Abstract
Caspase-8, an aspartate-specific cysteine protease that primarily functions as an initiator caspase to induce apoptosis, can downregulate innate immunity in part by cleaving RIPK1 and IRF3. However, patients with caspase-8 mutations or deficiency develop immunodeficiency and are prone to viral infections. The molecular mechanism underlying this controversy remains unknown. Whether caspase-8 enhances or suppresses antiviral responses against influenza A virus (IAV) infection remains to be determined. Here, we report that caspase-8 is readily activated in A549 and NL20 cells infected with the H5N1, H5N6, and H1N1 subtypes of IAV. Surprisingly, caspase-8 deficiency and two caspase-8 inhibitors, Z-VAD and Z-IETD, do not enhance but rather downregulate antiviral innate immunity, as evidenced by decreased TBK1, IRF3, IκBα, and p65 phosphorylation, decreased IL-6, IFN-β, MX1, and ISG15 gene expression; and decreased IFN-β production but increased virus replication. Mechanistically, caspase-8 cleaves and inactivates CYLD, a tumor suppressor that functions as a deubiquitinase. Caspase-8 inhibition suppresses CYLD cleavage, RIG-I and TAK1 ubiquitination, and innate immune signaling. In contrast, CYLD deficiency enhances IAV-induced RIG-I and TAK1 ubiquitination and innate antiviral immunity. Neither caspase-3 deficiency nor treatment with its inhibitor Z-DEVD affects CYLD cleavage or antiviral innate immunity. Our study provides evidence that caspase-8 activation in two human airway epithelial cell lines does not silence but rather enhances innate immunity by inactivating CYLD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Host cells detect intracellular viral infection mainly by sensing viral RNA and DNA [1]. Toll-like receptor 3 (TLR3) recognizes viral double-stranded RNA (dsRNA). TLR3 recruits the adaptor protein TRIF (TIR domain-containing adaptor inducing interferon-β) to activate two E3 ubiquitin ligases, TRAF3 (TNF receptor-associated factor 3) and TRAF6 [1, 2]. Retinoic acid-inducible gene-I (RIG-I) is another RNA sensor that preferentially binds single-stranded, 5’-phosphorylated RNA [2,3,4]. RIG-I interacts with the mitochondrial antiviral signaling protein (MAVS), an adaptor that activates TRAF3 and TRAF6 [2, 3]. TRAF3 activates TANK-binding kinase 1 (TBK1) to phosphorylate interferon regulatory factor 3 (IRF3) [5, 6]. TRAF3 also activates IKKε to phosphorylate IKK-γ (NEMO), which phosphorylates and activates NF-κB [5, 6]. TRAF6 cooperates with RIPK1 to activate TGF-β-activated kinase 1 (TAK1), which activates IKKα/β to phosphorylate IκB and the p65 subunit of NF-κB [1, 2, 6, 7]. Activated IRF3 and NF-κB translocate to the nucleus and play important roles in regulating type I interferon production [8]. In addition, MAVS also activates TBK1 or IKKε through the RIPK1-FADD-TRADD (tumor necrosis factor receptor 1-associated death domain protein) complex [9]. RIPK1 or FADD deficiency inhibits vesicular stomatitis virus (VSV)- and poly(I:C)-induced interferon (IFN) expression [10]. Both TLR3 and RIG-I regulate a complex network of innate signaling pathways.
Caspase-8 is a member of the cysteine protease family that primarily functions as an initiator caspase to induce apoptosis [11]. Accumulating evidence suggests that caspase-8 can positively or negatively regulate innate antiviral immunity [11]. Caspase-8 cleaves RIPK1 to disrupt its interaction with MAVS and TBK1, thereby suppressing the activation of the TBK1-IRF3 pathway [10, 12]. Moreover, caspase-3, a downstream effector caspase of caspase-8, can cleave IRF3, MAVS, and cyclic GMP–AMP synthase (cGAS), further dampening the activation of innate immunity [13]. Caspase-8 can cleave and inactivate CYLD, a tumor suppressor protein that deubiquitinates the M1 and K63 ubiquitin chains in RIG-I and TAK1 [14,15,16,17]. Thus, caspase-8 activation may enhance the activation of the TBK1-IRF3 and IKK-NF-κB pathways by preventing RIG-I and TAK1 deubiquitination [11, 18]. Indeed, several studies have shown that caspase-8 is required for RIG-I-, TLR4-, and TCR-induced NF-κB activation [18,19,20,21,22]. Moreover, individuals with caspase-8 deficiency or mutations develop immunodeficiency characterized by recurrent sinopulmonary and herpes simplex virus (HSV) infections and poor responses to immunization [22]. Caspase-8 appears to play an opposing role in innate antiviral immunity [23].
Influenza A virus (IAV) is a highly contagious pathogen that belongs to the Orthomyxoviridae family. H1N1 and H3N2 viruses are the most prevalent subtypes of human infections [24]. Avian H5N1 and its derivative H5Nx subtypes can cause sporadic and fatal infections in humans [25, 26]. IAV, a segmented, single-stranded RNA virus, replicates rapidly in airway epithelial cells [27]. The binding of viral RNA to Z-DNA binding protein 1 (ZBP1) activates caspase-8 through the ZBP1-RIPK3-RIPK1-FADD-caspase-8 complex [28, 29]. However, how caspase-8 activation in IAV-infected airway epithelial cells regulates antiviral responses remains to be determined. Here, we report that caspase-8 activation by IAV infection inactivates CYLD, prevents RIG-I and TAK1 deubiquitination, and enhances antiviral responses. RIPK1, whether cleaved by caspase-8, is dispensable for IAV-induced IRF3 and NF-κB activation. Our study provides mechanistic insights into the mechanisms by which caspase-8 regulates innate antiviral responses against IAV infection in human airway epithelial cells.
Results
IAV activates caspase-8 to cleave CYLD
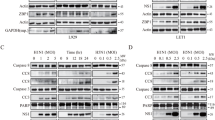
IAV infection readily activates caspase-8 to induce apoptosis [30]. CYLD is a deubiquitinase that can be cleaved and inactivated by caspase-8 [14, 15]. We first tested whether caspase-8 activation by IAV infection also cleaved CYLD in A549 cells, a human lung adenocarcinoma cell line, and NL20, a human noncancerous bronchial epithelial cell line. Two H5N1 strains (SY & CK10) and one H5N6 strain (Y6) dose- and time-dependently induced caspase-8 cleavage to generate a 43-kDa protein and induced CYLD cleavage to generate a 25-kDa protein in A549 and NL20 cells (Fig. 1A & B). Similar observations were made in A549 cells infected with the H1N1 virus (PR8) (Fig. S1). These observations suggest that CYLD is a substrate that is cleaved upon caspase-8 activation.
IAV induces caspase-8 and CYLD cleavage. (A & B) A549 and NL20 cells were infected with the indicated MOIs of the H5N1 (SY & CK10) and H5N6 (Y6) viruses for 24 h (A) or infected with the indicated virus at an MOI of 1 and incubated for the indicated durations (B). Cell lysates were analyzed for caspase-8 and CYLD cleavage by Western blotting. The viral NS1 protein was used as an infection control; β-Actin was used as a loading control. The experiment was repeated three times with samples from 5 batches. CASP8, caspase-8
Inhibition of caspase activity blocks CYLD cleavage and activation of the NF-κB pathway
CYLD deubiquitinates K63 ubiquitin chains in RIG-I, TAK1, NEMO, and TBK1 and suppresses innate immunity [14, 15]. We hypothesized that the inhibition of CYLD cleavage by a caspase inhibitor may attenuate the activation of the TLR3 and RIG-I pathways. We first tested whether Z-VAD, a pancaspase inhibitor, could downregulate the IAV-activated NF-κB pathway. The three H5 strains induced TAK1, IκB, and p65 phosphorylation in A549 and NL20 cells (Fig. 2A). Z-VAD dose-dependently blocked caspase-8 and CYLD cleavage as well as TAK1, IκB, and p65 phosphorylation in both A549 and NL20 cells (Fig. 2B). Immunofluorescence (IF) revealed that p65 was present in the cytoplasm of uninfected or Z-VAD-treated A549 cells (Fig. 2C). IAV infection induced p65 nuclear translocation, which was partially blocked by Z-VAD (Fig. 2C). Western blot analysis revealed that IAV infection increased the levels of phosphorylated p65 in the nuclear fraction of A549 cells, which was partially prevented by Z-VAD (Fig. 2D). A luciferase reporter assay revealed that IAV infection significantly increased NF-κB binding site-driven luciferase expression, which was largely inhibited by Z-VAD (Fig. 2E). These observations collectively suggest that caspase activation in IAV-infected cells enhances the activation of TAK1 and its downstream NF-κB transcription factor.
