mountains revealed the effect of climate fluctuation on vegetation (Umer et al. 2007; Gil-Romera et al. 2019; Groos et al. 2021; Mekonnen et al. 2022). Umer et al. (2007) reported that the ericaceous vegetation extended to the Sanetti Plateau during the beginning of the Holocene in response to the warm and humid climate, and they suggested that the vegetation expansion started to decrease during the mid-Holocene due to increasing aridity. Kidane et al. (2022) and Chala et al. (2016) assumed that the current climate warming might alter the spatial arrangement of the ericaceous vegetation in the Bale Mountains by supporting its expansion to the Plateau and simultaneously the extinction of afro-alpine vegetation.
At present, the growth of Erica on the Sanetti Plateau is mainly restricted to boulder-rich sites. In contrast, areas nearby without boulders are generally free of Erica and mainly covered by afro-alpine species such as Alchemilla haumannii, Helichrysum splendidum and Festuca abyssinica. These Erica patches are assumed to be relics of the ericaceous vegetation expansion during the humid Early Holocene (Miehe and Miehe 1994; Umer et al. 2007). Fire has been prominent in the Bale Mountains for hundreds to thousands of years (Gil-Romera et al. 2019; Mekonnen et al. 2022). This is because the pastoralists in the Bale Mountains believe that fire stimulates the growth of new grass for cattle grazing, thus improving fodder quality, controlling insect pests, and protecting their cattle from predators (Miehe and Miehe 1994; Fetene et al. 2006; Belayneh et al. 2013). As a result, the spatial extent of different vegetation groups in the Bale Mountains, including the isolated patches of Erica, has drastically changed over time (Kidane et al. 2012). Grazing is also considered to play a significant role in controlling the expansion of Erica. According to Johansson et al. (2010), cattle, goats, and sheep are the main grazing domestic animals in the ericaceous vegetation. Furthermore, according to Gebremedhin et al. (2016), Erica arborea is a highly preferred diet by domestic goats in the Semien Mountains. In particular, goats and sheep intensively graze the Erica seedlings sprouting after fire, thus putting immense pressure on their regeneration. However, in addition to these biotic factors, temperature, precipitation, soil quality, and wind speed have determinant effects on the growth of trees (Jacob et al. 2015). Miehe and Miehe (1994) hypothesized that temperature and moisture availability could be factors limiting the growth of Erica on the Sanetti Plateau. Moreover, Groos et al (2022) have presented ground temperature data sets for the Bale Mountains high-elevation sites. However, up to now, except for observational descriptions, there has been no quantified evidence that can explain the patchy occurrence of Erica on boulder-rich slopes of the Sanetti Plateau above ca. 3800 m asl.δ13C analyses of leaves and soils are used to determine historical alterations in the boundary between C3 woodland and C4 grasslands (Eshetu 2002). This is mainly due to the differences in the photosynthetic pathways between C3 and C4 plants. While C3 plants are characterized by δ13C values of – 22 to – 35‰, C4 plants show δ13C values of – 11 to – 17‰ (Marshall et al. 2007; Tiunov 2007). In addition, δ13C is a prominent proxy for determining plant water status. It decreases the δ13C value in dry tissue due to stomatal constraints on gas diffusion during periods of biomass production (Körner 2012a). Black carbon (BC) is a highly condensed carbonaceous product of organic matter combustion (Glaser et al. 1998; Brodowski et al. 2005). Due to its highly stable polycyclic hydrocarbons, black carbon is widely used as a proxy for fire history reconstruction (Kuzyakov et al. 2014). Applying this proxy in this study is promising because fire is a common incidence in the ericaceous vegetation of the Bale Mountains. In addition, the patchy distribution of Erica on the Sanetti Plateau is assumed to document fire disturbance (Miehe and Miehe 1994; Wesche et al. 2000; Gil-Romera et al. 2019).
In this study, we aim to contribute to a better understanding of the distribution of Erica on the Sanetti Plateau by comparing Erica and non-Erica plots (control plots) based on different environmental and biogeochemical proxies such as topography and selected soil properties. Specifically, our research questions are: (i) Is topography (elevation, exposition, inclination) a limiting factor, and is stone cover a favoring factor for the growth of Erica? (ii) Do Erica sites have different soil properties than control plots (e.g., pH, TOC, N contents and stocks, stable isotopes, and BC contents)? Finally, (iii) Is there any evidence that Erica has previously occupied the control plots?
Materials and methods
Study area
Geology, vegetation, and climate
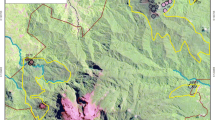
The Bale Mountains National Park is located in the Oromia National Regional State, south-east Ethiopia (6°29′–7°10′N and 39°28′–39°57′E; Fig. 1), covering an area of ~ 2200 km2 (Hillman 1988). The park was established in 1970 to conserve its endemic fauna and flora. The Bale Mountains were formed by volcanic eruptions during the Miocene and Oligocene, releasing large amounts of basalt, rhyolite, and ignimbrite (Μohr 1983). They rise from the eastern highlands (2500 m asl) alongside the Ethiopian rift valley to the Sanetti Plateau (3800–4000 m asl) and Tullu Dimtu (4377 m asl) (Miehe and Miehe 1994). Besides, the Bale Mountains were one of the most glaciated mountains in Ethiopia during the Late Pleistocene (Osmaston et al. 2005; Mark and Osmaston 2008; Groos et al. 2021).
