Abstract
Evidence from the literature keeps highlighting the impact of mutualistic bacterial communities of the gut microbiota on human health. The gut microbita is a complex ecosystem of symbiotic bacteria which contributes to mammalian host biology by processing, otherwise, indigestible nutrients, supplying essential metabolites, and contributing to modulate its immune system. Advances in sequencing technologies have enabled structural analysis of the human gut microbiota and allowed detection of changes in gut bacterial composition in several common diseases, including cardiometabolic disorders. Biological signals sent by the gut microbiota to the host, including microbial metabolites and pro-inflammatory molecules, mediate microbiome–host genome cross-talk. This rapidly expanding line of research can identify disease-causing and disease-predictive microbial metabolite biomarkers, which can be translated into novel biodiagnostic tests, dietary supplements, and nutritional interventions for personalized therapeutic developments in common diseases. Here, we review results from the most significant studies dealing with the association of products from the gut microbial metabolism with cardiometabolic disorders. We underline the importance of these postbiotic biomarkers in the diagnosis and treatment of human disorders.
Similar content being viewed by others
Introduction
The human gut microbiota is a complex system of mutualistic microorganisms, hosting an impressive 100 trillion bacteria from 500 to 1000 species representing 1–3% of body mass and encoding for over 4M genes [1, 2]. It functions as a bioreactor with enormous metabolic capacity and cooperates with the host in many biological functions to form a symbiotic mammalian superorganism [3]. Gut bacteria release bioactive molecules in the gut that can be used by gut mucosal cells or absorbed in the circulation and transported to the liver where they are transformed. The gut microbiota has raised considerable interest due to the possibility of carrying out deep microbiome sequence analysis [4, 5] and to use this information in association studies with various disease conditions. Changes in the architecture of the gut microbiome have been consistently associated with type 2 diabetes and obesity [6,7,8], which may be accounted for by low microbial gene richness suggesting reduced gut bacterial diversity in patients [9]. Relative resistance to diet-induced obesity and improved glucose tolerance in germ-free rodents also suggest that gut microbiota depletion affects host metabolism and susceptibility to diabetes and obesity [10]. However, the effects of antibiotic-mediated reduction in gut microbiota diversity on host metabolism and insulin sensitivity in humans remain controversial [11, 12].
The gut microbiota is the central regulator of mammalian fuel intake by processing nutrients into absorbable compounds. It also produces vitamins and essential metabolites that are not synthesised by the host. Changes in the architecture of the gut microbiota are likely to have important repercussion on the regulation of host biochemical pathways and metabolic networks. Many bacterial metabolite end-products of the gut microbiota play crucial roles in the host metabolic homeostasis, immunological processes, and neurobiology, and underline the fundamental importance of this extended genome in human health and disease [13,14,15]. This article reviews results from association studies of phenotypes relevant to cardiometabolic disorders with products from gut microbial metabolism illustrated in Fig. 1. We do not address intestinal nutrient sensing that triggers humoral and neural responses underlying important gut–brain cross-talk signaling mechanisms in diabetes and obesity, which has been recently reviewed [16] and addressed in landmark papers [17, 18]. This review paper underlines the enormous metabolic capacity of gut bacteria essential for the host and further underscores the importance of high-density metabolic data acquisition from biological samples to address whole-body regulation of biological processes mediated by microbial metabolites.
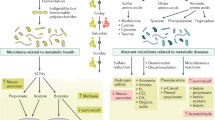
Examples of essential metabolites synthesised by gut bacteria. Metabolites produced by the gut microbiota from dietary substrates are transported to the liver where they can undergo enzymatic modification (e.g., TMA to TMAO), prior to transfer to the circulation and to other organs. SCFAs (predominantly butyrate) can be used locally as an energy source by gut mucosal cells. BCAAs branched-chain amino acids, SCFAs short-chain fatty acids
Short-chain fatty acids
Short-chain fatty acids (SCFAs) are produced by anaerobic gut bacteria in the caecum and the proximal colon principally through the fermentation of dietary fibers (e.g., nonstarch polysaccharides and low-digestible saccharides), and to a lesser extent of proteins and peptides [19]. The most abundant SCFAs are butyrate, acetate, and propionate [20]. SCFAs are used as an energy source by gut mucosal cells or transferred to the circulation to generate an important source of calory and energy for the organism and to act as signaling molecules. Upon synthesis by the gut microbiota, both propionate and butyrate have local effects as the primary energy source in by gut mucosal cells (butyrate) and by activating intestinal gluconeogenesis (propionate) through distinct mechanisms [18, 21]. Distal effects of SCFAs are illustrated by propionate-mediated stimulation of liver gluconeogenesis, de novo lipid synthesis, and protein synthesis, whereas acetate is a precursor for cholesterol synthesis.