The pancaspase inhibitor Z-VAD inhibits the activation of the NF-κB pathway. (A) A549 and NL20 cells were infected with H5N1 (SY & CK10) or H5N6 (Y6) at the indicated MOIs for 24 h. Cell lysates were analyzed for the expression of proteins in the NF-κB pathway by Western blot. (B) A549 and NL20 cells infected with the H5N1 (SY) virus at an MOI of 1 were incubated for 24 h in the absence or presence of the indicated concentrations of Z-VAD. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated three times with samples from 5 batches. (C) A549 cells were infected with H5N1 (SY) virus at an MOI of 1 and then incubated for 24 h in the absence or presence of Z-VAD (100 µM). The cells were fixed and stained with an antibody against p65, followed by nuclear staining with DAPI. Scale bar, 25 μm. (D) A549 cells were infected with 1 MOI of H5N1 (SY) virus and then incubated for 24 h in the absence or presence of Z-VAD (100 µM). Cytosolic and nuclear fractions were analyzed for p65 phosphorylation and then reprobed for total protein by Western blot. GAPDH and Sp1 were detected as loading controls for the cytosolic and nuclear fractions, respectively. The experiment was repeated twice with similar results. (E) A549 cells were transfected with the NF-κB-driven luciferase reporter gene (pGL3/NF-κB-Luc). After incubation for 24 h, the cells were infected with H5N1 (SY) virus at an MOI of 1 and then incubated for 24 h in the absence or presence of Z-VAD (100 µM). The cells were harvested and analyzed for luciferase activity. The results represent the mean ± SD of triplicate data from one of three independent experiments with similar results. **P < 0.01
Inhibition of caspase activity suppresses the activation of the TBK1-IRF3 pathway
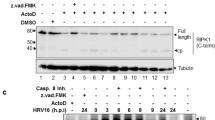
We next tested whether Z-VAD could also inhibit the activation of the TBK1-IRF3 pathway in IAV-infected cells. IAV infection increased TBK1 and IRF3 phosphorylation in A549 and NL20 cells in a dose-dependent manner (Fig. 3A). Z-VAD dose-dependently suppressed IAV-induced caspase-8 cleavage and the phosphorylation of TBK1 and IRF3 in A549 and NL20 cells (Fig. 3B). Z-VAD inhibited caspase-8 and CYLD cleavage as well as TBK1 and IRF3 phosphorylation in A549 cells infected with IAV for 16–24 h (Fig. S2A). In contrast, Z-VAD partially inhibited CYLD cleavage but dramatically increased TBK1 and IRF3 phosphorylation in murine RAW264.7 (Fig. S2B) and human J774A.1 (Fig. S2C) macrophage lines infected with IAV for various lengths of time. Z-VAD also decreased the levels of the viral NS1 protein of IAV in these two cell lines (Fig. S2B & C). These observations suggest that the ability of Z-VAD to repress or enhance TBK1 and IRF3 phosphorylation is cell-type specific. To determine the physiological relevance of caspase-8 activation in regulating innate immunity, we examined the effect of Z-VAD on primary human alveolar epithelial cells and found that Z-VAD was able to effectively inhibit TBK1 and IRF3 phosphorylation in these cells (Fig. S2D). Z-VAD also effectively inhibited caspase-8, caspase-3, and CYLD cleavage in these cells (Fig. S2D). We speculate that the effect of caspase-8 activation on innate antiviral immunity is universal in human airway epithelial cells.
The pancaspase inhibitor Z-VAD inhibits the activation of the TBK1-IRF3 pathway. (A) A549 and NL20 cells were infected with H5N1 (SY & CK10), H5N6 (Y6), or H1N1 (PR8) at the indicated MOIs for 24 h. Cell lysates were analyzed for the expression of proteins in the TBK1-IRF3 pathway by Western blotting. The experiment was repeated twice with two batches of samples. (B) A549 and NL20 cells infected with the H5N1 (SY) virus at an MOI of 1 were incubated for 24 h in the absence or presence of the indicated concentrations of Z-VAD. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated three times with samples from 5 batches. (C) A549 cells were infected with H5N1 (SY) virus at an MOI of 1 and then incubated for 24 h in the absence or presence of Z-VAD (100 µM). The cells were fixed and stained with an antibody against IRF3. Scale bar, 25 mm. (D) A549 cells were infected with 1 MOI of H5N1 (SY) virus and then incubated for 24 h in the absence or presence of Z-VAD (100 µM). The cytosolic and nuclear fractions were analyzed for IRF3 phosphorylation and then reprobed for total protein by Western blot. GAPDH and Sp1 were detected as loading controls for the cytosolic and nuclear fractions, respectively. The experiment was repeated twice with similar results. (E) A549 cells were transfected with the IRF3 binding site- or IFN-β promoter-driven luciferase reporter gene (pGL3/IRF3-Luc & pGL3/IFN-β-Luc). After incubation for 24 h, the cells were infected with H5N1 (SY) virus at an MOI of 1 and then incubated for 24 h in the absence or presence of Z-VAD (100 µM). The cells were harvested and analyzed for luciferase activity. The results represent the mean ± SD of triplicate data from one of three independent experiments with similar results. **p < 0.01. (F) A549 cells were left uninfected or infected with H5N1 (SY) virus at an MOI of 1 and then incubated for 24 h in the absence or presence of Z-VAD (100 µM). Total RNA was extracted and quantified, followed by RT‒PCR analysis of the IL-6, IFN-β, MX1, and ISG15 genes. The results are presented as the means ± SDs of three independent experiments with similar results. *P < 0.05; **P < 0.01
IF staining revealed that IRF3 signals were located in the cytoplasm of uninfected or Z-VAD-treated A549 cells. IAV infection induced IRF3 translocation into the nucleus, which was partially inhibited by Z-VAD (Fig. 3C). Western blot analysis revealed that IAV infection increased the level of phosphorylated IRF3 in the nuclear fraction, which was partially inhibited by Z-VAD (Fig. 3D). A luciferase reporter assay revealed that IAV infection significantly increased IRF3- or IFN-β promoter-driven luciferase expression, which was partially inhibited by Z-VAD (Fig. 3E). RT‒PCR revealed that IAV infection dramatically increased IL-6, IFN-β, MX1, and ISG15 mRNA levels in A549 cells, and these effects were partially inhibited by Z-VAD (Fig. 3F).
Caspase-8 inhibition suppresses innate antiviral immunity
To determine whether the inhibition of the NF-κB and TBK1 pathways by Z-Vad was due to the inhibition of caspase-8 or other caspases, we tested whether Z-IETD, a caspase-8-specific inhibitor, could also inhibit the activation of innate immunity in IAV-infected cells. Indeed, Z-IETD blocked H5N1-induced caspase-8, caspase-3, and CYLD cleavage (Fig. 4A) and TBK1, IRF3, and p65 phosphorylation in A549 and NL20 cells (Fig. 4B). Consistently, caspase-8 deficiency decreased IAV-induced caspase-3 and CYLD cleavage and TBK1, IRF3, TAK1, and p65 phosphorylation and prevented IAV-induced RIG-I expression in A549 cells (Fig. 4C & D). Similar observations were made in NL20 cells (Fig. S3A & B). Caspase-8 deficiency also abrogated Sendai virus (SeV)-induced TBK1 and IRF3 phosphorylation in A549 and NL20 cells (Fig. S3C & D). In contrast, caspase-8 deficiency enhanced IAV-induced TBK1 and IRF3 phosphorylation but decreased viral NS1 protein expression in LET1 cells, a murine alveolar epithelial cell line (Fig. S3E). These observations suggest that caspase-8 activation in IAV-infected cells enhances the activation of the NF-κB and IRF3 pathways.
Caspase-8 inhibition suppresses the activation of the NF-κB and IRF3 pathways. (A & B) A549 and NL20 cells infected with 1 MOI of the H5N1 (SY) virus for 24 h were incubated in the absence or presence of the indicated concentrations of Z-IETD. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated twice with the samples from two separate batches. CASP8, caspase-8; CASP3, caspase-3. (C & D) pLenti-V2-transfected wild-type (WT) control and Caspase-8-deficient A549 cells were infected with the indicated MOI of H5N1 (SY) virus for 24 h. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated three times, each with a separate batch of cell lysates. ΔCASP8, caspase-8 deficiency. (E-H) WT control and caspase-8-deficient A549 cells were infected with H5N1 (SY) at an MOI of 1 for 24 h. The cells were fixed and stained with an antibody against p65 (E) or an antibody against IRF3 (G), followed by nuclear staining with DAPI. Scale bar, 25 mm. The percentage of cells with p65 (F) or IRF3 (H) nuclear staining was calculated. The results are presented as the means ± SDs of three independent experiments. **P < 0.01
Caspase-8 deficiency blocked IAV-induced IRF3 and p65 nuclear translocation (Fig. 4E-H). A luciferase reporter assay revealed that the caspase-8 inhibitor Z-IETD (Fig. 5A) or caspase-8 deficiency (Fig. 5B) decreased NF-κB, IRF-3-, or IFN-β promoter-driven luciferase gene expression. RT‒PCR analysis revealed that Z-IETD or caspase-8 deficiency significantly decreased the mRNA levels of IL-6, IFN-β, MX1, and ISG15 in IAV-infected A549 cells (Fig. 5C & D). Caspase-8 deficiency decreased the levels of basal and IAV-induced JAK1 phosphorylation (Fig. 5E) and IFN-β production (Fig. 5F) but enhanced IAV replication in A549 cells infected with various MOIs of H5N1 virus (Fig. 5G). Similar observations were made in caspase-8-deficient NL20 cells (Fig. S4A-C). These observations collectively suggest that caspase-8 activation enhances innate antiviral immunity by promoting IFN production.