Map showing the geographical location of the Bale Mountains in Ethiopia and the study sites; five covered by Erica patches (E1–E5) and five located nearby without Erica (Control sites C1–C5), covered by grass and Helichrysum and temperature loggers installed by Groos et al. (2022) at the site 1 (TM09m and TM08) and site 2 (TM07 and TM10)
Spatial and temporal variability prevailed in the different vegetation compositions of the Bale Mountains along elevation. The northern and southern declivities between 1450 and 3200 m asl are characterized as dry and moist Afromontane forests, respectively. One of the dominant vegetation types in the Bale Mountains is the Ericaceous belt mediating between Afromontane and afro-alpine vegetation (Miehe and Miehe 1994). It spans between about 3200 and 3800 m asl, dominated by Erica arborea L. and Erica trimera (Engl.) Beentje (Hedberg 1951) in the form of shrubland and moist forest along the northern and southern slopes, respectively. The afro-alpine vegetation dominates above 3800 m asl up to 4377 m asl, punctuated by patches of Erica (Miehe and Miehe 1994). The dominant plant species in each elevational zone are covered in depth elsewhere (Hedberg 1951; Friis 1986; Miehe and Miehe 1994).
The climatic conditions of the Bale Mountains are defined by topography and the movement of the Intertropical Convergence Zone and the Congo air basin (Levin et al. 2009; Costa et al. 2014). As a result, the climate is characterized by a dry and bimodal rainy season. The dry season spans from November to February, and two rainy seasons prevail from March to June and from July to October, respectively. In Dinsho (the Bale Mountains National Park headquarters at 3070 m asl), the mean annual precipitation is 1069 mm, and the mean annual temperature is 11.8 °C. The southwestern part of the mountains experiences higher precipitation, with 1000–1500 mm per year, than the northern part, which exhibits annual rainfall ranging between 800 and 1000 mm per year (Hillman 1986; Tiercelin et al. 2008). The watershed of the Plateau is characterized by flat, swampy areas and many small, shallow lakes crucial for stream and river flow regulation (Belayneh et al. 2013). While the northern winds from the Arabian Peninsula dominate during the dry season, the southeasterly monsoon transports moisture from the Indian Ocean during the rainy seasons (Lemma et al. 2020). During the rainy season, snow can fall on the Plateau and highest peaks but usually does not persist for longer than a few hours or some days (Miehe and Miehe 1994).
Sample collection
Five sites were identified along a NE-SW transect over the Sanetti Plateau (Fig. 1). For each site two plots were identified; one covered by dense Erica patches (Erica plot) and the second one only by grass spp., Helichrysum, and a few Alchemilla plants (Figs. 1, 2) but without Erica (control plot). After testing the soil homogeneity using a soil auger, a total of ten representative soil profiles (five under Erica patches and five in adjacent control plots) were dug until the bedrock was reached, and the main topographic variables (elevation, aspect, and slope) and soil depth were recorded. Soil profiles were described (e.g., color, texture, stone contents in vol.%, and root content, see Table S1 and Bodenkundliche Kartieranleitung (Ad-hoc-Arbeitsgruppe Boden 2005)), and samples were taken from each soil horizon, air dried, and stored in plastic bags. In addition to soil samples, leaves of Erica, grass (Festuca), and Helichrysum, the dominant afro-alpine species, were collected randomly from the study plots.
Pictures showing sampling sites. a Site of Erica patch (E4) at 3970 m asl in north exposition b Erica patch E2 with Erica shrubs growing between stones and boulders. c Soil profile below Erica shrubs (E2). d Erica seedlings growing beside a single big boulder on a control plot. e Control plot (C1) covered by Helichrysum sp at 4150 m asl. f Soil profile below control plot (C3)
Laboratory analyses and statistical evaluation
pH and electrical conductivity were measured using 10 g of O-layer and 20 g of mineral soil samples. After placing the samples in containers, 25 ml of distilled water was added. The suspensions were shaken for 30 min and allowed to settle. Subsequently, electrical conductivity and pH were measured with a glass electrode. Soil texture analyses were done using PARIO Soil Particle Analyzer (METER Group, Munich, Germany). Before measurement, samples were treated using 50 ml of 30% H2O2 and 40 g/L solution of Na4P2O7. Total organic carbon (TOC), total nitrogen (N), and the natural abundance of δ13C and δ15N were measured using an elemental analyzer coupled to an isotope ratio mass spectrometer (EL-IRMS). While sucrose (ANU, IAEA, Vienna, Austria) and CaCO3 (NBS 19, TS limestone) were used as calibration standards for δ13C, IAEA 305A, IAEA N2, IAEA NO3, and USGS 41 were used for δ15N. The precision of δ13C and δ15N measurements was 0.2‰ and 0.3‰, respectively.
Soil organic C and N stocks of each mineral soil horizon and the whole soil profile were calculated according to Batjes (Batjes 1996).
where SOCstock is the total amount of organic carbon (in kg m−2), SOC is the proportion of organic carbon (kg Mg−1) in layer i, BD is the bulk density (Mg m−3) of layer i, t is the thickness of this layer (m), and S is the volume of the fraction of fragments > 2 mm.
The carbon stock of the O-layers was calculated differently from that of the mineral soils:
The organic material has been sampled using a 20 cm × 20 cm metal frame. This procedure provides the weight of all the organic material sampled per unit area, independently of its depth.
BC was analyzed using benzene polycarboxylic acids (BPCAs) as molecular markers, following Glaser et al. (1998) with modifications by Brodowski et al. (2005). Five hundred mg of each sample were hydrolyzed with 10 ml 4 M TFA for 4 h at 105 °C. The hydrolyzed samples were filtrated on glass fiber filters and rinsed several times with de-ionized water to remove polyvalent cations. Subsequently, the samples were digested with 65% nitric acid for 8 h at 170 °C in a high-pressure digestion apparatus. The solution was passed through Dowex 50 W resin columns (200 to 400 meshes) to remove polyvalent cations. After derivatization, the BPCAs were separated using gas chromatography (SHIMADZU, GC-2010, Kyoto, Japan) and detected using a flame ionization detector (FID) with an injection temperature of 300 °C. All statistical analyses were done using R software.
