Abstract
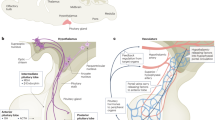
The pituitary plays a pivotal role in maintaining systemic homeostasis by secreting several hormones. During fetal development, the pituitary develops from the oral ectoderm in contact with the adjacent hypothalamus. This process is regulated by the fine-tuned expression of transcription and growth factors. Impairments of this process result in congenital pituitary hypoplasia leading to dysfunction of the pituitary. Although animal models such as knockout mice have helped to clarify these underlying mechanisms, the developmental processes of the human pituitary gland and the mechanisms of human pituitary disorders have not been fully understood. This is because, at least in part, of the lack of a human pituitary developmental model. Recently, methods for in vitro induction of the pituitary gland from human pluripotent stem cells were developed. These models can be utilized not only for regenerative medicine but also for human pituitary studies on developmental biology and for modeling of pituitary disorders, such as hypopituitarism and pituitary tumors. In this review, we provide an overview of recent progress in the applications of pluripotent stem cells for pituitary research and discuss further perspectives for pituitary studies.



Similar content being viewed by others
References
Nakaguma M, Correa FA, Santana LS, Benedetti AFF, Perez RV, Huayllas MKP, Miras MB, Funari MFA, Lerario AM, Mendonca BB, Carvalho LRS, Jorge AAL, Arnhold IJP (2019) Genetic diagnosis of congenital hypopituitarism by a target gene panel: novel pathogenic variants in GLI2, OTX2 and GHRHR. Endocr Connect 8(5):590–595. https://doi.org/10.1530/EC-19-0085
Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM (1998) Embryonic stem cell lines derived from human blastocysts. Science 282(5391):1145–1147. https://doi.org/10.1126/science.282.5391.1145
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131(5):861–872. https://doi.org/10.1016/j.cell.2007.11.019
Avior Y, Sagi I, Benvenisty N (2016) Pluripotent stem cells in disease modelling and drug discovery. Nat Rev Mol Cell Biol 17(3):170–182. https://doi.org/10.1038/nrm.2015.27
Dincer Z, Piao J, Niu L, Ganat Y, Kriks S, Zimmer B, Shi SH, Tabar V, Studer L (2013) Specification of functional cranial placode derivatives from human pluripotent stem cells. Cell Rep 5(5):1387–1402. https://doi.org/10.1016/j.celrep.2013.10.048
Ozone C, Suga H, Eiraku M, Kadoshima T, Yonemura S, Takata N, Oiso Y, Tsuji T, Sasai Y (2016) Functional anterior pituitary generated in self-organizing culture of human embryonic stem cells. Nat Commun 7:10351. https://doi.org/10.1038/ncomms10351
Ogawa K, Suga H, Ozone C, Sakakibara M, Yamada T, Kano M, Mitsumoto K, Kasai T, Kodani Y, Nagasaki H, Yamamoto N, Hagiwara D, Goto M, Banno R, Sugimura Y, Arima H (2018) Vasopressin-secreting neurons derived from human embryonic stem cells through specific induction of dorsal hypothalamic progenitors. Sci Rep 8(1):3615. https://doi.org/10.1038/s41598-018-22053-x
Couly G, Le Douarin NM (1988) The fate map of the cephalic neural primordium at the presomitic to the 3-somite stage in the avian embryo. Development 103(Suppl):101–113