Caspase-8 inhibition suppresses innate antiviral immunity. (A) A549 cells were transfected with the luciferase reporter gene driven by the NF-κB or IRF3 binding sites or by the IFN-β promoter. After incubation for 24 h, the cells were infected with H5N1 (SY) virus at an MOI of 1 and then incubated for 24 h in the absence or presence of Z-IETD (100 µM). The cells were harvested and analyzed for luciferase activity. **p < 0.01. (B) WT control and caspase-8-deficient A549 cells were transfected with the luciferase reporter gene driven by the NF-κB or IRF3 binding sites or by the IFN-β promoter. After incubation for 24 h, the cells were infected with 1 MOI of H5N1 (SY) virus for another 24 h, and the cells were harvested and analyzed for luciferase activity. The results represent the mean ± SD of triplicate data from one of three independent experiments with similar results. **P < 0.01. (C) A549 cells were left uninfected or infected with H5N1 (SY) virus at an MOI of 1 and then incubated for 24 h in the absence or presence of Z-IETD (100 µM). (D) WT control and caspase-8-deficient A549 cells were infected with H5N1 (SY) at an MOI of 1 for 24 h. Total RNA was extracted and quantified, followed by RT-PCR analysis of IL-6, IFN-β, MX1, and ISG15 mRNA levels. The results are presented as the means ± SDs of three independent experiments. **P < 0.01. (E) WT control and caspase-8-deficient A549 cells were infected with H5N1 (SY) at the indicated MOIs for 24 h. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated twice with similar results. (F and G) WT control and caspase-8-deficient A549 cells were infected with 1 MOI of H5N1 virus (F) or the indicated MOI of H5N1 virus (G) for 24 h. Conditioned media were collected and analyzed for IFN-b concentrations by using an ELISA kit. The virus titers in the conditioned media were analyzed by measuring TCID50 values. The IFN-β concentrations and virus titers are presented as the means ± SDs of three independent experiments. **P < 0.01
Caspase-3 deficiency does not affect IAV-elicited innate immunity
Caspase-3 cleaves several proteins involved in innate immunity, including cGAS, MAVS, and IRF3 [13, 31]. Since caspase-3 is an effector caspase downstream of caspase-8, we also evaluated the role of caspase-3 in regulating innate immunity in IAV-infected cells. Z-DEVD, an inhibitor of caspase-3 with relatively low specificity [32], blocked IAV-induced caspase-3 cleavage but had little effect on caspase-8 and CYLD cleavage. Z-DEVD had almost no effect on TBK1, IRF3, or p65 phosphorylation (Fig. 6A). A lack of effect of caspase-3 on innate immunity was confirmed in caspase-3-deficient cell lines. Caspase-3 deficiency had little effect on caspase-8 cleavage and no effect on CYLD cleavage or TBK1, IRF3, or p65 phosphorylation in A549 and NL20 cells (Fig. 6B). IAV infection equally cleaved CYLD in wild-type and caspase-3-deficient A549 cells (Fig. S5A). IAV infection cleaved CYLD in caspase-3-deficient but not in caspase-8-deficient A549 cells (Fig. S5B). Z-DEVD and caspase-3 deficiency did not affect IAV-induced luciferase gene expression driven by the IFN-β promoter or by the IRF3 or NF-κB binding sites (Fig. 6C & D). Z-DEVD also did not affect the mRNA levels of IL-6, IFN-β, MX1, or ISG15 in IAV-infected A549 cells (Fig. 6E) or on IFN-β production (Fig. 6F). These observations collectively suggest that caspase-3 activation does not affect innate antiviral immunity.
Caspase-3 inhibition does not affect the activation of the NF-κB and IRF3 pathways. (A) A549 and NL20 cells infected with the H5N1 (SY) virus at an MOI of 1 for 24 h were incubated in the absence or presence of the indicated concentrations of Z-DEVD. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated twice with similar results. (B) WT control and caspase-3-deficient A549 and NL20 cells were infected with the indicated MOI of the H5N1 (SY) virus for 24 h. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated three times, each with three batches of cell lysates. (C) A549 cells were transfected with the luciferase reporter gene driven by the NF-κB or IRF3 binding sites or by the IFN-β promoter. After incubation for 24 h, the cells were infected with H5N1 (SY) virus at an MOI of 1 and then incubated for 24 h in the absence or presence of Z-DEVD (80 µM). The cells were harvested and analyzed for luciferase activity. ns, not significant. (D) WT control and caspase-3-deficient A549 cells were transfected with the NF-κB, IRF3 and IFN-β promoter-driven luciferase reporters. After incubation for 24 h, the cells were infected with 1 MOI of H5N1 (SY) virus for another 24 h, and the cells were harvested and analyzed for luciferase activity. The results represent the mean ± SD of triplicate data from one of three independent experiments with similar results. ns, not significant. (E) A549 cells were left uninfected or infected with H5N1 (SY) virus at an MOI of 1 and then incubated for 24 h in the absence or presence of Z-EDVD (80 µM). Total RNA was extracted and quantified, followed by RT‒PCR analysis of the IL-6, IFN-β, MX1, and ISG15 genes. The results are presented as the means ± SDs of three independent experiments. ns, not significant. (F) WT control and caspase-3-deficient A549 cells were infected with the H5N1 virus (1 MOI) for 24 h. Conditioned media were collected and analyzed for the concentration of IFN-b by using an ELISA kit. The results are presented as the means ± SDs of three independent experiments. **P < 0.01
CYLD deficiency enhances IAV-elicited innate immunity
CYLD removes K63 ubiquitin from TAK1 and RIG-I to inhibit their activation [16, 33]. To determine whether CYLD cleavage was responsible for caspase-8-enhanced innate immunity, we tested whether CYLD deficiency also enhanced IAV-induced immune responses. Indeed, CYLD deficiency in A549 cells increased IAV-induced TAK1, p65, TBK1, and IRF3 phosphorylation compared to that in wild-type cells (Fig. 7A & B). Unexpectedly, CYLD deficiency also inhibited IAV-induced caspase-3 cleavage in A549 cells but did not affect caspase-8 cleavage (Fig. 7A). Similar observations were made in NL20 cells (Fig. S6A & B). IAV infection induced RIG-I expression to a significantly greater extent in CYLD-deficient A549 cells than in control A549 cells (Fig. 7B). Re-expression of CYLD in CYLD-deficient A549 cells blocked IAV-induced TBK1 and IRF3 phosphorylation and RIG-I expression (Fig. 7C), whereas re-expression of an uncleavable CYLD mutant (D215A) further reduced IAV-induced TBK1 and IRF3 phosphorylation and decreased the levels of IAV-induced RIG-I expression compared to those in CYLD-deficient cells transfected with the wild-type CYLD expression vector (Fig. 7D).
CYLD deficiency enhances the activation of the NF-κB and IRF3 pathways. (A & B) WT control and CYLD-deficient A549 cells were infected with the indicated MOI of H5N1 (SY) virus for 24 h. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated twice with the samples from two separate batches. (C) CYLD-deficient A549 cells were transfected with pCAGGS or the vector encoding CYLD. After incubation for 24 h, the cells were left uninfected or infected with the indicated MOI of H5N1 (SY) virus for another 24 h. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated twice with similar results. (D) CYLD-deficient A549 cells were first transfected with a human wild-type CYLD gene or a mutant CYLD gene (D215A). After incubation for 24 h, the cells were infected with H5N1 (SY) at the indicated MOI and then incubated for another 24 h. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. *, the truncated 25-kDa CYLD protein. (E-H) WT control and CYLD-deficient A549 cells were infected with H5N1 (SY) at an MOI of 1 for 24 h. The cells were fixed and stained with an antibody against p65 (E) or an antibody against IRF3 (G), followed by nuclear staining with DAPI. Scale bar, 25 μm. The percentage of cells with p65 (F) and IRF3 (G) nuclear staining was calculated. The results are presented as the means ± SDs of three independent experiments. **p < 0.01
We further investigated the effect of CYLD deficiency on p65 and IRF3 activation, as well as their transcriptional activity. IAV-induced p65 and IRF3 nuclear translocation were much more prominent in CYLD-deficient A549 cells than in their vector controls (Fig. 7E-H). A luciferase reporter assay revealed that NF-κB-, IRF3-, and IFN-β promoter-driven luciferase activity was much greater in IAV-infected CYLD-deficient A549 cells than in vector controls (Fig. 8A). The levels of IL-6, IFN-β, ISG15, and MX1 mRNA were much greater in the IAV-infected CYLD-deficient A549 cells than in their wild-type counterparts (Fig. 8B). CYLD deficiency enhanced IAV-induced JAK1 phosphorylation in A549 cells (Fig. 8C) and IFN-β production (Fig. 8D). Notably, the basal level of JAK1 phosphorylation appeared to be greater in CYLD-deficient A549 cells than in wild-type cells (Fig. 8C). The titers of IAV in CYLD-deficient A549 cells were significantly lower than those in their wild-type counterparts (Fig. 8E). Similar observations were made in NL20 cells (Fig. S6C-E). These observations collectively suggest that caspase activation enhances innate antiviral immunity by inactivating CYLD.