SCFAs have multiple regulatory roles in energy homeostasis, insulin sensitivity, and glucose and lipid metabolism [22]. Association between elevated fecal SCFAs and obesity has emerged from studies in humans [23] and in mice treated with low doses of antibiotics [24]. However, there is a general agreement for their association with reduced risk of cardiovascular and metabolic diseases. Increased plasma insulin in response to propionate was initially demonstrated in healthy volunteers following long-term intracolonic administration of propionate, and was subsequently confirmed in vitro in human islets incubated with propionate [25]. Treatment of overweight individuals with colonic delivery of propionate results in reduced energy intake, adiposity, and lipid liver content, and increased plasma levels of peptide YY (PYY) and glucagon-like peptide 1 (GLP-1) produced locally by enteroendocrine L cells [26]. The effect of propionate on the production of PYY and GLP-1 was confirmed in mouse and rat models following intracolonic propionate administration, and in isolated colonocytes incubated with propionate [27]. Results from an extensive study in obese patients showed that rectal administration of individual SCFAs was associated with increased fasting fat oxidation and energy expenditure, decreased carbohydrate oxidation, and reduced whole-body lipolysis [28]. Further evidence of the beneficial role of microbial SCFAs on human health was obtained in a series of experiments in mice transplanted with gut microbiota from twin pairs discordant for obesity [29]. Mice inoculated with microbiota from lean co-twins showed higher caecal levels of butyrate and propionate than mice inoculated with gut bacteria from obese co-twins, suggesting that capacity to breakdown and ferment polysaccharides into SCFAs is greater in the microbiota from lean individuals than in that of obese patients.
Experiments in animal models have confirmed the associations of SCFAs with host metabolism and diseases. Glucose tolerance and insulin sensitivity are significantly improved in rats treated with diet enriched in butyrate or propionate [18]. Diet supplementation with butyrate in high-fat diet (HFD) fed mice resulted in broad ranging effects, including reduced obesity, glucose intolerance and insulin resistance, enhanced thermogenesis and mitochondrial function, and increased expression of PGC-1α in brown adipose tissue, muscle, and liver, which may be explained by increased activity of AMPK and p38 [30,31,32]. HFD-induced insulin resistance and obesity in rats is associated with increased plasma concentration of acetate produced by the gut microbiota [17]. Relationship between acetate and phenotypes related to diabetes and obesity is consistent with results from oral administration of acetate in the Otsuka Long Evans Tokushima Fatty (OLETF) rat model of genetically determined obesity and diabetes, which improved glucose tolerance and decreased body weight, hepatic lipid content, and abdominal fat [33].
Data in humans and in experimental models also demonstrated that elevated SCFA can contribute to lowering blood pressure and improving vascular phenotypes. Results from two independent meta-analyses of randomized-controlled trials suggested that increased SCFA induced by probiotics or dietary fiber intake was associated with a reduction in blood pressure in patients with hypertension [34, 35]. More recently, results from the metabolic analyses in the INTERMAP study population showed that 24-h urinary excretion of formate was positively correlated with urinary sodium excretion and inversely associated with both systolic and diastolic blood pressure [36]. Experiments in vivo in mice and in in vitro systems confirmed these observations. Intravenous administration of propionate in vivo in mice lowered blood pressure [37]. Mice fed diet rich in fiber or supplemented with acetate showed a significant reduction in high blood pressure, cardiac fibrosis, and left-ventricular hypertrophy induced by deoxycorticosterone acetate (DOCA) treatment in mice [38]. Repeated intraperitoneal injections of butyrate in mice chronically infused with angiotensin II were able to significantly reduce blood pressure [39]. In vitro experiments showed that butyrate, propionate, and acetate induced dilatation of human colonic resistance vessels [40], and exhibited vasorelaxant properties when tested in isolated rat caudal artery [41].
The cellular mediators of the effects of SCFAs are partly elucidated. Signaling of SCFAs is mediated by the G protein-coupled receptors GPR41 (FFAR3) and GPR43 (FFAR2), predominantly expressed in adipose tissue, intestine, and immune cells [42], as well as GPR109A (HCAR2) [43], and probably other as yet unknown mediators. In a series of elegant experiments in mice and in 3T3-L1 adipocytes, Kimura and colleagues showed that activation of GPR43 by SCFAs results in the inhibition of insulin signaling and reduction of lipid accumulation in fat [44]. On the other hand, another study suggests that relative resistance to obesity in mice-fed HFD supplemented with butyrate and propionate is independent from GPR41 activation [32]. GPR41 is also expressed in the vascular endothelium where it mediates the role of SCFAs in blood pressure regulation [37, 45]. Butyrate also plays an important role in gene expression through its inhibitory effect of histone deacetylases [30], which affects chromatin structure by deacetylation of proteins, and may affect nucleosome positioning [31].
Methylamines
Results from extensive studies of the metabolism of choline and methylamines in humans and rodent models are, perhaps, the most compelling illustrations played by the role of gut bacterial metabolites in cardiometabolic diseases. Trimethylamine (TMA) is an amine synthesised from dietary components, l-carnitine, lecithin, choline, and betaine by microbial enzymes (Fig. 2). TMA is oxidised into trimethylamine-N-oxide (TMAO) in the liver predominantly by the enzyme flavin-containing monooxygenase 3 (FMO3) [46]. We have recently demonstrated retroconversion of TMA from TMAO by gut bacteria (mostly Enterobacteriaceae) in vivo in mice, which can, therefore, increase the pool of circulating TMA that can be oxidised back to TMAO by FMO3 in the liver [47].