Glasgow E, Karavanov AA, Dawid IB (1997) Neuronal and neuroendocrine expression of lim3, a LIM class homeobox gene, is altered in mutant zebrafish with axial signaling defects. Dev Biol 192(2):405–419. https://doi.org/10.1006/dbio.1997.8761
Takuma N, Sheng HZ, Furuta Y, Ward JM, Sharma K, Hogan BL, Pfaff SL, Westphal H, Kimura S, Mahon KA (1998) Formation of Rathke’s pouch requires dual induction from the diencephalon. Development 125(23):4835–4840
Treier M, Gleiberman AS, O’Connell SM, Szeto DP, McMahon JA, McMahon AP, Rosenfeld MG (1998) Multistep signaling requirements for pituitary organogenesis in vivo. Genes Dev 12(11):1691–1704
Dasen JS, Rosenfeld MG (2001) Signaling and transcriptional mechanisms in pituitary development. Annu Rev Neurosci 24:327–355. https://doi.org/10.1146/annurev.neuro.24.1.327
Treier M, O’Connell S, Gleiberman A, Price J, Szeto DP, Burgess R, Chuang PT, McMahon AP, Rosenfeld MG (2001) Hedgehog signaling is required for pituitary gland development. Development 128(3):377–386
Hui CC, Slusarski D, Platt KA, Holmgren R, Joyner AL (1994) Expression of three mouse homologs of the Drosophila segment polarity gene cubitus interruptus, Gli, Gli-2, and Gli-3, in ectoderm- and mesoderm-derived tissues suggests multiple roles during postimplantation development. Dev Biol 162(2):402–413. https://doi.org/10.1006/dbio.1994.1097
Ericson J, Norlin S, Jessell TM, Edlund T (1998) Integrated FGF and BMP signaling controls the progression of progenitor cell differentiation and the emergence of pattern in the embryonic anterior pituitary. Development 125(6):1005–1015
Maruoka Y, Ohbayashi N, Hoshikawa M, Itoh N, Hogan BL, Furuta Y (1998) Comparison of the expression of three highly related genes, Fgf8, Fgf17 and Fgf18, in the mouse embryo. Mech Dev 74(1–2):175–177. https://doi.org/10.1016/s0925-4773(98)00061-6
Celli G, LaRochelle WJ, Mackem S, Sharp R, Merlino G (1998) Soluble dominant-negative receptor uncovers essential roles for fibroblast growth factors in multi-organ induction and patterning. EMBO J 17(6):1642–1655. https://doi.org/10.1093/emboj/17.6.1642
Hermesz E, Mackem S, Mahon KA (1996) Rpx: a novel anterior-restricted homeobox gene progressively activated in the prechordal plate, anterior neural plate and Rathke’s pouch of the mouse embryo. Development 122(1):41–52
Spieler D, Baumer N, Stebler J, Koprunner M, Reichman-Fried M, Teichmann U, Raz E, Kessel M, Wittler L (2004) Involvement of Pax6 and Otx2 in the forebrain-specific regulation of the vertebrate homeobox gene ANF/Hesx1. Dev Biol 269(2):567–579. https://doi.org/10.1016/j.ydbio.2004.01.044
Mortensen AH, Schade V, Lamonerie T, Camper SA (2015) Deletion of OTX2 in neural ectoderm delays anterior pituitary development. Hum Mol Genet 24(4):939–953. https://doi.org/10.1093/hmg/ddu506
Charles MA, Suh H, Hjalt TA, Drouin J, Camper SA, Gage PJ (2005) PITX genes are required for cell survival and Lhx3 activation. Mol Endocrinol 19(7):1893–1903. https://doi.org/10.1210/me.2005-0052
Szeto DP, Rodriguez-Esteban C, Ryan AK, O’Connell SM, Liu F, Kioussi C, Gleiberman AS, Izpisúa-Belmonte JC, Rosenfeld MG (1999) Role of the Bicoid-related homeodomain factor Pitx1 in specifying hindlimb morphogenesis and pituitary development. Genes Dev 13(4):484–494. https://doi.org/10.1101/gad.13.4.484