CYLD deficiency enhances IAV-elicited innate immunity. (A) WT control and CYLD-deficient A549 cells were transfected with the luciferase reporter gene driven by the NF-κB or IRF3 binding sites or by the IFN-β promoter. After incubation for 24 h, the cells were infected with 1 MOI of H5N1 (SY) virus for another 24 h, and the cells were harvested and analyzed for luciferase activity. The results represent the mean ± SD of triplicate data from one of three independent experiments with similar results. **p < 0.01. (B) WT control and CYLD-deficient A549 cells were infected with H5N1 (SY) at an MOI of 1 for 24 h. Total RNA was extracted and quantified, followed by RT‒PCR analysis of the IL-6, IFN-β, MX1, and ISG15 genes. The results are presented as the means ± SDs of three independent experiments. **P < 0.01. (C) WT control and CYLD-deficient A549 cells were infected with the indicated MOI of the H5N1 (SY) virus for 24 h. Cell lysates were analyzed for the expression of the indicated proteins by Western blot. The experiment was repeated twice with similar results. The relative phosphorylation levels were analyzed by quantifying the density of the phosphorylated protein bands normalized to their corresponding total proteins. The results are presented in a bar graph. The data are presented as the means ± SDs of three independent experiments. *p < 0.05, **p < 0.01, compared to the uninfected controls. (D and E) WT control and CYLD-deficient A549 and NL20 cells were infected with the H5N1 virus (1 MOI) for 24 h. Conditioned media were collected and analyzed for IFN-b concentrations by using an ELISA kit. The virus titers in the conditioned media were analyzed by measuring TCID50 values. The IFN-β concentrations and virus titers are presented as the means ± SDs of three independent experiments. **P < 0.01
Caspase-8 enhances RIG-I and TAK1 ubiquitination by inactivating CYLD
CYLD antagonizes antiviral immunity in part by deubiquitinating RIG-I and TAK1 [14]. We hypothesized that blocking CYLD cleavage by inhibiting caspase-8 activity would enhance RIG-I and TAK1 ubiquitination. The levels of total protein ubiquitination were slightly greater in the IAV-infected A549 cells than in the mock-infected control cells (Fig. 9A & C). Z-VAD further increased protein ubiquitination in IAV-infected A549 cells (Fig. 9A & C). FLAG immunoprecipitation (IP) revealed that IAV infection dramatically increased RIG-I (Fig. 9A & B) and TAK1 (Fig. 9C & D) ubiquitination in A549 cells, which was blocked by Z-VAD (Fig. 9A & C). Consistent with these observations, caspase-8 deficiency blocked IAV-induced RIG-I and TAK1 ubiquitination, whereas CYLD deficiency increased RIG-I and TAK1 ubiquitination in uninfected and IAV-infected A549 cells (Fig. 9B & D). The effect of caspase-8 activation on RIG-I and TAK1 K63 ubiquitination was confirmed in A549 cells transfected with an HA-tagged Lys63-specific ubiquitin gene (HA-Ub-K63O). Z-VAD and caspase-8 deficiency both reduced RIG-I and TAK1 K63 ubiquitination (Fig. S7A-D), whereas CYLD deficiency enhanced RIG-I and TAK1 K63 ubiquitination in A549 cells (Fig. S7A-D). These observations collectively suggest that caspase-8 activation enhances innate antiviral immunity by inactivating CYLD and enhancing TAK1 and RIG-I K63 ubiquitination.
Caspase-8 enhances RIG-I and TAK1 ubiquitination by inactivating CYLD. (A-D) A549 cells were cotransfected with the expression vector encoding the HA-ubiquitin gene plus the FLAG-RIG-I gene (A) or the FLAG-TAK1 gene (C). pLenti-V2-transfected WT control and caspase-8- or CYLD-deficient A549 cells were cotransfected with the expression vector encoding the HA-ubiquitin gene plus the FLAG-RIG-I gene (B) or the FLAG-TAK1 gene (D). After incubation for 24 h, the cells were left uninfected or infected with H5N1 (SY) virus (1 MOI) and then incubated for another 24 h. Cell lysates were prepared and immunoprecipitated with an antibody against FLAG. Whole-cell lysates (WCLs) and immunoprecipitates were analyzed with antibodies against FLAG and HA. The data represent one of three independent experiments. (E) Schematic model of caspase-8-mediated immune activation. Influenza A virus activates TBK1 and TAK1 through RIG-I and TRL3, which then activate IRF3 and NF-κB. IAV infection activates caspase-8 in a ZBP1-dependent (NL20 cells) and ZBP1-independent (A549 cells) manner. Activated caspase-8 cleaves and inactivates CYLD, a deubiquitinase that removes the K63 sidechains of RIG-I and TAK1. Increased RIG-I and TAK1 ubiquitination enhances the activation of the TBK1-IRF3 and TAK1-NF-κB pathways, leading to strong antiviral responses such as increased IFN and ISG gene expression and the restriction of virus replication
Discussion
Caspase-8 and caspase-3 suppress the antiviral response by cleaving several proteins involved in innate immunity, including RIPK1, IRF3, MAVS, and cGAS [34,35,36]. However, ample evidence suggests that caspase-8 can positively regulate innate immunity [11, 23]. Our present study aimed to investigate the role of caspase-8 in regulating the antiviral response in IAV-infected cells. We provide evidence that caspase-8 enhances IAV-induced innate immunity by inactivating CYLD and preventing RIG-I and TAK1 K63 deubiquitination in two human airway epithelial cell lines. Our findings highlight the complexity of caspase regulation of innate antiviral immunity.
Caspase-8 primarily functions to execute extrinsic apoptosis [37]. However, numerous studies have suggested that caspase-8 can negatively regulate innate immunity. For example, Rajput et al. [12] reported that caspase-8 cleaves RIPK1 to produce a small 25-kD fragment that competes with full-length RIPK1 for RIG-I binding, leading to the inhibition of TBK1-IRF3 activation in Sendai virus-induced MEFs and HeLa cells. Wang et al. [10] reported that caspase-8 cleaves RIPK1 to disrupt the interaction between RIPK1 and TBK1, thus inhibiting TBK1 activation and antiviral responses. In contrast, caspase-8 knockdown in HEK293 cells and caspase-8 deficiency in MEFs reduce the expression of dsRNA-induced inflammatory cytokine genes [18]. Patients with caspase-8 deficiency or mutations, which are associated with autoimmune lymphoproliferative syndrome (ALPS), develop immunodeficiency and are prone to human herpesvirus infections [22, 38,39,40]. Caspase-8 is also required for RIG-I-, TLR4-, and TCR-induced NF-κB activation [18,19,20,21,22]. Whether caspase-8 suppresses or enhances innate immunity seems to be cell type-specific and context dependent.
Our present study showed that caspase-8 deficiency and treatment with caspase-8 inhibitors decreased innate antiviral responses in A549 and NL20 cells infected with IAV. Caspase-8 deficiency also blocked SeV-induced TBK1 and IRF3 phosphorylation in these two human cell lines (Fig. S3C), suggesting that caspase-8 can enhance innate antiviral immunity. In contrast, caspase-8 deficiency in LET1 cells, a murine alveolar epithelial cell line, enhanced IAV-induced TBK1 and IRF3 phosphorylation (Fig. S3E). An in-depth investigation revealed that IAV infection indeed increased RIPK1 cleavage in LET1 cells and NL20 cells (Fig. S8A). However, caspase-8 deficiency blocked RIPK1 cleavage in LET1 but not NL20 cells (Fig. S8A). This finding suggested that RIPK1 could be cleaved in NL20 cells by another caspase, caspase-10, which is expressed in humans but not in rodents [11]. Furthermore, RIPK1 deficiency attenuated IAV-induced TBK1 and IRF3 phosphorylation in LET1 cells but not in NL20 cells (Fig. S8C), suggesting that RIPK1 is required for RIG-I-mediated TBK1 activation in LET1 cells but not in NL20 and A549 cells. However, why RIPK1 plays a different role in TBK1 activation in these two cell lines remains to be determined. Wang et al. [34] recently reported that both human and murine full-length RIPK1 can bind TBK1, whereas the truncated RIPK1 fragment cleaved by caspase-8 cannot bind TBK1. These authors further demonstrated that RIPK1 is required for optimal TBK1 and IRF3 phosphorylation in murine bone marrow-derived monocytes. Whether RIPK1 is also required for TBK1 phosphorylation in human monocytes remains to be investigated.