Representation of the methylamine pathway illustrating the microbiota–host co-metabolism. Bacterial enzymes use dietary substrates, choline, betaine, l-carnitine, γ-butyrobetaine, trimethylamine-N-oxide (TMAO) to synthesise trimethylamine (TMA), which is transferred across the intestinal endothelium to the circulation and transported to the liver where it is metabolised into TMAO by the enzyme flavin-containing monooxygenase (FMO3). The TMA substrate betaine can be synthesised from choline and l-carnitine. 1: Choline TMA lyase; 2: betaine reductase; 3: carnitine reductase; 4: carnitine TMA lyase; 5: TMAO reductase; 6: choline dehydrogenase; 7: betaine aldehyde dehydrogenase; 8: l-carnitine dehydrogenase; 9: γ-butyrobetaine dioxygenase; 10: γ-butyrobetainyl-CoA: carnitine CoA transferase
Relationships between TMAO and cardiovascular risk are based on correlative inferences. The multiple detrimental roles of TMAO on human health are still debated [48,49,50] and thus keep raising interest. For example, elevated plasma levels of TMAO secondary to l-carnitine treatment in ApoE knock-out mice expressing the cholesteryl-ester transfer protein (CEPT) resulted in a reduction of aortic lesions regardless of plasma lipid and lipoprotein levels, and did not alter the formation of macrophage foam cell [51]. The initial observations in humans showing that choline deficiency results in hepatic steatosis reversible by choline supplementation have demonstrated the importance of choline metabolism for the host [52]. Evidence of the pathophysiological impact of choline and methylamines on insulin resistance, fatty liver disease, and obesity was obtained by analyses of plasma and urine metabolomic profiles of mouse strains strongly susceptible (129S6) or resistant (BALB/c) to HFD-induced glucose intolerance, obesity, and fatty liver disease [53]. Fat-fed 129S6 mice exhibited the disruptions of choline metabolism characterised by reduction in plasma levels of phosphatidylcholine and elevated urinary excretion of dimethylamine, TMA, and TMAO. Conversion of choline into methylamines by microbiota in 129S6 mice on HFD resulted in the reduction of the bioavailability of choline.
Subsequent investigations in humans mostly focused on the endpoint of the methylamine pathway (TMAO), which is synthesised by the mammalian metabolism, rather than on its substrate (TMA) synthesised by the gut microbiome. Elevated plasma levels of TMAO have been associated with increased risk of type 2 diabetes mellitus [54], cardiovascular and cerebrovascular diseases [55,56,57], incident thrombosis risk [58], and carotid intima-media thickness [59] in population-based and intervention studies. Further experiments in mice indicated that elevated levels of circulating TMAO resulted in enhanced aortic atherosclerotic plaque lesions, increased thrombus formation in the carotid artery, perturbed bile acid metabolism, downregulated expression of genes involved in reverse cholesterol transport, and stimulated expression of two macrophage scavenger receptors (CD36, SRA), without significant changes in plasma lipids, glycemia, and hepatic triglycerides [56,57,58]. In vitro studies showed that TMAO enhanced the function of human platelets and increased platelet adhesion through increased intracellular release of Ca2+ and stimulation of inositol-1,4,5-triphosphate signaling [58].
Most recently, a study in HFD-fed mice showed that urinary TMAO prior to the dietary challenge is the most significant predictive marker of future heterogeneity in physiological and behavioral anomalies [60]. Variable phenotypic adaptation to HFD in inbred mice is a well-known phenomenon [61, 62], which was recently investigated in groups of HFD-fed isogenic C57BL/6J mice through deep physiological and behavioral phenotyping coupled with the combined analyses of the urine metabolome and adipose tissue transcriptome [60]. Variations in urine levels of choline, TMAO, dimethylamine (DMA), and monomethylamine (MMA) are metabolic markers of adaptation to HFD feeding in isogenic mice (Fig. 3). These changes already exist prior to the dietary challenge and predicted future divergence in disease patterns characterised by various degrees of glucose intolerance and obesity, which correlated with significant divergences in insulin secretion, circulating triglycerides and lipoproteins, and measures of anxiety and activity between extreme responder groups [60]. For example, mice that developed glucose intolerance in response to HFD exhibited a significant increase in urine concentration of products of choline metabolism (e.g., TMA, TMAO, DMA, and dimethylglycine—DMG) prior to HFD-feeding induction, when compared to HFD-fed mice that maintained normal glucose tolerance. This study also showed that chronic subcutaneous infusion of TMAO in HFD-fed mice resulted in paradoxical improvement of both glucose tolerance and glucose-induced insulin secretion, which was confirmed in vitro in isolated pancreatic islets incubated with TMAO. These findings suggest the existence of differences in the composition or the activity of gut bacteria between isogenic individuals prior to any dietary stimulus contribute to the biosynthesis of TMA and predispose to future development of disease phenotypes. The possible implication of the epigenome in this phenomenon is supported by a recent study, showing that depletion of circulating choline in mice caused by intestinal colonisation of choline-utilising bacterial communities results in reduced DNA methylation in several organs and deteriorated metabolic and behavioral phenotypes [63].