Results
Environmental features and soil properties
Erica and control plots of a given site generally have comparable topographic features; they are located in southern (sites 1, 2, and 3) and north-eastern (sites 4 and 5) expositions between 3850 and 4150 m asl (Table 1).
Except for E4, usually, the inclination is weak. Erica plots are generally covered by big boulders (average 60%), with Erica shrubs (~ 1.5 m tall) growing between the boulders (Fig. 2a, b). In contrast, control plots are completely covered by grass and afro-alpine plant species with no visible big stones/boulders on the surface (Fig. 2e, f).
The soils under study are characterized by thick Ah horizons (up to 75 cm), which were further subdivided into Ah1, Ah2, and Ah3. Additionally, B, C, and transitional horizons could be identified based on morphological properties such as color, stone content, roots, etc. (Fig. 2c, f; Table S1). Due to the very high stone contents of the C horizons, the comparison between Erica plots and the corresponding control plots was restricted to O, Ah, and Bw horizons; only the SOC stocks were calculated for the whole soil profile. Since the percentage of stones is a very influential factor for root penetration, water storage capacity, carbon, and nutrient accumulation, stoniness was determined per horizon. The results showed that, even though not statistically significant, stone contents are often higher below Erica than in soils of control plots (Fig. 3). For instance, the Ah1 horizons of control and Erica plots have an average stone content of 36 and 45, respectively. In the Bw horizons of Erica and control plots, stone contents increased to ca. 90 (Fig. 3). Bulk density of control soils was significantly higher than that below Erica (Fig. S1, p = 0.002).
Stone content in soil horizons of all Erica and control sites, estimated according to Bodenkundliche Kartieranleitung (2005). The notched box plots indicate the median (solid lines between the boxes) and interquartile range (IQR) with upper (75%) and lower (25%) quartiles. The notches display the 95% confidence interval of the median. The lines extending outside the box (whiskers) show variability outside the quartiles
Soil texture fractions are highly variable along the soil depth of Erica and control profiles (Fig. S2). However, no significant difference is obtained between Erica and the control plots. pH values range between 5.6 and 6.8 in Erica and control soils (Fig. 4). However, the pH values do not vary significantly between Erica and control plots, except for the O layers below Erica, which show a slight decrease in pH values (x̄ = 5.6). EC values vary between 45 and 198 µS cm−1 below Erica and between 31 and 488 µS cm−1 in control soils (Fig. 4). Moreover, EC values decrease with increasing soil depth in both Erica and control soil profiles.
Electrical conductivity (EC) and pH in soil horizons of Erica and control sites. The notched box plots indicate the median (solid lines between the boxes) and interquartile range (IQR) with upper (75%) and lower (25%) quartiles. The notches display the 95% confidence interval of the median. The lines extending outside the box (whiskers) show variability outside the quartiles. The circles represent outliers
Soil organic carbon and nitrogen contents
TOC values of Erica plants show significantly higher values than Helichrysum and Festuca (p = 0.009) (Fig. 5). However, there is no significant difference between the TOC contents in the soils of Erica and control plots. Nevertheless, the mineral soil horizons of the Erica plots tend to have higher TOC contents than the control profiles. TOC values of organic layers vary between 13 and 33% in control plots and range from 23 to 46% in Erica plots. Moreover, the TOC values of the Ah3 layers below Erica are higher (x̄ = 8.2%) than those of the control plots (x̄ = 5.7%). Similar to TOC, the N contents of Erica leaves are higher than those of Festuca and Helichrysum (Fig. S3). In the organic and Ah1 horizons of control plots, N contents are slightly higher than below Erica, while N in the other horizons is somewhat enriched below Erica. N values, like TOC values, decline with increasing depth in all soil profiles.
Total organic carbon (TOC) and TOC/N ratio of plant leaves and soil horizons of Erica and control sites. The notched box plots indicate the median (solid lines between the boxes) and interquartile range (IQR) with upper (75%) and lower (25%) quartiles. The notches display the 95% confidence interval of the median. The lines extending outside the box (whiskers) show variability outside the quartiles. The circles represent outliers
In soils, TOC/N ratios range from 10 to 45 below Erica and 9–26 in control plots (Table S2). They are, in general, significantly higher in Erica plots than in control plots (p = 0.006). While TOC/N ratios in control soils drastically decrease with increasing soil depth (from Ah1 to Ah3), they remain significantly high in Ah2 (p = 0.03) and Ah3 (p = 0.01) soil horizons under Erica (Fig. 5).
Soil organic carbon and nitrogen stocks
Figure 6 depicts soil organic carbon stocks restricted to soil depths of 0–30 cm, 0–50 cm, and the whole profile that considers the profile's maximum depth in each plot. The results show that, even if not statistically significant, SOC stocks are slightly higher in control plots than in Erica plots (Fig. 6).
Soil organic carbon (SOC) in soil profiles under Erica and control sites. The notched box plots indicate the median (solid lines between the boxes), and interquartile range (IQR) with upper (75%) and lower (25%) quartiles. The notches display the 95% confidence interval of the median. The lines extending outside the box (whiskers) show variability outside the quartiles. The circles represent outliers
Black carbon
Our results demonstrate significantly higher BC contents (related to sample: p = 0.01 and related to TOC: p = 0.02) in Erica soils than in control soils (Fig. 7a, b). Moreover, the BPCA pattern shows a higher contribution of B4CA, B5CA, and B6CA and a lower percentage of B3CA in both Erica and control plots (Fig. S4). The B5CA/B6CA ratios range from 0.63 to 1.39 below Erica and from 0.74 to 2.51 in the control plots (Fig. 7c). Topsoils of control plots have significantly higher B5CA/B6CA ratios (p = 0.02) than the Erica profiles. The Bw horizons of the control soils always have lower BC and B6CA contents than those below Erica.