In addition to cleaving RIPK1 and negatively regulating innate immunity, caspase-8 also downregulates antiviral responses by cleaving and inactivating IRF3 [11, 13]. Sears et al. [41] reported that caspase-8 can directly cleave IRF3 in IRF3-transfected P2.1 cells, a subclone of HT1080 cells that express little endogenous IRF3. We found that IAV infection did not cleave IRF3 in LET1 cells but did not affect IRF3 in NL20 cells, which could not be blocked by caspase-8 deficiency (Fig. S8C). This finding suggested that IRF3 is not cleaved by caspase-8 or by downstream caspase-3. Furthermore, the amount of cleaved IRF3 in NL20 cells was lower than that of full-length IRF3 (Fig. S5A). IAV infection did not lead to IRF3 cleavage in A549 cells (Fig. S8C). These observations suggest that IAV-activated caspase-8 cannot negatively regulate antiviral responses in A549 and NL20 cells because caspase-8 has no effect on RIPK1 or IRF3 cleavage and that RIPK1 plays no role in the activation of the TBK1-IRF3 pathway.
CYLD is a deubiquitinase that removes K63 and M1 polyubiquitin chains from RIG-I, TAK1, NEMO, TRAF2, and TRAF6 [14]. CYLD suppresses NF-κB activation induced by TLRs, T and B-cell antigen receptors, and the TNF-α receptor [14]. CYLD silencing activates NF-κB to enhance HIV replication in Jurkat cells in vitro [42]. CYLD also deubiquitinates RIG-I and TBK1 to suppress Sendai virus-induced antiviral responses [16]. CYLD overexpression decreased RIG-I, TBK1, and IKKε ubiquitination [16]. Consistent with these observations, our study showed that CYLD deficiency significantly enhanced TAK1 and RIG-I ubiquitination in IAV-infected cells (Fig. 9), leading to increased innate immunity. CYLD is cleaved at aspartate 215 by caspase-8, which generates an N-terminal 25-kDa fragment [17, 43]. The C-terminal fragment, which contains the catalytic domain, is degraded [14, 15]. We found that inhibition of CYLD cleavage by caspase-8 inhibitors or by gene deficiency decreased RIG-I and TAK1 ubiquitination. Since RIPK1, TBK1, NEMO, TRAF3, and TRAF6 can also be targeted by CYLD [14], it is likely that the removal of K63-linked ubiquitin in these molecules also contributes to its inhibitory effect on innate antiviral immunity.
Caspase-8 can also indirectly suppress innate immunity by activating caspase-3 [11]. Caspase-3 cleaves numerous proteins, such as cGAS, MAVS, IRF3 and the NF-κB members p65/RelA, RelB, and c-Rel [13, 44]. Caspase-3 deficiency increases TBK1 and IRF3 phosphorylation and the production of IFNs and ISG54 in peritoneal macrophages activated by the RIG-I or cGAS pathway or infected with viruses, including VSV, VACV, HSV-1, and SeV [13]. We found that caspase-3 inhibitors or caspase-3 deficiency did not decrease the total protein levels of IRF3 and p65 and had no effect on TBK1, IRF3, or p65 phosphorylation in IAV-infected A549 and NL20 cells (Fig. 6). These observations suggest that caspase-3 does not suppress antiviral responses in these two cell lines.
Programmed cell death is an integral part of the host’s defence against invading intracellular microbes [30, 45]. Caspase-8-mediated pyroptosis by GSDMD cleavage and activation promotes antimicrobial defence against Yersinia infection [46,47,48]. IAV induces multiple forms of cell death, including apoptosis, necroptosis, and pyroptosis [28]. Caspase-8 plays a crucial role in IAV-induced apoptosis and pyroptosis but inhibits necroptosis [28, 29], probably in part due to CYLD inactivation [17]. Furthermore, caspase-8 activation can enhance IAV-induced IL-1b production and inflammatory cell death in macrophages [49]. Our present study showed that caspase inhibition decreased innate antiviral immunity in two human airway epithelial cell lines and in primary alveolar epithelial cells. These observations suggest that caspase-8 activation may exert its antiviral effects by enhancing antiviral responses and inducing cell death and inflammatory responses to alert the immune system [50]. Since caspase inhibition enhanced innate immunity in macrophages (Fig. S2B and S2C) and LET1 cells (Fig. S3E), inhibition of caspase-8 likely promoted antiviral responses in rodents. Indeed, mice deficient in caspase-8 generate higher levels of type I interferons than wild-type mice and are resistant to norovirus infection [34]. Chu et al. [51] recently reported that the administration of z-LEHD-fmk, a caspase-9-specific inhibitor that inhibits intrinsic apoptosis, can prolong the survival of mice infected with the Middle East respiratory syndrome coronavirus. Since caspase-8 deficiency has opposite effects on the innate immune response in IAV-infected LET1 and NL20 cells and because RIPK1 is required for IAV-induced TBK1-IRF3 activation in murine LET1 cells but not in human NL20 cells (Fig. S8C), great caution should be taken when translating observations made in rodents into a human setting.
In summary, our study provides evidence that caspase-8 can enhance innate antiviral immunity against IAV infection in two cell lines. Mechanistically, caspase-8 cleaves and inactivates CYLD, leading to increased RIG-I and TAK1 ubiquitination and antiviral responses (Fig. 9E). Caspase-3, on the other hand, does not appear to play a significant role in regulating the innate antiviral response. Our study suggested that the role of caspase-8 in antiviral immunity is complex; whether RIPK1 is required for RIG-I-mediated TBK1 activation may determine whether caspase-8 enhances or suppresses antiviral responses.
Materials and methods
Reagents
Z-VAD (OMe)-FMK (HY-16658), Z-IETD (HY-101297), and Z-DEVD (HY-12466) were purchased from MedChemExpress (MCE) LLC (Shanghai, China). 4,6-Diamidino-2-phenylindole (DAPI) was purchased from InvivoGen, Inc. (Shanghai, China). Antibodies against β-Actin (#sc-47778), NS1 (#sc-130568), Sp1 (#sc-420), and GAPDH (#sc-166574) were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). Antibodies against cleaved caspase-8 (#9496), cleaved caspase-8 (#4790), cleaved caspase-3 (#9664), cleaved caspase-3 (#14220), CYLD (#8462), phospho-p65 (#3033), p65 (#3308), phospho-IκBα (#9341 s), IκBα (#9242), phospho-TBK1S172 (#5483), TBK1 (#3504), phospho-IRF3S396 (#29047), IRF3 (#4302), RIG-I (#3743), RIPK1 (#sc3493), phospho-TAK1S187 (#4536), TAK1 (#4505), JAK1 (#50996), and HA tag (#2367) were purchased from Cell Signaling Technology, Inc. (Danvers, MA, USA). Antibodies against Flag tag (DYKDDDDK) (#HT201-01) and GFP (#HT801-01) were purchased from TransGen Biotech Co., Ltd. (TransGen, Shanghai, China). An antibody against phospho-JAK1 (#abs138005) was purchased from Absin Bioscience, Inc. (Absin, China). The anti-PB2 antibody was purchased from GeneTex (#GTX125926) (Shanghai, China). Alexa Fluor® 488-conjugated anti-rabbit IgG (#AB_2338840) was purchased from Jackson ImmunoResearch Laboratories, Inc. (West Grove, PA, USA). The expression vector encoding FLAG-RIG-I was a generous gift from Dr Jianzhong Zhu (College of Veterinary Medicine, Yangzhou University, China). The expression vector encoding FLAG-TAK1 was a generous gift from Dr. Xin Lin (Tsinghua University, Beijing, China). The expression vector encoding HA-tagged Lys63-specific ubiquitin (HA-Ub-K63O) was kindly provided by Dr. Zhenhai Chen (College of Veterinary Medicine, Yangzhou University, China). The HA-ubiquitin gene was cloned and inserted into the pcDNA3.1 expression vector, as reported in our previous publication [52].
Cell cultures and viruses
A549 (a human lung cancer cell line of alveolar epithelial cell origin), NL20 (an immortalized, noncancerous human bronchial epithelial cell line), Madin-Darby canine kidney (MDCK), RAW264.7, and J774A.1 cells were purchased from the American Tissue Culture Collection (Manassas, VA). LET1 cells, a murine lung epithelial type I cell line, were kindly provided by BEI Resources (Manassas, VA). A549, LET1, RAW264.7, and J774A.1 cells were grown in DMEM supplemented with 10% fetal bovine serum (FBS). NL20 cells were grown in F12 media supplemented with 4% FBS, hydrocortisone (500 ng/ml), transferrin (1 mg/ml), insulin (5 mg/ml), EGF (epidermal growth factor, 10 ng/ml), and nonessential amino acid (0.1 mM). All cell lines tested negative for mycoplasma contamination according to PCR. Primary human alveolar epithelial cells were prepared according to previous publications [53]. Use of lung tissues was approved by the Medical Experiment Ethics Committee of College of Medicine, Yangzhou University Institutional with the informed consent by patients. Lung tissue adjascent to adenocarcinomas from lung cancer patients were obtained from the Affiliated Hospital of Yangzhou University. The tissues were sliced into approximately 0.5 mm3 cubicles, rinsed three times in HEPES-buffered balanced salt solution (HBSS) and then digested in HBSS containing trypsin (10,000 U/mL), elastase (5.0 U/mL) and DNase I (10,000 U/mL) for 45 min at 37 °C. Floating epithelial cells were obtained by three rounds of adherence to tissue culture dishes to remove macrophages. The cells were then cultured in DMEM/F12 medium supplemented with hydrocortisone (500 ng/ml), transferrin (1 mg/ml), insulin (5 mg/ml), EGF (epidermal growth factor, 10 ng/ml), and nonessential amino acid (0.1 mM). A/mallard/Huadong/S/2005 (SY strain, H5N1), A/chicken/Jiangsu/K0402/2010 (CK10 strain, H5N1), A/goose/Guangdong/Y6/2015 (Y6 strain, H5N6), and A/PR8/34 (H1N1) viruses have been reported previously [54]. These viruses were prepared by inoculating 10-day-old specific-pathogen-free embryonic chicken eggs. Viral titers were determined by a 10-fold serial dilution (101 to 109) of each dilution from 105 to 109 in MDCK cells. The Reed and Muench method was used to determine the 50% tissue culture infection dose (TCID50 per 100 ml).