Adapted from Dumas et al. [60]
Methylamine-mediated discrimination of adaptation to dietary challenge in isogenic mice. Orthogonal partial least-squares discriminant analysis (O-PLS-DA) scores plots built on 1H NMR metabolomics of 24-h urinary collections was used to discriminate isogenic C57BL/6J mice fed carbohydrate diet (blue) or obesogenic high-fat diet (red) (a). Assigned O-PLS-DA model coefficient plot underlines the power of TMA, TMAO and choline to discriminate diet groups (b).
Despite growing interest in TMAO biology, its cellular ligands remain unknown. Identification of cellular ligands of TMAO is the next milestone to demonstrate causality with diseases. In contrast, a specific ligand for its precursor TMA has been identified [64]. Following a screening of 42 amines and amine-related compounds, TMA, and to a lesser extent dimethylethylamine (DMEA), activate the trace amine-associated receptor TAAR5, an olfactory G protein-coupled receptor, in a concentration-dependent manner. However, little is known about the biological roles of TMA. Using a mass spectrometry (MS) quantitative assay targeting carnitine, choline, betaine, TMA, and TMAO, a significant association between coronary calcified plaque and cardiovascular risk in humans and serum TMA, and not TMAO, was reported [65]. Urinary levels of TMA, DMA, and DMG, another product of microbial metabolism of choline, have also been associated with Body Mass Index in humans [66]. The existence of multiple TMA substrates (choline, phosphatidylcholine, l-carnitine, betaine, γ-butyrobetaine, and TMAO) and the wide variety of food sources (red meat, fish, egg, and dairy products) that contain TMA or substrates that the gut microbiota can use for TMA biosynthesis, perhaps, prevent systematic nutritional recommendations based on the results obtained until now on methylamines.
Branched-chain amino acids
The branched-chain amino acids (BCAAs), leucine, isoleucine, and valine, are among the nine essential amino acids synthesised by gut bacteria [67]. They are characterised structurally by aliphatic side-chains with a “branch” (one carbon in the centre linked to three or more carbon atoms). A significant association of the biosynthesis of BCAAs and tryptophan with the bacterial species Prevotella copri provided the evidence of functional relationships between changes in the composition of the gut microbiome and these metabolites, as well as potentially indolelactic acid, in insulin-resistant patients [68]. The direct role of BCAAs in the stimulation of insulin secretion [69] and correlations between elevated concentration of plasma BCAAs with obesity and serum insulin [70] have been known for decades. The possible exploitation of BCAAs as biomarkers of cardiometabolic diseases has been addressed in multiple independent patient and population studies, and underlined the complex relationships between circulating BCAAs and disease risk, and the possible implication of confounding factors. Association of plasma BCAAs with insulin resistance was reported in obese [71], non-obese [72], and non-diabetic [68, 73, 74] individuals, and confirmed in twins [75]. Similarly, association between levels of circulating BCAAs and adipokine was evidenced in both diabetic [76] and non-diabetic [74] individuals. Using various study designs and analytical systems, plasma concentrations of all three BCAAs were also found significantly increased in diabetic patients of the German KORA study [77], in female patients for the TwinsUK collection [78], and in a Swedish prospective case control study [79]. Urinary levels of BCAAs were also associated with Body Mass Index in the INTERMAP study [66].
The impact of BCAAs on cardiometabolic phenotypes has been further explored in rodent models and in humans. Addition of BCAAs to HFD in rats reduced the increase in body growth normally caused by HFD, independently from any to significant changes in insulin resistance, which was associated with increased activity of mTOR and JNK [71]. Conversely, dietary restriction in BCAAs improved glucose homeostasis in mice fed control chow diet [80] and reduced adiposity and glucose intolerance induced by HFD in mice [81]. Inoculation of gut microbiota prepared from twin pairs discordant for obesity in mice showed that BCAAs were more elevated when the donor was the obese co-twin, leading to an obese phenotype in transplanted mice, than when the donor was the lean co-twin [29].
Evidence for a causal role of BCAAs in human diabetes was suggested in longitudinal and genetic studies. 12-year follow-up and prospective studies in normoglycemic individuals demonstrated that BCAAs individually and in combination predict future development of type 2 diabetes, suggesting that BCAAs may actually assist the identification of individuals at risk of developing the disease [82]. Most recently, extensive statistical analysis based on Mendelian randomisation in several large populations showed association between variables related to insulin resistance (homeostasis model assessment of insulin resistance—HOMA-IR, fasting insulin) and fasting plasma levels of these BCAAs, but failed to identify significant causal effect of BCAAs on either fasting insulin or HOMA-IR [83]. In contrast, the genetic risk score for insulin resistance traits was significantly associated with increased concentration of plasma BCAAs, indicating a causal impact of insulin resistance on circulating BCAAs. In a large-scale genome-wide association study (GWAS), causal relationships between increased type 2 diabetes risk and high levels of BCAAs determined genetically by genetic polymorphisms at five independent genomic regions were further evidenced through Mendelian randomisation in a meta-analysis [84]. The strongest evidence of association was found with BCAA-raising polymorphisms upstream the gene encoding the protein phosphatase PPM1K on chromosome 4q22.1, which activates the mitochondrial branched-chain alpha-ketoacid dehydrogenase (BCKD) and is, therefore, an attractive candidate to explain this genetic association.