Suh H, Gage PJ, Drouin J, Camper SA (2002) Pitx2 is required at multiple stages of pituitary organogenesis: pituitary primordium formation and cell specification. Development 129(2):329–337
Sheng HZ, Zhadanov AB, Mosinger B Jr, Fujii T, Bertuzzi S, Grinberg A, Lee EJ, Huang SP, Mahon KA, Westphal H (1996) Specification of pituitary cell lineages by the LIM homeobox gene Lhx3. Science 272(5264):1004–1007. https://doi.org/10.1126/science.272.5264.1004
Kelberman D, Rizzoti K, Lovell-Badge R, Robinson ICAF, Dattani MT (2009) Genetic Regulation of Pituitary Gland Development in Human and Mouse. Endocr Rev 30(7):790–829. https://doi.org/10.1210/er.2009-0008
Sheng HZ, Moriyama K, Yamashita T, Li H, Potter SS, Mahon KA, Westphal H (1997) Multistep control of pituitary organogenesis. Science 278(5344):1809–1812. https://doi.org/10.1126/science.278.5344.1809
Davis SW, Keisler JL, Pérez-Millán MI, Schade V, Camper SA (2016) All hormone-producing cell types of the pituitary intermediate and anterior lobes derive from prop1-expressing progenitors. Endocrinology 157(4):1385–1396. https://doi.org/10.1210/en.2015-1862
Olson LE, Tollkuhn J, Scafoglio C, Krones A, Zhang J, Ohgi KA, Wu W, Taketo MM, Kemler R, Grosschedl R, Rose D, Li X, Rosenfeld MG (2006) Homeodomain-mediated beta-catenin-dependent switching events dictate cell-lineage determination. Cell 125(3):593–605. https://doi.org/10.1016/j.cell.2006.02.046
Zhao L, Bakke M, Parker KL (2001) Pituitary-specific knockout of steroidogenic factor 1. Mol Cell Endocrinol 185(1–2):27–32. https://doi.org/10.1016/s0303-7207(01)00621-9
Wataya T, Ando S, Muguruma K, Ikeda H, Watanabe K, Eiraku M, Kawada M, Takahashi J, Hashimoto N, Sasai Y (2008) Minimization of exogenous signals in ES cell culture induces rostral hypothalamic differentiation. Proc Natl Acad Sci USA 105(33):11796–11801. https://doi.org/10.1073/pnas.0803078105
Suga H, Kadoshima T, Minaguchi M, Ohgushi M, Soen M, Nakano T, Takata N, Wataya T, Muguruma K, Miyoshi H, Yonemura S, Oiso Y, Sasai Y (2011) Self-formation of functional adenohypophysis in three-dimensional culture. Nature 480(7375):57–62. https://doi.org/10.1038/nature10637
Chambers SM, Fasano CA, Papapetrou EP, Tomishima M, Sadelain M, Studer L (2009) Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat Biotechnol 27(3):275–280. https://doi.org/10.1038/nbt.1529
Zimmer B, Piao J, Ramnarine K, Tomishima MJ, Tabar V, Studer L (2016) Derivation of Diverse Hormone-Releasing Pituitary Cells from Human Pluripotent Stem Cells. Stem Cell Rep 6(6):858–872. https://doi.org/10.1016/j.stemcr.2016.05.005
Kasai T, Suga H, Sakakibara M, Ozone C, Matsumoto R, Kano M, Mitsumoto K, Ogawa K, Kodani Y, Nagasaki H, Inoshita N, Sugiyama M, Onoue T, Tsunekawa T, Ito Y, Takagi H, Hagiwara D, Iwama S, Goto M, Banno R, Takahashi J, Arima H (2020) Hypothalamic contribution to pituitary functions is recapitulated in vitro using 3d-cultured human iPS cells. Cell Rep 30(1):18–24. https://doi.org/10.1016/j.celrep.2019.12.009e15
Studer L, Tabar V (2016) Human Pluripotent-Derived Lineages for Repairing Hypopituitarism. In: Pfaff D, Christen Y (eds) Stem Cells in Neuroendocrinology. Springer, Cham, pp 25–34. https://doi.org/10.1007/978-3-319-41603-8_3