Gene knockout
Caspase-8, caspase-3, CYLD, and RIPK1 were knocked out in the A549, NL20, and LET1 cell lines with the LentiCRISPR V2 (pLenti-V2) vector encoding a guide RNA. The targeting sequences were as follows: Human CASP8, 5’-TGATCGACCCTCCGCCAGAA-3’; Murine CASP8, 5’-GCAGGTCCCACCGACTGATG-3’ and 5’-CTTCCTAGACTGCAACCGAG-3’; Human CASP3, 5’-TAGTTAATAAAGGTATCCA-3’; Human CYLD, 5’-AAGCTCCTTAAAGTACCGAA-3’; Human RIPK1, 5’-AGTGCAGAACTGGACAGCGG-3’ and 5’-GAATACTGCGTCGGGCACGG-3’; and Murine RIPK1, 5’-AGACAGCGGAGGCTTCGGGA-3’. Three cell lines (A549, NL20, and LET1) were transfected with the CRISPR expression vector and selected in media supplemented with puromycin (1–2 µg/ml) for 14 days. Individual colonies were expanded and analyzed for the expression of caspase-8, caspase-3, CYLD, and RIPK1 by Western blotting. The colonies screened from the cells transfected with the pLenti-V2 empty vector were used as controls. All experiments were carried out with at least two colonies.
Recombinant DNA
Total cellular mRNA was extracted from 293T cells and reverse-transcribed by using a reverse transcription kit. cDNA was used as the template to amplify the full-length CYLD gene, which was subsequently cloned and inserted into the Kpn I/Nhe I-digested pCAGGS vector with a Seamless Cloning and Assembly Kit (TransGen, Beijing, China). The sequences of the primers used for PCR amplification were 5’-CATCGATGCATGGTACATGAGTTCAGGCTTATGG-3’ and 5’-GGCAGAGGGAAAAAGATC-TGTTATTTGTACAAACTC-3’. Mutation of the CYLD D215A site was performed with a Fast Mutagenesis System (TransGen, Beijing, China) with the following primers: 5’-CACTGCATTGGAAAGTGCTTACGCAGG-3’ and 5’-GCACTTTCCAATGCAGTGTCATCATCTTC-3’. The expression vector pCAGGS-CYLD and its mutant pCAGGS-CYLDD215A were sequenced to confirm the mutation.
Real-time quantitative PCR analysis
Total RNA was extracted from A549 and NL20 cells using TRIzol (Sangon, China). RNA integrity was verified by electrophoresis. Reverse transcription of RNA was performed using Hiscript III RT SuperMix for qPCR (Vazyme, China) according to the manufacturer’s instructions. cDNA was subjected to quantitative real-time PCR using 2x ChamQ Universal SYBR qPCR Master Mix (Vazyme, China). The sequences of the primers used for IFN-β were as follows: 5’-CTATTGTTGAGAACCTCCTGGCTA-3’, 5’-CCACTCTGACTATGGTCCAGGCAC-3’; ISG15: 5’-GGACAAATGCGACGAACCTC-3’, 5’-GCTCACTTGCTGCTT-CAGG-3’; MX1: 5’-TGCA-GGACAAGGACACCTAC-3’, 5’-TCACCACGGCTAACGGATAA-3’; and IL-6: 5’-AGACAGCCACTCACCTCTTCAG-3’, 5’-TTCTGCCAGTGCCTCTTTGCTG-3’. The amplification of human β-actin was included as a control. Relative mRNA levels were normalized to the ΔΔCT by the β-actin mRNA level. Fold changes were calculated as the ratio of the normalized value of each sample to that of the corresponding untreated control sample. All real-time RT‒PCRs were performed in triplicate. The results from three independent experiments were pooled and statistically analyzed.
Western blot
Cells grown in 12-well plates were harvested and lysed in NP-40 lysis buffer (50 mM Tris-HCl, pH 8.0; 150 mM NaCl; 1% NP-40; 5 mM EDTA, 10 mg/ml aprotinin, 10 mg/ml leupeptin, and 1 mM phenylmethylsulfonyl fluoride). After ultrasonication, cell lysates were prepared and analyzed for the proteins of interest by Western blotting with specific antibodies, followed by incubation with horseradish peroxidase-conjugated goat anti-rabbit or anti-mouse IgG and SuperSignal Western Pico enhanced chemiluminescence substrate (Pierce Chemical Co., Rockford, IL). All Western blot experiments were repeated at least twice, each with the detection of b-actin as a loading control. Protein phosphorylation was first analyzed by probing with an antibody against phosphorylated protein. The membrane was stripped and reprobed with an antibody against total protein. Sliced blots from the same membrane were used for detecting full-length and cleaved proteins such as CYLD, caspase-8 and caspase-3. The relative phosphorylation levels were analyzed by quantifying the density of the phosphorylated protein bands normalized to their corresponding total proteins. The results are presented as bar graphs.
Luciferase reporter assay
A549 cells seeded in a 96-well plate were transfected with a luciferase reporter gene driven by multiple NF-κB or IRF3 binding sites or by the IFN-b gene promoter by using TurboFect (Thermo Fisher Scientific, NY, USA) following the manufacturer’s instructions. The b-actin promoter-driven Renilla luciferase reporter plasmid was cotransfected as an internal control. Twenty-four hours after transfection, the cells were infected with 1 MOI (multiplicity of infection) of the H5N1 virus and incubated for another 24 h in the absence or presence of Z-VAD (100 µM). Cells were harvested and analyzed for luciferase activity by using a double-luciferase reporter assay kit (TransDetect, Beijing, China) and read in a Tecan plate reader (Phenix Research Products, Phenix, AZ). The relative light intensity of each sample was normalized to that of the b-actin promoter-driven Renilla luciferase control. The data are presented as the means ± standard deviations (SD) of triplicate data from one experiment. The experiments were repeated at least twice with similar results. Experiments in caspase-8-, caspase-3-, and CYLD-deficient A549 cells were similarly carried out. Wild-type A549 cells transfected with the pLenti-V2 vector were used as controls.
ELISA
pLenti-V2-transfected wild-type (WT) control and caspase-8-, caspase-3-, or CYLD-deficient A549 cells were infected with H5N1 virus (SY) at an MOI of 1 for 24 h. Conditioned media were collected and analyzed for the concentration of IFN-b by using an ELISA kit (Elabscience, Wuhan, China). The results are presented as the means ± SDs of three independent experiments.
Immunofluorescence staining
A549 cells seeded on coverslips in 24-well flat-bottomed plates were infected with the H5N1 virus and then incubated in the absence or presence of Z-VAD (100 µM). Dimethyl sulfoxide (DMSO), used as a solvent for Z-VAD, was added as a negative control at a concentration of 2%. The cells were fixed with cold 100% methanol and permeabilized with 0.1% Triton X-100 in PBS for 20 min, followed by rinsing three times with PBS. The coverslips were blocked at room temperature with 5% bovine serum albumin (BSA) in PBS for 1 h and then probed with an anti-IRF3 or p65 antibody in a blocking buffer overnight at 4 °C. The cells were then stained with Alexa 488-conjugated anti-rabbit IgG (1:100) for 1 h at room temperature. Finally, the cells were stained with 10 µM DAPI for 5 min. Fluorescence images were captured under a Leica SP8 confocal laser scanning microscope. The percentage of cells with IRF3- or p65-positive nuclei in eight randomly selected fields, each with a range of 20–45 cells per field, was calculated. Experiments with caspase-8 and CYLD-deficient A549 cells were carried out similarly. Wild-type A549 cells transfected with the pLenti-V2 vector were used as controls.
Immunoprecipitation
Cells were lysed in 1% Triton X-100 lysis buffer (20 mM Tris-HCl, pH 7.5; 100 mM NaCl; 1% Triton X-100; 10% glycerol; 10 mM EDTA; and a protease and phosphatase inhibitor cocktail (P002, NCM Biotech)) on ice, followed by sonication. After centrifugation at 12,000 ×g for 15 min, the supernatant was incubated with rabbit anti-FLAG IgG overnight, followed by the addition of protein A/G agarose beads (#53132, Thermo Scientific) and incubation at 4 °C for 3 h on a rotator. The immune complexes were rinsed in lysis buffer three times and then boiled in 2× loading buffer for 5 min. RIG-I and TAK1 ubiquitination was detected by Western blotting with an anti-HA mAb. RIG-I and TAK1 were detected with an antibody against FLAG.