Black carbon contents (BC) (a), black carbon contribution to TOC (b), and B5CA/B6CA ratios c of Erica and control sites soils. The notched box sites indicate the median (solid lines between the boxes), and interquartile range (IQR) with upper (75%) and lower (25%) quartiles. The notches display the 95% confidence interval of the median. The lines extending outside the box (whiskers) show variability outside the quartiles. The circles represent outliers
Stable isotopes
δ13C values of leaves sampled from the Erica and control plots range between – 28.6 and – 23.8‰. Erica leaves have significantly higher values than Festuca and Helichrysum (p = 0.03) (Fig. 8). δ13C values vary between – 24.7 and – 22.3 in soils of Erica plots and from – 27 to – 22‰ in control plot soils, with the highest values recorded in Bw horizons (Fig. 8). In general, δ13C values are significantly higher in soils under Erica (p = 0.02) than in control profiles, and they increase with increasing soil depth in both Erica and control plots. δ15N values vary between – 6.5 and 0.1 in leaves (Fig. 8). Helichrysum and Festuca show higher δ15N values than Erica leaves. Similarly, soils under Erica shrubs reveal δ15N values ranging from – 3.4 to 7.0‰, with the highest values recorded in Bw horizons. Control plot soil exhibits δ15N values between – 2.7 and 9.0‰, with the highest value recorded in Ah3 horizons (Fig. 8).
δ13C and δ15N values of leaves and soil layers of Erica and control plots. The notched box plots indicate the median (solid lines between the boxes) and interquartile range (IQR) with upper (75%) and lower (25%) quartiles. The notches display the 95% confidence interval of the median. The lines extending outside the box (whiskers) show variability outside the quartiles. The circles represent outliers
Discussion
Our results illustrate similarity in site exposition and environmental features of Erica and control plots established per site. The most striking difference is the high amount of boulders covering the Erica plots. The Sanetti Plateau is characterized by harsh climatic conditions with strong winds, extreme solar radiation, heating, desiccation, and frequent frost at night (Miehe and Miehe 1994; Wesche 2003; Groos et al. 2022). Alpine plants adapt to such extreme conditions, whereas woody plants face difficulties regulating the ambient temperature (Wesche 2003; Wesche et al. 2008; Körner 2012b). According to Körner (2012a, b), the upper tree line is globally seen as controlled by the mean temperature during the growing season. Therefore, we assume that the big boulders positioned above the upper timber line of the Erica belt protect the Erica patches against wind and provide warmth and shade, particularly for the Erica seedlings (Wesche et al. 2008). On the other hand, afro-alpine plants are rather shade-intolerant due to their high photosynthetic light-compensation points (Billings and Mooney 1968; Johansson et al. 2018). Therefore, Erica seedlings growing beside boulders may outcompete the afro-alpine plants and mature into shrubs. This interpretation is supported by Fig. 2d, which depicts Erica seedlings growing in the south exposition alongside a single big boulder on a control plot. However, the Erica plants were confined to short stature due to intensive grazing.
Groos et al. (2022) installed temperature data loggers at the elevation range from 3493 to 4377 m asl. From these temperature data loggers, four were installed at 10 cm depth at our Erica (E1 and E2, TM09m and TM10m) and control (C1 and C2, TM08m and TM07m) plots (see Fig. 1). The results show no difference in the temperature pattern between Erica and control plots (Fig. S5). At site 1, a high temperature was recorded during the dry season, decreasing during the rainy season at both Erica and control plots. In contrast, at site 2, the temperature decreased during the dry season and increased during the rainy season at both the control and Erica plots. According to Groos et al. (2022), the temperature difference between the sites can be attributed to a difference in exposition. Nevertheless, since the number of data loggers installed at our study sites was very low, further studies are required to check whether there is a difference in microclimate between Erica and the control plots.
Except for bulk density, TOC, TOC/N and BC, other biogeochemical proxies do not show a significant difference between Erica and control plots. The significantly lower soil bulk density under Erica could be caused by a high organic matter content and increased stone contents (Fig. 3). Even though statistically not significant, the lower pH values recorded in soils of Erica plots (Fig. 4) can be explained by the acidifying effects of the thick, slowly decomposing Erica litter (Dahlgren et al. 1997). Nevertheless, pH values recorded in our Erica profiles are slightly higher than those reported by Johansson (2013) from the Erica belt in the Bale Mountains. These high pH values might be caused by increased alkaline dust inputs from the surroundings on the Sanetti Plateau. Moreover, the decrease of EC values along soil depth in both Erica and control soil profiles might be due to the ascendance of water and ions during the dry period.
Higher soil TOC and N contents of Erica plots compared to the control plots can be attributed to differences in quality and quantity of the litter input (see Fig. 5 and S3, high TOC and N values of Erica leaves) (Andrén and Kätterer 1997). Furthermore, increased insolation on control plots (Miehe and Miehe 1994) might support organic matter degradation, whereas soils under Erica benefit from the shade provided by large rocks and the Erica canopy. Moreover, the Ah3 and Bw horizons at the bottom of the soil profiles of the control plots contain less TOC and N than the control soils, which may indicate that Erica was not a common vegetation component on these plots in the past. Despite lower N values of plant species growing on control plots, their O and Ah layers show higher N values (Fig. S3), likely due to fecal N input from grazing cattle (Baron et al. 2002; Johansson et al. 2012). High TOC/N values recorded in our Erica plots agree with Mekonnen et al. (2019) and Zech (2006), reporting that such high values characterize the Erica vegetation and its soils at high elevations in other African Mountains. The low TOC/N ratios, especially of the Ah3 and Bw horizons of control plots, also support the interpretation that the control plots have never been fully covered by Erica. Besides, the higher TOC/N ratios of the mineral soils below Erica reflect that Erica litter is less decomposed by soil microorganisms (Jacob et al. 2015).