Bile acids
Bile acids are steroid molecules produced in the liver from cholesterol and, subsequently, processed into secondary bile acids by the gut microbiota. Knowledge of the relationship between gut microbiota and bile acid homeostasis stems from experiments in germ-free mice [85], in animals treated with broad-spectrum antibiotics [86] and in gastrectomised mice [87]. The effects of bile acids on the host metabolism are mediated through regulation of the cholesterol 7-α hydroxylase (CYP7A1) and binding to the nuclear farnesoid X receptor (FXR, NR1H4) and a G protein-coupled receptor (TGR5, GPBAR1). The broad ranging biological functions of bile acids can be explained by the expression of these mediators in many organs involved in cardiovascular and metabolic diseases (liver, adipose tissue, skeletal muscle, pancreatic β cells, and heart), as well as in gut epithelial cells. Bile acid metabolism in health and disease and the central roles of FXR and TGR5 in the concomitant regulation of gut microbiota composition, glucose and lipid metabolism, as well as inflammation and energy expenditure have all been recently reviewed [88,89,90] and are not addressed in detail here. Bile acid metabolism has been more particularly investigated in response to bariatric surgery (e.g., Roux-en-Y gastric bypass; vertical sleeve gastrectomy). There is now compelling evidence that weight independent metabolic benefits and diabetes remission following bariatric surgery in humans and experimental models is associated with changes in FXR signaling [91], increased plasma levels of bile acids and changes in their composition, as well as altered gut microbiota ecology [91,92,93]. Both modulation of bile acid metabolism and utilisation of FXR- and TGR5-agonists, therefore, represent attractive though challenging, therapeutic options in type 2 diabetes, non-alcoholic fatty liver disease (NAFLD), and non-alcoholic steatohepatitis (NASH) [94,95,96,97], which are, nevertheless, complicated by the expression of FXR and TGR5 in many tissues.
Polyphenols and indole derivatives
Polyphenols are plant metabolites formed by hydroxylated phenolic rings. Fermentation of dietary polyphenols (mainly hydroxycinnamic acids and flavonoids) by the gut microbiota results in the production of small bioactive compounds consisting of phenolic acids, which can be absorbed by the gut. Products of plant polyphenols have been identified experimentally through systems of in vitro fermentation by human colonic microbiota [98, 99] and incubation with primary hepatocytes [99]. Polyphenols affect the gut microbiota architecture and stimulate bacterial production of SCFAs [100], and can, therefore, have indirect beneficial effects on host metabolism. Absorption and transport of phenolic acids to target tissues are reduced in diabetes and obesity in humans and rodent models [101].
Many intestinal bacteria are able to generate phenolic compounds and indoles from dietary amino acids [102, 103]. 4-cresol (p-cresol, 4-methylphenol) is a phenol metabolised from tyrosine by gut bacteria, mostly by Firmicutes (Clostridiaceae, Eubacteriaceae, Lacnospiraceae, Ruminococcaceae, and Staphylococcaceae) and by bacteroidetes, actinobacteria, and fusobacteria. It is also naturally present at low levels in foods (tomatoes, asparagus, cheeses, butter, bacon, and smoked products) and drinks (coffee, black tea, wine, scotch, whiskey, brandy, and rum). As for the metabolism of choline and methylamines discussed above, clinical and fundamental research focused on 4-cresyl sulfate, which is the product of 4-cresol sulfation in the gut mucosa and liver and is a well-known protein-bound uremic toxin associated with chronic kidney disease [102]. However, the metabolic effects of 4-cresol are largely unexplored. 4-cresol inhibits hepatic drug-metabolising cytochrome P450 and UDP-glucuronosyltransferase enzymes in vitro in human liver microsomes [104]. High concentrations of 4-cresol (100–200 μM) inhibit the differentiation of 3T3-L1 preadipocytes into mature adipocytes, induce apoptosis, and decrease glucose uptake [105], thus indicating that 4-cresol metabolism affects the function of other organs than kidney. Vanillic is another example of a product of biotransformation of polyphenols by the gut microbiota, which has the capacity to reduce hyperinsulinemia, hyperglycemia, hyperlipidemia, and hepatic insulin resistance in high-fat diet-fed rats [106].