Statistical analysis
Differences in mRNA levels, luciferase activity, virus titers, and IRF3 and p65 nuclear staining were statistically analyzed by using an unpaired Student’s t-test. A P-value < 0.05 was considered to indicate statistical significance. All the statistical analyses were performed with GraphPad Prism 8 software (San Diego, CA).
Data availability
The authors declare that the data supporting the findings of this study are available within the article and from the corresponding author upon request.
Abbreviations
- BMDM:
-
Bone marrow-derived monocytes
- BSA:
-
Bovine serum albumin
- CASP:
-
Caspase
- cGAS:
-
Cyclic GMP-AMP synthase
- CYLD:
-
Cylindromatosis
- DAPI:
-
4,6-diamidino-2-phenylindole
- DMSO:
-
Dimethyl sulfoxide
- dsRNA:
-
Double-stranded RNA
- FADD:
-
FAS-associated protein with death domain
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- GFP:
-
Green fluorescent protein
- GSDMD:
-
Gasdermin D
- HA:
-
Hemagglutinin
- HMGB1:
-
High mobility group box 1
- HSV1:
-
Herpes simplex virus type 1
- IAV:
-
Influenza A virus
- IF:
-
Immunofluorescence
- IKKε:
-
IkappaB kinase ε
- IFN:
-
Interferon
- IRF3:
-
IFN regulatory factor 3
- IP:
-
Immunoprecipitation
- MEF:
-
Murine embryonic fibroblast
- MNV1:
-
Murine norovirus-1
- MOI:
-
Multiplicity of infection
- NA:
-
Neuraminidase
- RT‒PCR:
-
Reverse-transcription-PCR
- RIG-I:
-
Retinoic acid-inducible gene-I
- RIPK:
-
Receptor interacting protein kinase
- SD:
-
Standard deviation
- SeV:
-
Sendai virus
- TAK1:
-
Transforming growth factor-β activating protein kinase 1
- TBK1:
-
TANK-binding kinase 1
- TLR3:
-
Toll-like receptor 3
- TNF:
-
Tumor necrosis factor
- TRAF3:
-
TNF receptor-associated factor 3
- TRADD:
-
Tumor necrosis factor receptor type 1-associated death domain protein
- ZBP1:
-
Z-DNA binding protein 1
- WCL:
-
Whole cell lysate
- WT:
-
Wild-type
References
Li D, Wu M (2021) Pattern recognition receptors in health and diseases. Signal Transduct Target Ther 6:291
Wu W, Zhang W, Duggan ES, Booth JL, Zou MH, Metcalf JP (2015) RIG-I and TLR3 are both required for maximum interferon induction by influenza virus in human lung alveolar epithelial cells. Virology 482:181–188
Onomoto K, Onoguchi K, Yoneyama M (2021) Regulation of RIG-I-like receptor-mediated signaling: interaction between host and viral factors. Cell Mol Immunol 18:539–555
Rehwinkel J, Gack MU (2020) RIG-I-like receptors: their regulation and roles in RNA sensing. Nat Rev Immunol 20:537–551
Madiraju C, Novack JP, Reed JC, Matsuzawa SI (2022) K63 ubiquitination in immune signaling. Trends Immunol 43:148–162
Antonia RJ, Hagan RS, Baldwin AS (2021) Expanding the View of IKK: New substrates and New Biology. Trends Cell Biol 31:166–178
Zhang Q, Zhang X, Lei X, Wang H, Jiang J, Wang Y, Bi K, Diao H (2022) Influenza A virus NS1 protein hijacks YAP/TAZ to suppress TLR3-mediated innate immune response. PLoS Pathog 18:e1010505
Fang R, Jiang Q, Zhou X, Wang C, Guan Y, Tao J, Xi J, Feng JM, Jiang Z (2017) MAVS activates TBK1 and IKKε through TRAFs in NEMO dependent and independent manner. PLoS Pathog 13:e1006720
Udawatte DJ, Rothman AL (2021) Viral Suppression of RIPK1-Mediated Signaling. mBio 12, e0172321
Wang Y, Karki R, Mall R, Sharma BR, Kalathur RC, Lee S, Kancharana B, So M, Combs KL, Kanneganti T-D (2022) Molecular mechanism of RIPK1 and caspase-8 in homeostatic type I interferon production and regulation. Cell Rep 41
Chen H, Ning X, Jiang Z (2017) Caspases control antiviral innate immunity. Cell Mol Immunol 14:736–747
Rajput A, Kovalenko A, Bogdanov K, Yang SH, Kang TB, Kim JC, Du J, Wallach D (2011) RIG-I RNA helicase activation of IRF3 transcription factor is negatively regulated by caspase-8-mediated cleavage of the RIP1 protein. Immunity 34:340–351
Ning X, Wang Y, Jing M, Sha M, Lv M, Gao P, Zhang R, Huang X, Feng JM, Jiang Z (2019) Apoptotic caspases suppress type I Interferon Production via the cleavage of cGAS, MAVS, and IRF3. Mol Cell 74:19–31e17
Lork M, Verhelst K, Beyaert R (2017) CYLD, A20 and OTULIN deubiquitinases in NF-kappaB signaling and cell death: so similar, yet so different. Cell Death Differ 24:1172–1183
Sun SC (2010) CYLD: a tumor suppressor deubiquitinase regulating NF-kappaB activation and diverse biological processes. Cell Death Differ 17:25–34
Friedman CS, O’Donnell MA, Legarda-Addison D, Ng A, Cárdenas WB, Yount JS, Moran TM, Basler CF, Komuro A, Horvath CM, Xavier R, Ting AT (2008) The tumour suppressor CYLD is a negative regulator of RIG-I-mediated antiviral response. EMBO Rep 9:930–936
O’Donnell MA, Perez-Jimenez E, Oberst A, Ng A, Massoumi R, Xavier R, Green DR, Ting AT (2011) Caspase 8 inhibits programmed necrosis by processing CYLD. Nat Cell Biol 13:1437–1442
Takahashi K, Kawai T, Kumar H, Sato S, Yonehara S, Akira S (2006) Roles of caspase-8 and caspase-10 in innate immune responses to double-stranded RNA. J Immunol 176:4520–4524
Horn S, Hughes MA, Schilling R, Sticht C, Tenev T, Ploesser M, Meier P, Sprick MR, MacFarlane M, Leverkus M (2017) Caspase-10 negatively regulates caspase-8-Mediated cell death, switching the response to CD95L in Favor of NF-kappaB activation and cell survival. Cell Rep 19:785–797
Lemmers B, Salmena L, Bidere N, Su H, Matysiak-Zablocki E, Murakami K, Ohashi PS, Jurisicova A, Lenardo M, Hakem R, Hakem A (2007) Essential role for caspase-8 in toll-like receptors and NFkappaB signaling. J Biol Chem 282:7416–7423
Weng D, Marty-Roix R, Ganesan S, Proulx MK, Vladimer GI, Kaiser WJ, Mocarski ES, Pouliot K, Chan FK, Kelliher MA, Harris PA, Bertin J, Gough PJ, Shayakhmetov DM, Goguen JD, Fitzgerald KA, Silverman N, Lien E (2014) Caspase-8 and RIP kinases regulate bacteria-induced innate immune responses and cell death. Proc. Natl. Acad. Sci. U. S. A 111, 7391–7396
Chun HJ, Zheng L, Ahmad M, Wang J, Speirs CK, Siegel RM, Dale JK, Puck J, Davis J, Hall CG, Skoda-Smith S, Atkinson TP, Straus SE, Lenardo MJ (2002) Pleiotropic defects in lymphocyte activation caused by caspase-8 mutations lead to human immunodeficiency. Nature 419:395–399
Orning P, Lien E (2021) Multiple roles of caspase-8 in cell death, inflammation, and innate immunity. J Leukoc Biol 109:121–141
Paules C, Subbarao K (2017) Influenza Lancet 390:697–708
Lai S, Qin Y, Cowling BJ, Ren X, Wardrop NA, Gilbert M, Tsang TK, Wu P, Feng L, Jiang H, Peng Z, Zheng J, Liao Q, Li S, Horby PW, Farrar JJ, Gao GF, Tatem AJ, Yu H (2016) Global epidemiology of avian influenza a H5N1 virus infection in humans, 1997–2015: a systematic review of individual case data. Lancet Infect Dis 16:e108–e118
Neumann G, Chen H, Gao GF, Shu Y, Kawaoka Y (2010) H5N1 influenza viruses: outbreaks and biological properties. Cell Res 20:51–61
Meischel T, Villalon-Letelier F, Saunders PM, Reading PC, Londrigan SL (2020) Influenza A virus interactions with macrophages: lessons from epithelial cells. Cell Microbiol 22:e13170
Balachandran S, Mocarski ES (2021) Viral Z-RNA triggers ZBP1-dependent cell death. Curr Opin Virol 51:134–140
Balachandran S, Rall GF (2020) Benefits and perils of Necroptosis in Influenza Virus infection. J Virol 94
Atkin-Smith GK, Duan M, Chen W, Poon IKH (2018) The induction and consequences of Influenza A virus-induced cell death. Cell Death Dis 9:1002
Kesavardhana S, Malireddi RKS, Kanneganti TD (2020) Caspases in cell death, inflammation, and Pyroptosis. Annu Rev Immunol 38:567–595