SOC and N stocks of Erica plots are partly lower than those of control plots (Fig. 6; Fig. S3). This is mainly due to their higher stone contents which are negatively correlated with the SOC and N stocks (R = – 0.5). Furthermore, the PCA results demonstrate the impact of stone contents on the carbon and nitrogen stock accumulation (Fig. S6). Similarly, Gebrehiwot et al. (2018) found higher SOC stocks in the afro-alpine grassland soils of the Abune Yosef Mountain in Northern Ethiopia compared to soils below the ericaceous forest. In a study about soils under different vegetation in the Bale Mountains, Yimer et al. (2006) noted higher carbon stocks in 0.3–1 m soil depth below Erica. However, this difference can be explained by the variability of climatic conditions and environmental factors between the lower elevation of the Erica belt and our high-elevation sites on the Sanetti Plateau.
Our results further show that soils under Erica are enriched in BC compared to soils of the control plots (Fig. 7a, b). This indicates a higher amount of combustible fuel on Erica plots than on control plots. Furthermore, the significantly increased BC contents, especially of Ah2 and Ah3 horizons in soils below Erica (Fig. 7a, b), support our interpretation that Erica did not intensively cover these plots previously. Otherwise, a higher accumulation of recalcitrant BC would have been preserved during the burning of former Erica shrubs.
In addition to BC contents, the relative contribution of benzene polycarboxylic acids (BPCA) provides information on fire temperature and fuel source (Schneider et al. 2010; Wolf et al. 2013). For instance, benzene rings with four and five carboxylic groups (B4CA and B5CA) are primarily produced at low temperatures (~ 300 °C), whereas benzene rings with six carboxylic groups (B6CA) are mainly produced at high temperatures (600 °C). Moreover, the B5CA/B6CA ratio is supposed to indicate the type of vegetation burned in the area. According to Wolf et al. (2013), forest ground and grass fires have B5CA/B6CA ratios of 1.5–2.0, whereas shrub fires have B5CA/B6CA ratios of 0.8–1.7. Our result correlates with the range suggested by Wolf et al. (2013) for shrubs and grassland fires. Moreover, significantly high B5CA/B6CA values in control plots indicate low temperature, while low B5CA/B6CA values in Erica plots indicate high combustion temperature. The latter is attributed to the high flammability of Erica twigs due to low moisture content and high concentrations of oils, waxes, and terpenes, which are readily volatile and contribute to the energy released by burning. In contrast, Helichrysum, the main vegetation constituent of the control sites, is naturally less flammable (Johansson et al. 2012).
The δ13C values of plants from our Erica and control plots are within the range reported for C3 plants (Marshall et al. 2007; Tiunov 2007) (Fig. 8). This is consistent with results of our previous transect study in the Bale Mountains reporting that the dominant plants including grasses, growing between 2550 and 4377 m asl are characterized as C3 plants (Mekonnen et al. 2019). However, the significantly high δ13C values of Erica plants (Fig. 8) seem to contrast with the finding of the transect study (Mekonnen et al. 2019), where no significant difference between Erica and other dominant plants could be identified. This discrepancy might be due to the effect of elevation on δ13C values and the different ways plants adjust their gas exchange to mitigate the decline in atmospheric CO2 pressure along elevation (Körner et al. 1991). To prove this, we ran a correlation analysis between elevation and δ13C values of dominant plants from the transect study. The results showed that elevation correlates positively with δ13C of Erica leaves (R = 0.5) but negatively with Festuca (R = – 0.7), whereas δ13C of Alchemilla leaves did not change with elevation. The isotope pattern found for Erica in this study is in agreement with the elevation effect (Körner et al. 1991), whereas the isotope pattern of Festuca does not. A similar statistical test was not possible for Helichrysum due to limited data. Moreover, the relatively positive δ13C values of Erica leaves from our Erica plots on the Sanetti Plateau might suggest temporary water stress, e.g., during the dry season. In contrast, Festuca does not respond similarly, most likely due to distinct physiological conditions that enable it to mitigate water scarcity (Hedberg 1964). Moreover, the high stone content of the soil profiles under Erica patches reduces the plant-available water storage capacity. δ13C values of the O, Ah1, and Ah2 horizons under Erica correspond to the values of the Erica leaves. The more negative δ13C values of these horizons from control soils correlate with the negative δ13C values of Helichrysum and Festuca. The increase from O to Bw shows progressing organic matter mineralization with increasing soil depth in both plots. This interpretation is supported by the negative correlation between δ13C and carbon stocks (Fig. S6). Generally, higher δ15N values in control profiles correlate with high δ15N values of Helichrysum and Festuca. The continual increase of δ15N along soil depth is attributed to the loss of δ14N during microbial decomposition of organic matter (Natelhoffer and Fry 1988; Eshetu 2004; Andersson et al. 2012). Besides, the frequent vegetation fires at the high elevations of the Bale Mountains could also be responsible for increased δ15N values, recorded in both Erica and control plot soils (Zech et al. 2011; Johansson 2013). Generally, higher δ15N values in control profiles likely correlate with the high δ15N values of Helichrysum and Festuca, implying that control plots were previously dominated by afro-alpine vegetation.
Conclusions
In this study, we examined potential factors responsible for the patchy occurrence of Erica above the upper ericaceous ecotone on the Sanetti Plateau, Bale Mountains. Apart from the boulder cover, topographic features, soil texture, and EC did not show significant differences between Erica and control plots. High TOC and TOC/N values were recorded below Erica and can be attributed to increased fresh organic matter input. Still, slightly higher SOC stocks were calculated for the control plots due to their lower stone contents. Erica leaves on the Sanetti Plateau were characterized by more positive δ13C values compared to leaves of Helichrysum and grass growing on the control plots. This difference is also reflected in the soils, probably influenced by water stress on the Erica plots. In addition, Erica sites are characterized by high BC contents indicating a high amount of combustible fuel at Erica plots. Low TOC, TOC/N ratio, and BC contents but high B5CA/B6CA ratios in the control plots indicate that Erica did not occupy the control plots in former times. However, Erica shrubs growing between the surface boulders appear to benefit from the improved microclimate created by the big dark basal rocks and from physical protection against grazing and fire. We conclude that, in general, the soil conditions of most control plots would allow the growth of Erica, but in the absence of boulders, the microclimatic conditions above 3800 m asl are too severe. Therefore, further investigations should focus on a detailed assessment of the microclimate conditions, soil moisture availability, and water potential of the Erica patches on the Sanetti Plateau.