The role of indole derivatives in cardiovascular and metabolic diseases is emerging from studies in humans. Indole is a signaling molecule produced exclusively by bacterial tryptophanases from the essential amino acid tryptophan supplied in the diet [107]. Indoxylsulfate is a protein-bound uremic toxin formed through hydroxylation of indole in the liver followed by O-sulfation. Serum concentrations of indoxylsulfate are associated with aortic calcification, arterial stiffness, and increased cardiovascular mortality in patients with chronic kidney disease [108]. Furthermore, indoxylsulfate, as well as indoxyl acetate, activate the aryl hydrocarbon receptor (AHR) pathway in primary human aortic vascular smooth muscle cells and promote thrombosis through upregulation of the expression of tissue factor and inhibition of ubiquitination and degradation of tissue factor [109, 110]. Finally, human population studies showed that serum indole propionate was associated with reduced risk of type 2 diabetes and preserved β-cell function, and negatively correlated with low-grade inflammation. Elevated serum indole propionate was associated with dietary fiber intake, consistent with a functional relationship between diet, gut microbial metabolism, and disease risk [111].
Other metabolites
Benzoic acid is an aromatic carboxylic acid synthesised in the gut through fermentation by colonic microbiota of dietary aromatic compounds, including aromatic amino acids present in plants [98]. It is used as a food preservative (E211), and is often found in sodas and ready-made meals. It is conjugated in the liver with glycine to produce hippuric acid, which is further conjugated and eliminated in the urine. In a genetic study, we identified that elevated levels of plasma benzoic acid in the Goto–Kakizaki rat model of polygenic type 2 diabetes is controlled by a genetic locus containing a deletion in a uridine diphosphate glucuronosyltransferase [112]. This study was the first demonstration of the genetic control of products of microbial metabolism by host genes that contribute to disease susceptibility. Metabolome-wide association studies in humans identified associations between type 2 diabetes and benzoic acid levels in plasma [113, 114] and urine [113]. On the other hand, results from a randomized, cross-over trial in overweight subjects showed that oral administration of sodium benzoic acid had no significant effects on glucose homeostasis [115].
Biosynthesis and excretion of hippuric acid, the product of benzoic acid degradation, have been reviewed in detail [116]. The implication of this product of microbial–mammalian co-metabolism in human diseases was initially obtained in a metabolome-wide association study of the INTERMAP UK cohort, which demonstrated inverse correlation between urine levels of hippuric acid and hypertension [36] and Body Mass Index [66]. Investigations in small groups of patients and lean controls confirmed association between low levels of urinary hippuric acid and obesity [117] and type 2 diabetes [118]. Interestingly, hippuric acid strongly increased in obese or diabetic individuals who underwent bariatric surgery [117, 119], suggesting that this metabolite may represent a clinical biomarker of disease remission. Urinary levels of hippuric acid were decreased in spontaneously obese rats of the Zucker (fa/fa) strain mutant for the leptin receptor gene when compared to lean controls heterozygous for the fa mutation [118]. This observation was replicated in spontaneously hypertensive rats (SHR) [120] when compared to Wistar Kyoto rats. Variations in urinary levels of hippuric acid prior to HFD feeding in isogenic mice were significantly associated with the future development of obesity, suggesting that changes in the architecture and/or function of the gut microbiota predict disease risk independently of genetic variations [60]. Evidence of direct relationships between hippuric acid and the gut microbiome was recently suggested through the observation of a correlation between urine hippuric acid levels and microbiota ecological diversity determined by 16S rDNA motifs frequency [121].
Fermentation of dietary fibers by the gut microbiota can produce other metabolites than SCFAs that are also synthesised by the host. The organic acid succinate is an intermediate in the synthesis of propionate by gut bacteria and also an abundant product of microbial metabolism which is used as a substrate for intestinal gluconeogenesis [122]. A metabolome-wide association study in the INTERMAP population identified an association between urinary levels of succinate and Body Mass Index [66]. Dietary supplementation with succinate in mice improves glucose tolerance, insulin sensitivity, and energy metabolism, and reduces body growth [122].
Concluding remarks
The gut microbiome sits at the interface between the host, nutritional, and inflammatory environments, and there is growing evidence of a role of bacterial metabolites as biomarkers of pathophysiological features of cardiometabolic diseases, with applications in disease diagnostics and prognosis in precision medicine. Application of high-density metabolite profiling systems is now essential to broaden the spectrum of microbial metabolites simultaneously detected in biological samples, particularly in biofluids (blood, urine), which can be collected in large populations with minimally invasive methods, as well as in extracts from organ biopsies. Regardless of the biological matrix (plasma, serum, urine, cell preparations, and organ extracts) and the technology used (nuclear magnetic resonance spectroscopy and mass spectrometry), metabolomics generates high-resolution spectra for hundreds to thousands of metabolite signals [123, 124], and has enormous potential in systematic metabolic phenotyping [125], with numerous applications in toxicology [126], prediction of treatment outcomes [127, 128], and molecular epidemiology [36, 66]. It is also a powerful system to document in detail the global functional output of changes in the gut microbiome architecture through qualitative and quantitative analyses of the mammalian-microbial co-metabolome [129], to provide novel insights into disease-predictive and disease-associated biomarkers. It is particularly important, since many foodstuff in westernised societies, including ready-made meals, often contain many synthetic and natural additives (e.g., sweeteners, stabilizers, emulsifiers, thickeners, flavor enhancers, and coloring agents) which modify qualitatively and quantitatively the gut microbiota ecosystem, and can have unpredictable beneficial or adverse biological effects in the host. Artificial sweeteners and emulsifiers (carboxymethylcellulose, polysorbate-80) in the diet alter gut microbiota architecture, and promote glucose intolerance, low-grade inflammation, and obesity [130, 131]. Of note, artificial additives may be metabolised by gut bacteria to unforeseen compounds, which can be detected in biological samples only through systematic untargeted metabolomic profiling methods (i.e., analysis of full-metabolomic spectral data).