Thornberry NA, Rano TA, Peterson EP, Rasper DM, Timkey T, Garcia-Calvo M, Houtzager VM, Nordstrom PA, Roy S, Vaillancourt JP, Chapman KT, Nicholson DW (1997) A combinatorial approach defines specificities of members of the caspase family and granzyme B. Functional relationships established for key mediators of apoptosis. J Biol Chem 272:17907–17911
Lork M, Verhelst K, Beyaert R (2017) CYLD, A20 and OTULIN deubiquitinases in NF-κB signaling and cell death: so similar, yet so different. Cell Death Differ 24:1172–1183
Wang Y, Karki R, Mall R, Sharma BR, Kalathur RC, Lee S, Kancharana B, So M, Combs KL, Kanneganti TD (2022) Molecular mechanism of RIPK1 and caspase-8 in homeostatic type I interferon production and regulation. Cell Rep 41:111434
Fang Y, Peng K (2022) Regulation of innate immune responses by cell death-associated caspases during virus infection. FEBS J 289:4098–4111
Brodsky IE (2020) Caspase-8 protein cuts a brake on immune defences. Nature 587:201–203
Pang J, Vince JE (2023) The role of caspase-8 in inflammatory signalling and pyroptotic cell death. Semin Immunol 70:101832
Niemela J, Kuehn HS, Kelly C, Zhang M, Davies J, Melendez J, Dreiling J, Kleiner D, Calvo K, Oliveira JB, Rosenzweig SD (2015) Caspase-8 Deficiency presenting as late-onset multi-organ lymphocytic infiltration with Granulomas in two adult siblings. J Clin Immunol 35:348–355
Lehle AS, Farin HF, Marquardt B, Michels BE, Magg T, Li Y, Liu Y, Ghalandary M, Lammens K, Hollizeck S, Rohlfs M, Hauck F, Conca R, Walz C, Weiss B, Lev A, Simon AJ, Gross O, Gaidt MM, Hornung V, Clevers H, Yazbeck N, Hanna-Wakim R, Shouval DS, Warner N, Somech R, Muise AM, Snapper SB, Bufler P, Koletzko S, Klein C, Kotlarz D (2019) Intestinal inflammation and dysregulated immunity in patients with inherited Caspase-8 Deficiency. Gastroenterology 156:275–278
Su H, Bidere N, Zheng L, Cubre A, Sakai K, Dale J, Salmena L, Hakem R, Straus S, Lenardo M (2005) Requirement for caspase-8 in NF-kappaB activation by antigen receptor. Science 307:1465–1468
Sears N, Sen GC, Stark GR, Chattopadhyay S (2011) Caspase-8-mediated cleavage inhibits IRF-3 protein by facilitating its proteasome-mediated degradation. J Biol Chem 286:33037–33044
Manganaro L, Pache L, Herrmann T, Marlett J, Hwang Y, Murry J, Miorin L, Ting AT, Konig R, Garcia-Sastre A, Bushman FD, Chanda SK, Young JA, Fernandez-Sesma A, Simon V (2014) Tumor suppressor cylindromatosis (CYLD) controls HIV transcription in an NF-kappaB-dependent manner. J Virol 88:7528–7540
Legarda D, Justus SJ, Ang RL, Rikhi N, Li W, Moran TM, Zhang J, Mizoguchi E, Zelic M, Kelliher MA, Blander JM, Ting AT (2016) CYLD Proteolysis protects macrophages from TNF-Mediated auto-necroptosis Induced by LPS and licensed by type I IFN. Cell Rep 15:2449–2461
Wu D, Wang Z, Zhang J, Robinson AG, Lyu B, Chen Z, Wang C, Wei B, Xia X, Zhang Q, Zhou X (2022) Apoptotic caspase inhibits innate immune signaling by cleaving NF-kappaBs in both mammals and flies. Cell Death Dis 13:731
Verburg SG, Lelievre RM, Westerveld MJ, Inkol JM, Sun YL, Workenhe ST (2022) Viral-mediated activation and inhibition of programmed cell death. PLoS Pathog 18:e1010718
Demarco B, Grayczyk JP, Bjanes E, Le Roy D, Tonnus W, Assenmacher CA, Radaelli E, Fettrelet T, Mack V, Linkermann A, Roger T, Brodsky IE, Chen KW, Broz P (2020) Caspase-8-dependent gasdermin D cleavage promotes antimicrobial defense but confers susceptibility to TNF-induced lethality. Sci Adv 6
Orning P, Weng D, Starheim K, Ratner D, Best Z, Lee B, Brooks A, Xia S, Wu H, Kelliher MA, Berger SB, Gough PJ, Bertin J, Proulx MM, Goguen JD, Kayagaki N, Fitzgerald KA, Lien E (2018) Pathogen blockade of TAK1 triggers caspase-8-dependent cleavage of gasdermin D and cell death. Science 362:1064–1069
Sarhan J, Liu BC, Muendlein HI, Li P, Nilson R, Tang AY, Rongvaux A, Bunnell SC, Shao F, Green DR, Poltorak A (2018) Caspase-8 induces cleavage of gasdermin D to elicit pyroptosis during Yersinia infection. Proc. Natl. Acad. Sci. U. S. A 115, E10888-E10897
Lei X, Chen Y, Lien E, Fitzgerald KA (2023) MLKL-Driven Inflammasome Activation and Caspase-8 Mediate Inflammatory Cell Death in Influenza A Virus Infection. mBio 14, e0011023
Verdonck S, Nemegeer J, Vandenabeele P, Maelfait J (2022) Viral manipulation of host cell necroptosis and pyroptosis. Trends Microbiol 30:593–605
Chu H, Shuai H, Hou Y, Zhang X, Wen L, Huang X, Hu B, Yang D, Wang Y, Yoon C, Wong BH, Li C, Zhao X, Poon VK, Cai JP, Wong KK, Yeung ML, Zhou J, Au-Yeung RK, Yuan S, Jin DY, Kok KH, Perlman S, Chan JF, Yuen KY (2021) Targeting highly pathogenic coronavirus-induced apoptosis reduces viral pathogenesis and disease severity. Sci Adv 7
Ruan T, Sun Y, Zhang J, Sun J, Liu W, Prinz RA, Peng D, Liu X, Xu X (2022) H5N1 infection impairs the alveolar epithelial barrier through intercellular junction proteins via itch-mediated proteasomal degradation. Commun Biology 5:186
Mao P, Wu S, Li J, Fu W, He W, Liu X, Slutsky AS, Zhang H, Li Y (2015) Human alveolar epithelial type II cells in primary culture. Physiol Rep 3
Ruan T, Sun J, Liu W, Prinz RA, Peng D, Liu X, Xu X (2020) H1N1 influenza virus cross-activates Gli1 to disrupt the intercellular junctions of alveolar epithelial cells. Cell Rep 31:107801
Acknowledgements
We thank Dr. Feng Ma for kindly providing GFP-tagged Sendai virus, Dr. Xin Lin (Tsinghua University) for kindly providing the TAK1 expression vector, Dr. Zhenhai Chen for kindly providing the HA-tagged K63 ubiquitin expression vector, Dr. Jianzhong Zhu (Yangzhou University, Jiangsu Province, China) for kindly providing the FLAG-RIG-I expression vector, Dr. Liqian Zhu (College of Veterinary Medicine, Yangzhou University) for kindly providing the H1N1 (PR8) virus, and BEI Resources for providing the LET1 cell line.
Funding
This work was supported in part by the National Key Research and Development Project of China (2021YFD1800202) (X. Wang), the Priority Academic Program Development of Jiangsu Higher Education Institutions (X. Xu), and the State Key Laboratory for Animal Disease Control and Prevention, Lanzhou Veterinary Research Institute, Chinese Academy of Agricultural Sciences (SKLVEB-KFKT-02) (P. Liu).
Author information
Authors and Affiliations
Contributions
XX and HY conceptualization, HY, YS, JZ, WZ, HL, PZ investigation, WL, P. L, KL, JS supervision and coordination, XW, XL, PL. critical resources and fund acquisition, HY, manuscript drafting. XX. wrote and edited the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Human lung tissues were obtained the the Affiliated Hospital of Yangzhou University. The study was approved by the Medical Experiment Ethics Committee of College of Medicine, Yangzhou University with the informed consent by patients.
Consent for publication
All authors have read the manuscript and agree with submission for publication.
Conflict of interest
The authors declare that the study was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yu, H., Sun, Y., Zhang, J. et al. Influenza A virus infection activates caspase-8 to enhance innate antiviral immunity by cleaving CYLD and blocking TAK1 and RIG-I deubiquitination. Cell. Mol. Life Sci. 81, 355 (2024). https://doi.org/10.1007/s00018-024-05392-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-024-05392-z