Kelava I, Lancaster Madeline A (2016) Stem cell models of human brain development. Cell Stem Cell 18(6):736–748. https://doi.org/10.1016/j.stem.2016.05.022
Protze SI, Lee JH, Keller GM (2019) Human pluripotent stem cell-derived cardiovascular cells: from developmental biology to therapeutic applications. Cell Stem Cell 25(3):311–327. https://doi.org/10.1016/j.stem.2019.07.010
Sterneckert JL, Reinhardt P, Scholer HR (2014) Investigating human disease using stem cell models. Nat Rev Genet 15(9):625–639. https://doi.org/10.1038/nrg3764
Ferran JL, Puelles L, Rubenstein JL (2015) Molecular codes defining rostrocaudal domains in the embryonic mouse hypothalamus. Front Neuroanat 9(46):1–14. https://doi.org/10.3389/fnana.2015.00046
Dateki S, Fukami M, Sato N, Muroya K, Adachi M, Ogata T (2008) OTX2 mutation in a patient with anophthalmia, short stature, and partial growth hormone deficiency: functional studies using the IRBP, HESX1, and POU1F1 promoters. J Clin Endocrinol Metab 93(10):3697–3702. https://doi.org/10.1210/jc.2008-0720
Iwama S, Sugimura Y, Kiyota A, Kato T, Enomoto A, Suzuki H, Iwata N, Takeuchi S, Nakashima K, Takagi H, Izumida H, Ochiai H, Fujisawa H, Suga H, Arima H, Shimoyama Y, Takahashi M, Nishioka H, Ishikawa SE, Shimatsu A, Caturegli P, Oiso Y (2015) Rabphilin-3A as a targeted autoantigen in lymphocytic infundibulo-neurohypophysitis. J Clin Endocrinol Metab 100(7):E946–E954. https://doi.org/10.1210/jc.2014-4209
Bando H, Iguchi G, Fukuoka H, Taniguchi M, Kawano S, Saitoh M, Yoshida K, Matsumoto R, Suda K, Nishizawa H, Takahashi M, Morinobu A, Kohmura E, Ogawa W, Takahashi Y (2015) A diagnostic pitfall in IgG4-related hypophysitis: infiltration of IgG4-positive cells in the pituitary of granulomatosis with polyangiitis. Pituitary 18(5):722–730. https://doi.org/10.1007/s11102-015-0650-9
Garon-Czmil J, Petitpain N, Rouby F, Sassier M, Babai S, Yelehe-Okouma M, Weryha G, Klein M, Gillet P (2019) Immune check point inhibitors-induced hypophysitis: a retrospective analysis of the French Pharmacovigilance database. Sci Rep 9(1):19419. https://doi.org/10.1038/s41598-019-56026-5
Yamamoto M, Iguchi G, Takeno R, Okimura Y, Sano T, Takahashi M, Nishizawa H, Handayaningshi AE, Fukuoka H, Tobita M, Saitoh T, Tojo K, Mokubo A, Morinobu A, Iida K, Kaji H, Seino S, Chihara K, Takahashi Y (2011) Adult combined GH, prolactin, and TSH deficiency associated with circulating PIT-1 antibody in humans. J Clin Invest 121(1):113–119. https://doi.org/10.1172/jci44073
Bando H, Iguchi G, Fukuoka H, Yamamoto M, Hidaka-Takeno R, Okimura Y, Matsumoto R, Suda K, Nishizawa H, Takahashi M, Tojo K, Takahashi Y (2014) Involvement of PIT-1-reactive cytotoxic T lymphocytes in anti-PIT-1 antibody syndrome. J Clin Endocrinol Metab 99(9):E1744–E1749. https://doi.org/10.1210/jc.2014-1769
Kanie K, Bando H, Iguchi G, Muguruma K, Matsumoto R, Hidaka-Takeno R, Okimura Y, Yamamoto M, Fujita Y, Fukuoka H, Yoshida K, Suda K, Nishizawa H, Ogawa W, Takahashi Y (2019) Pathogenesis of Anti-PIT-1 Antibody Syndrome: PIT-1 Presentation by HLA Class I on Anterior Pituitary Cells. J Endocr Soc 3(11):1969–1978. https://doi.org/10.1210/js.2019-00243