Mapping the genetic control of microbial metabolites to the human genome is an important perspective, which will improve our understanding of transgenomic regulations [132] and infer causality to diseases when metabolites and disease traits are controlled by the same genetic loci. Ultimately, the effects of microbial metabolites on health can be altered by naturally occurring polymorphisms in host genes encoding proteins (transporters, receptors, and enzymes) that contribute to their metabolism. Positive correlation of circulating TMAO levels with body weight and adiposity in the hybrid mouse diversity panel provides opportunities to identify genes controlling TMAO metabolism [54]. Deciphering the implication of bacterial metabolites on epigenetic regulations, as demonstrated for TMAO and SCFAs, is also an area of promising research to understand the cross-talk between the gut microbiome and the genome of the host [133, 134].
Therapeutic applications of microbial metabolites require full characterisation of their cellular receptors and the underlying signaling pathways, which are in many cases unknown. Finally, potential therapeutic solutions of this research lie in the identification of bacterial ecosystems able to synthesise metabolites that play a beneficial role in the host metabolism, for their inoculation in patients.
Abbreviations
- AHR:
-
Aryl hydrocarbon receptor
- BCAAs:
-
Branched-chain amino acids
- CEPT:
-
Cholesteryl-ester transfer protein
- CYP7A1:
-
Cholesterol 7-α hydroxylase
- DMA:
-
Dimethylamine
- DMEA:
-
Dimethylethylamine
- DMG:
-
Dimethylglycine
- FMO3:
-
Flavin-containing monooxygenase 3
- FXR:
-
Farnesoid X receptor
- GLP-1:
-
Glucagon-like peptide 1
- HFD:
-
High-fat diet
- MMA:
-
Monomethylamine
- MS:
-
Mass spectrometry
- NAFLD:
-
Non-alcoholic fatty liver disease
- NASH:
-
Non-alcoholic steatohepatitis
- OLETF:
-
Otsuka Long Evans Tokushima Fatty
- PYY:
-
Peptide YY
- SCFAs:
-
Short-chain fatty acids
- TMA:
-
Trimethylamine
- TMAO:
-
Trimethylamine-N-oxide
References
Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, Gill SR, Nelson KE, Relman DA (2005) Diversity of the human intestinal microbial flora. Science 308:1635–1638
Blaser MJ, Falkow S (2009) What are the consequences of the disappearing human microbiota? Nat Rev Microbiol 7:887–894
Fischbach MA, Segre JA (2016) Signaling in host-associated microbial communities. Cell 164:1288–1300
Qin J, Li R, Raes J, Arumugam M, Burgdorf KS, Manichanh C, Nielsen T, Pons N, Levenez F, Yamada T, Mende DR, Li J, Xu J, Li S, Li D, Cao J, Wang B, Liang H, Zheng H, Xie Y, Tap J, Lepage P, Bertalan M, Batto JM, Hansen T, Le Paslier D, Linneberg A, Nielsen HB, Pelletier E, Renault P, Sicheritz-Ponten T, Turner K, Zhu H, Yu C, Jian M, Zhou Y, Li Y, Zhang X, Qin N, Yang H, Wang J, Brunak S, Doré J, Guarner F, Kristiansen K, Pedersen O, Parkhill J, Weissenbach J, Bork P, Ehrlich SD, M. Consortium (2010) A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464:59–65
Lloyd-Price J, Mahurkar A, Rahnavard G, Crabtree J, Orvis J, Hall AB, Brady A, Creasy HH, McCracken C, Giglio MG, McDonald D, Franzosa EA, Knight R, White O, Huttenhower C (2017) Strains, functions and dynamics in the expanded Human Microbiome Project. Nature 550:61–66
Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE, Sogin ML, Jones WJ, Roe BA, Affourtit JP, Egholm M, Henrissat B, Heath AC, Knight R, Gordon JI (2009) A core gut microbiome in obese and lean twins. Nature 457:480–484
Forslund K, Hildebrand F, Nielsen T, Falony G, Le Chatelier E, Sunagawa S, Prifti E, Vieira-Silva S, Gudmundsdottir V, Pedersen HK, Arumugam M, Kristiansen K, Voigt AY, Vestergaard H, Hercog R, Costea PI, Kultima JR, Li J, Jørgensen T, Levenez F, Dore J, Nielsen HB, Brunak S, Raes J, Hansen T, Wang J, Ehrlich SD, Bork P, Pedersen O, M. Consortium (2015) Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature 528:262–266