Data availability
All data generated or analysed during this study are included in the supplementary information files.
References
Andersson RA, Meyers P, Hornibrook E et al (2012) Elemental and isotopic carbon and nitrogen records of organic matter accumulation in a holocene permafrost peat sequence in the east European Russian arctic. J Quat Sci 27:545–552. https://doi.org/10.1002/jqs.2541
Andrén O, Kätterer T (1997) ICBM: the introductory carbon balance model for exploration of soil carbon balances. Ecol Appl 7:1226–1236. https://doi.org/10.1890/1051-0761(1997)007[1226:ITICBM]2.0.CO;2
Baron VS, Mapfumo E, Dick AC et al (2002) Grazing intensity impacts on pasture carbon and nitrogen flow. J Range Manag 55:535–541. https://doi.org/10.2307/4003996
Batjes NH (1996) Total carbon and nitrogen in the soils of the world. Eur J Soil Sci 47:151–163. https://doi.org/10.1111/j.1365-2389.1996.tb01386.x
Beck E, Scheibe R, Senser M (1983) The vegetation of the Shira Plateau and the western slopes of Kibo (Mt. Kilimanjaro, Tanzania). Phytocoenologia 11:1–30. https://doi.org/10.1127/phyto/11/1983/1
Belayneh A, Yohannes T, Worku A (2013) Recurrent and extensive forest fire incidence in the Bale Mountains National Park (BMNP), Ethiopia: extent, cause and consequences. Int J Environ Sci 2:29–39
Billings WD, Mooney HA (1968) The ecology of Arctic and Alpine plants. Biol Rev 43:481–529. https://doi.org/10.1111/J.1469-185X.1968.TB00968.X
Boden AhA (2005) Bodenkundliche Kartieranleitung, Hrsg, 5th edn. Bundesanstalt für Geowissenschaften und Rohstoffe in Zusammenarbeit mit den Staatliche Geologische Dienste, Hannover
Brodowski S, Rodionov A, Haumaier L et al (2005) Revised black carbon assessment using benzene polycarboxylic acids. Org Geochem 36:1299–1310. https://doi.org/10.1016/j.orggeochem.2005.03.011
Chala D, Brochmann C, Psomas A et al (2016) Good-bye to tropical alpine plant giants under warmer climates? Loss of range and genetic diversity in Lobelia rhynchopetalum. Ecol Evol 6:8931–8941. https://doi.org/10.1002/ece3.2603
Chala D, Zimmermann NE, Brochmann C, Bakkestuen V (2017) Migration corridors for alpine plants among the ‘sky islands’ of eastern Africa: do they, or did they exist? Alp Bot 127:133–144. https://doi.org/10.1007/s00035-017-0184-z
Costa K, Russell J, Konecky B, Lamb H (2014) Isotopic reconstruction of the African humid period and congo air boundary migration at Lake Tana, Ethiopia. Quat Sci Rev 83:58–67. https://doi.org/10.1016/j.quascirev.2013.10.031
Dahlgren RA, Boettinger JL, Huntington GL, Amundson RG (1997) Soil development along an elevational transect in the western Sierra Nevada, California. Geoderma 78:207–236. https://doi.org/10.1016/S0016-7061(97)00034-7
Eshetu Z (2002) Historical C3–C4 vegetation pattern on forested mountain slopes: its implication for ecological rehabilitation of degraded highlands of Ethiopia by afforestation. J Trop Ecol 18:743–758. https://doi.org/10.1017/S0266467402002481
Eshetu Z (2004) Natural15N abundance in soils under young-growth forests in Ethiopia. For Ecol Manag 187:139–147. https://doi.org/10.1016/S0378-1127(03)00315-3
Fagúndez J (2013) Heathlands confronting global change: drivers of biodiversity loss from past to future scenarios. Ann Bot 111:151–172. https://doi.org/10.1093/aob/mcs257
Fetene M, Assefa Y, Gashaw M, Woldu Z, Beck E (2006) Diversity of afroalpine vegetation and ecology of treeline species in the Bale Mountains, Ethiopia, and the influence of fire. In: SpehnEM, Liberman M, Korner C (eds) Land use change and mountain biodiversity. CRC Press, New York, pp 25–38
Friis I (1986) Zonation of forest vegetation on the south slope of Bale Mountains, South Ethiopia. SINET Ethiop J Sci 9:29–44
Gebrehiwot K, Desalegn T, Woldu Z et al (2018) Soil organic carbon stock in Abune Yosef afroalpine and sub-afroalpine vegetation, northern Ethiopia. Ecol Process 7:1–9. https://doi.org/10.1186/s13717-018-0117-9
Gebremedhin B, Flagstad O, Bekele A et al (2016) DNA metabarcoding reveals diet overlap between the endangered walia ibex and domestic goats-Implications for conservation. PLoS One. https://doi.org/10.1371/journal.pone.0159133
Gil-Romera G, Adolf C, Benito BM et al (2019) Long-term fire resilience of the Ericaceous Belt, Bale Mountains, Ethiopia. Biol Lett 15:20190357. https://doi.org/10.1098/rsbl.2019.0357
Glaser B, Haumaier L, Guggenberger G, Zech W (1998) Black carbon in soils: the use of benzenecarboxylic acids as specific markers. Org Geochem 29:811–819. https://doi.org/10.1016/S0146-6380(98)00194-6
Groos AR, Akçar N, Yesilyurt S et al (2021) Nonuniform Late Pleistocene glacier fluctuations in tropical Eastern Africa. Sci Adv. https://doi.org/10.1126/sciadv.abb6826
Groos AR, Niederhauser J, Lemma B et al (2022) An hourly ground temperature dataset for 16 high-elevation sites (3493–4377gmga.s.l.) in the Bale Mountains, Ethiopia (2017–2020). Earth Syst Sci Data 14:1043–1062. https://doi.org/10.5194/essd-14-1043-2022