Mandai M, Watanabe A, Kurimoto Y, Hirami Y, Morinaga C, Daimon T, Fujihara M, Akimaru H, Sakai N, Shibata Y, Terada M, Nomiya Y, Tanishima S, Nakamura M, Kamao H, Sugita S, Onishi A, Ito T, Fujita K, Kawamata S, Go MJ, Shinohara C, Hata K-i, Sawada M, Yamamoto M, Ohta S, Ohara Y, Yoshida K, Kuwahara J, Kitano Y, Amano N, Umekage M, Kitaoka F, Tanaka A, Okada C, Takasu N, Ogawa S, Yamanaka S, Takahashi M (2017) Autologous induced stem-cell–derived retinal cells for macular degeneration. N Engl J Med 376(11):1038–1046. https://doi.org/10.1056/NEJMoa1608368
Miyagawa S, Domae K, Yoshikawa Y, Fukushima S, Nakamura T, Saito A, Sakata Y, Hamada S, Toda K, Pak K, Takeuchi M, Sawa Y (2017) Phase I Clinical Trial of Autologous Stem Cell-Sheet Transplantation Therapy for Treating Cardiomyopathy. J Am Heart Assoc. https://doi.org/10.1161/jaha.116.003918
Takebe T, Sekine K, Enomura M, Koike H, Kimura M, Ogaeri T, Zhang RR, Ueno Y, Zheng YW, Koike N, Aoyama S, Adachi Y, Taniguchi H (2013) Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499(7459):481–484. https://doi.org/10.1038/nature12271
Wimmer RA, Leopoldi A, Aichinger M, Wick N, Hantusch B, Novatchkova M, Taubenschmid J, Hämmerle M, Esk C, Bagley JA, Lindenhofer D, Chen G, Boehm M, Agu CA, Yang F, Fu B, Zuber J, Knoblich JA, Kerjaschki D, Penninger JM (2019) Human blood vessel organoids as a model of diabetic vasculopathy. Nature 565(7740):505–510. https://doi.org/10.1038/s41586-018-0858-8
Benam KH, Novak R, Ferrante TC, Choe Y, Ingber DE (2020) Biomimetic smoking robot for in vitro inhalation exposure compatible with microfluidic organ chips. Nat Protoc 15(2):183–206. https://doi.org/10.1038/s41596-019-0230-y
Sonoyama T, Sone M, Honda K, Taura D, Kojima K, Inuzuka M, Kanamoto N, Tamura N, Nakao K (2012) Differentiation of human embryonic stem cells and human induced pluripotent stem cells into steroid-producing cells. Endocrinology 153(9):4336–4345. https://doi.org/10.1210/en.2012-1060
Takebe T, Sekine K, Kimura M, Yoshizawa E, Ayano S, Koido M, Funayama S, Nakanishi N, Hisai T, Kobayashi T, Kasai T, Kitada R, Mori A, Ayabe H, Ejiri Y, Amimoto N, Yamazaki Y, Ogawa S, Ishikawa M, Kiyota Y, Sato Y, Nozawa K, Okamoto S, Ueno Y, Taniguchi H (2017) Massive and reproducible production of liver buds entirely from human pluripotent stem cells. Cell Rep 21(10):2661–2670. https://doi.org/10.1016/j.celrep.2017.11.005
Acknowledgements
This work was supported by grants from the Japan Agency for Medical Research and Development (AMED, The acceleration Program for Intractable Disease Research utilizing Disease-specific iPS cells: JP 18bm0804012h0002), grants in aid for Scientific Research from The Ministry of Education, Culture, Sports, Science and Technology (17K19684, 16H05332, to Y.T., and 18K16232 to R.M.), grants from the Uehara Memorial Foundation and the Naito Foundation, and grants from the Centor for iPS Cell Research and Application (CiRA, The Fellowship Program of Development of Young Researchers to R.M.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors have nothing to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Matsumoto, R., Takahashi, Y. Human pituitary development and application of iPSCs for pituitary disease. Cell. Mol. Life Sci. 78, 2069–2079 (2021). https://doi.org/10.1007/s00018-020-03692-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-020-03692-8