Hedberg O (1951) Vegetation belts of the east african mountains, 45th edn. Sven. Bot. Tidskr, Stockholm
Hedberg O (1964) Features of Afroalpine plant ecology. Swedish Science Press, Uppsala
Hemp A (2005) Climate change-driven forest fires marginalize the impact of ice cap wasting on Kilimanjaro. Glob Chang Biol 11:1013–1023. https://doi.org/10.1111/j.1365-2486.2005.00968.x
Hemp A, Beck E (2001) Erica excelsa as a fire-tolerating component of Mt. Kilimanjaro’s forests. Phytocoenologia 31:449–475. https://doi.org/10.1127/phyto/31/2001/449
Hillman JC (1986) Conservation in Bale Mountains National Park, Ethiopia. Oryx 20:89–94. https://doi.org/10.1017/S0030605300026314
Hillman JC (1988) The Bale Mountains National Park Area, Southeast Ethiopia, and its Management. Mt Res Dev 8:253–258
Jacob M, Annys S, Frankl A et al (2015) Tree line dynamics in the tropical African highlands—Identifying drivers and dynamics. J Veg Sci 26:9–20. https://doi.org/10.1111/jvs.12215
Johansson M (2013) Fire and grazing in subalpine heathlands and forests of Bale Fire ecology and traditional use of fire. Swedish University of Agricultural Sciences
Johansson M, Rooke T, Fetene M, Granström A (2010) Browser selectivity alters post-fire competition between Erica arborea and E. trimera in the sub-alpine heathlands of Ethiopia. Plant Ecol 207:149–160. https://doi.org/10.1007/s11258-009-9661-9
Johansson MU, Fetene M, Malmer A, Granström A (2012) Tending for cattle: traditional fire management in ethiopian montane heathlands. Ecol Soc. https://doi.org/10.5751/ES-04881-170319
Johansson MU, Frisk CA, Nemomissa S, Hylander K (2018) Disturbance from traditional fire management in subalpine heathlands increases Afro-alpine plant resilience to climate change. Glob Chang Biol 24:2952–2964. https://doi.org/10.1111/gcb.14121
Kessler M (2000) Observations on a human-induced fire event at a humid timberline in the Bolivian Andes. Ecotropica 6:89–93
Kidane Y, Stahlmann R, Beierkuhnlein C (2012) Vegetation dynamics, and land use and land cover change in the Bale Mountains, Ethiopia. Environ Monit Assess 184:7473–7489. https://doi.org/10.1007/s10661-011-2514-8
Kidane YO, Hoffmann S, Jaeschke A et al (2022) Ericaceous vegetation of the Bale Mountains of Ethiopia will prevail in the face of climate change. Sci Rep 12:1–18. https://doi.org/10.1038/s41598-022-05846-z
Körner C (2007) Climatic treelines: conventions, global patterns, causes. Erdkd 4:316–324. https://doi.org/10.3112/erdkunde.2007.04.02
Körner C (2012a) Alpine treelines: functional ecology of the global high elevation tree limits. Springer, Basel
Körner C (2012b) Treelines will be understood once the functional difference between a tree and a shrub is. Ambio 41:197–206. https://doi.org/10.1007/s13280-012-0313-2
Körner C, Farquhar GD, Wong SC (1991) Carbon isotope discrimination by plants follows latitudinal and altitudinal trends. Oecologia 88:30–40. https://doi.org/10.1007/BF00328400
Kron KA, Judd WS, Stevens PF et al (2002) Phylogenetic classification of Ericaceae: molecular and morphological evidence. Bot Rev 68:335–423. https://doi.org/10.1663/0006-8101(2002)068[0335:pcoema]2.0.co;2
Kuzyakov Y, Bogomolova I, Glaser B (2014) Biochar stability in soil: decomposition during eight years and transformation as assessed by compound-specific14C analysis. Soil Biol Biochem 70:229–236. https://doi.org/10.1016/j.soilbio.2013.12.021
Lemma B, Kebede Gurmessa S, Nemomissa S et al (2020) Spatial and temporal 2H and 18O isotope variation of contemporary precipitation in the Bale Mountains, Ethiopia. Isot Environ Health Stud 56:122–135. https://doi.org/10.1080/10256016.2020.1717487
Levin NE, Zipser EJ, Ceding TE (2009) Isotopic composition of waters from Ethiopia and Kenya: insights into moisture sources for eastern Africa. J Geophys Res Atmos 114:1–13. https://doi.org/10.1029/2009JD012166
Mark BG, Osmaston HA (2008) Quaternary glaciation in Africa: key chronologies and climatic implications. J Quat Sci 23:589–608. https://doi.org/10.1002/jqs.1222
Marshall JD, Brooks JR, Lajtha K (2007) Sources of variation in the stable isotopic composition of plants, 2nd edn. Blackwell Publishing, Hoboken
Mcguire AF, Kron KA (2011) Phylogenetic relationships of European and African Ericas. Int J Plant Sci 166:311–318
Mekonnen B, Zech W, Glaser B et al (2019) Chemotaxonomic patterns of vegetation and soils along altitudinal transects of the Bale Mountains, Ethiopia, and implications for paleovegetation reconstructions—Part I: stable isotopes and sugar biomarkers. E G Quat Sci J 68:177–188. https://doi.org/10.5194/egqsj-68-189-2019
Mekonnen B, Glaser B, Zech R et al (2022) Climate, vegetation and fire history during the past 18,000 years, recorded in high altitude lacustrine sediments on the Sanetti Plateau, Bale Mountains (Ethiopia). Prog Earth Planet Sci. https://doi.org/10.1186/s40645-022-00472-9
Miehe G, Miehe S (1994) Ericaceous forests and heathlands in the Bale Mountains of South Ethiopia: ecology and man’s impact. T. Warnke Verlag, Hamburg
Μohr P (1983) Volcanotectonic aspects of Ethiopian Rift evolution. In: Popoff, M., Tiercelin, J.-J. (Eds) Ancient Rifts and Troughs, Symposium of the French National Centre of Scientific Research (CNRS), Marseilles, pp. 175–189. Bull Volcanol 46–1:320
Natelhoffer KJ, Fry B (1988) Controls on natural nitrogen-15 and carbon-13 abundances in forest soil organic matter. Soil Sci Soc Am J 52:1633–1640. https://doi.org/10.2136/sssaj1988.03615995005200060024x
Oliver EGH (1989) The Ericoideae and the Southern African heathers. Bot J Linn Soc 101:319–327. https://doi.org/10.1111/j.1095-8339.1989.tb00167.x
Osmaston HA, Mitchell WA, Osmaston JAN (2005) Quaternary glaciation of the Bale Mountains Ethiopia. J Quat Sci. https://doi.org/10.1002/jqs.931
Schneider MPW, Hilf M, Vogt UF, Schmidt MWI (2010) Organic geochemistry the benzene polycarboxylic acid (BPCA) pattern of wood pyrolyzed between 200 °C and 1000 °C. Org Geochem 41:1082–1088. https://doi.org/10.1016/j.orggeochem.2010.07.001
Tiercelin JJ, Gibert E, Umer M et al (2008) High-resolution sedimentary record of the last deglaciation from a high-altitude lake in Ethiopia. Quat Sci Rev 27:449–467. https://doi.org/10.1016/j.quascirev.2007.11.002
Tiunov AV (2007) Stable isotopes of carbon and nitrogen in soil ecological studies. Biol Bull 34:395–407. https://doi.org/10.1134/S1062359007040127
Umer M, Lamb HF, Bonnefille R et al (2007) Late Pleistocene and Holocene vegetation history of the Bale Mountains, Ethiopia. Quat Sci Rev 26:2229–2246. https://doi.org/10.1016/j.quascirev.2007.05.004
Wesche K (2003) The importance of occasional droughts for Afroalpine landscape ecology. J Trop Ecol 19:197–207. https://doi.org/10.1017/S0266467403003225
Wesche K, Miehe G, Kaeppeli M (2000) The significance of fire for Afroalpine Ericaceous vegetation. Mt Res Dev 20:340–347. https://doi.org/10.1659/0276-4741(2000)020[0340:TSOFFA]2.0.CO;2
Wesche K, Cierjacks A, Assefa Y et al (2008) Recruitment of trees at tropical alpine treelines: Erica in Africa versus Polylepis in South America. Plant Ecol Divers 1:35–46. https://doi.org/10.1080/17550870802262166
Wolf M, Lehndorff E, Wiesenberg GLB et al (2013) Towards reconstruction of past fire regimes from geochemical analysis of charcoal. Org Geochem 55:11–21. https://doi.org/10.1016/j.orggeochem.2012.11.002
Yimer F, Ledin S, Abdelkadir A (2006) Soil organic carbon and total nitrogen stocks as affected by topographic aspect and vegetation in the Bale Mountains, Ethiopia. Geoderma 135:335–344. https://doi.org/10.1016/j.geoderma.2006.01.005
Zech M (2006) Evidence for Late Pleistocene climate changes from buried soils on the southern slopes of Mt. Kilimanjaro Tanzania. Palaeogeogr Palaeoclimatol Palaeoecol 242(3–4):303–312. https://doi.org/10.1016/j.palaeo.2006.06.008
Zech M, Bimüller C, Hemp A et al (2011) Human and climate impact on 15N natural abundance of plants and soils in high-mountain ecosystems: a short review and two examples from the Eastern Pamirs and Mt. Kilimanjaro. Isot Environ Health Stud 47:286–296. https://doi.org/10.1080/10256016.2011.596277
Acknowledgements
We are grateful to the Bale Mountains National Park, the Ethiopian Biodiversity Institute, and the Ethiopian Wildlife Conservation Authority for granting permission for scientific fieldwork and for the ease of access to the plant and soil samples utilized in this study. We would like to thank the Department of Plant Biology and Biodiversity Management at Addis Ababa University for its scientific cooperation. We also appreciate Heike Maennicke's invaluable assistance in the lab and Marianne Benesch for quantifying the stable isotopes. We thank Rudolf Sämann for the soil texture analysis. Betelhem Mekonnen also expressed gratitude for the assistance provided by the Katholischer Akademischer Ausländer-Dienst (KAAD). We appreciate the constructive comments and suggestions from the anonymous reviewers that helped to improve this manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. This research was funded by the German Research Foundation within the DFG Research Unit 'The Mountain Exile Hypothesis, grant number GL327/18-1, ZE844/10-1.
Author information
Authors and Affiliations
Contributions
WZ and BG conceived the study; WZ and BM collected the samples, BM and TB performed the laboratory analyses and analyzed the data; BM wrote the original manuscript draft.; WZ, BG, MZ, SN, TB, TB reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mekonnen, B., Glaser, B., Zech, M. et al. Factors determining the distribution of Erica patches on the Sanetti Plateau, Bale Mountains, Ethiopia. Alp Botany 133, 135–147 (2023). https://doi.org/10.1007/s00035-023-00295-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00035-023-00295-4