Qin J, Li Y, Cai Z, Li S, Zhu J, Zhang F, Liang S, Zhang W, Guan Y, Shen D, Peng Y, Zhang D, Jie Z, Wu W, Qin Y, Xue W, Li J, Han L, Lu D, Wu P, Dai Y, Sun X, Li Z, Tang A, Zhong S, Li X, Chen W, Xu R, Wang M, Feng Q, Gong M, Yu J, Zhang Y, Zhang M, Hansen T, Sanchez G, Raes J, Falony G, Okuda S, Almeida M, LeChatelier E, Renault P, Pons N, Batto JM, Zhang Z, Chen H, Yang R, Zheng W, Yang H, Wang J, Ehrlich SD, Nielsen R, Pedersen O, Kristiansen K (2012) A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490:55–60
Le Chatelier E, Nielsen T, Qin J, Prifti E, Hildebrand F, Falony G, Almeida M, Arumugam M, Batto JM, Kennedy S, Leonard P, Li J, Burgdorf K, Grarup N, Jørgensen T, Brandslund I, Nielsen HB, Juncker AS, Bertalan M, Levenez F, Pons N, Rasmussen S, Sunagawa S, Tap J, Tims S, Zoetendal EG, Brunak S, Clément K, Doré J, Kleerebezem M, Kristiansen K, Renault P, Sicheritz-Ponten T, de Vos WM, Zucker JD, Raes J, Hansen T, Bork P, Wang J, Ehrlich SD, Pedersen O, M. Consortium (2013) Richness of human gut microbiome correlates with metabolic markers. Nature 500:541–546
Bäckhed F, Ding H, Wang T, Hooper LV, Koh GY, Nagy A, Semenkovich CF, Gordon JI (2004) The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci USA 101:15718–15723
Reijnders D, Goossens GH, Hermes GD, Neis EP, van der Beek CM, Most J, Holst JJ, Lenaerts K, Kootte RS, Nieuwdorp M, Groen AK, Olde Damink SW, Boekschoten MV, Smidt H, Zoetendal EG, Dejong CH, Blaak EE (2016) Effects of gut microbiota manipulation by antibiotics on host metabolism in obese humans: a randomized double-blind placebo-controlled trial. Cell Metab 24:63–74
Vrieze A, Out C, Fuentes S, Jonker L, Reuling I, Kootte RS, van Nood E, Holleman F, Knaapen M, Romijn JA, Soeters MR, Blaak EE, Dallinga-Thie GM, Reijnders D, Ackermans MT, Serlie MJ, Knop FK, Holst JJ, van der Ley C, Kema IP, Zoetendal EG, de Vos WM, Hoekstra JB, Stroes ES, Groen AK, Nieuwdorp M (2014) Impact of oral vancomycin on gut microbiota, bile acid metabolism, and insulin sensitivity. J Hepatol 60:824–831
Nicholson JK, Holmes E, Kinross J, Burcelin R, Gibson G, Jia W, Pettersson S (2012) Host–gut microbiota metabolic interactions. Science 336:1262–1267
Brown JM, Hazen SL (2018) Microbial modulation of cardiovascular disease. Nat Rev Microbiol 16:171–181
Sonnenburg JL, Bäckhed F (2016) Diet-microbiota interactions as moderators of human metabolism. Nature 535:56–64
Clemmensen C, Müller TD, Woods SC, Berthoud HR, Seeley RJ, Tschöp MH (2017) Gut–brain cross-talk in metabolic control. Cell 168:758–774
Perry RJ, Peng L, Barry NA, Cline GW, Zhang D, Cardone RL, Petersen KF, Kibbey RG, Goodman AL, Shulman GI (2016) Acetate mediates a microbiome–brain–β-cell axis to promote metabolic syndrome. Nature 534:213–217
De Vadder F, Kovatcheva-Datchary P, Goncalves D, Vinera J, Zitoun C, Duchampt A, Bäckhed F, Mithieux G (2014) Microbiota-generated metabolites promote metabolic benefits via gut–brain neural circuits. Cell 156:84–96
Koh A, De Vadder F, Kovatcheva-Datchary P, Bäckhed F (2016) From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell 165:1332–1345
LeBlanc JG, Chain F, Martín R, Bermúdez-Humarán LG, Courau S, Langella P (2017) Beneficial effects on host energy metabolism of short-chain fatty acids and vitamins produced by commensal and probiotic bacteria. Microb Cell Fact 16:79
Donohoe DR, Garge N, Zhang X, Sun W, O’Connell TM, Bunger MK, Bultman SJ (2011) The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metab 13:517–526
Canfora EE, Jocken JW, Blaak EE (2015) Short-chain fatty acids in control of body weight and insulin sensitivity. Nat Rev Endocrinol 11:577–591
Schwiertz A, Taras D, Schäfer K, Beijer S, Bos NA, Donus C, Hardt PD (2010) Microbiota and SCFA in lean and overweight healthy subjects. Obesity (Silver Spring) 18:190–195
Cho I, Yamanishi S, Cox L, Methé BA, Zavadil J, Li K, Gao Z, Mahana D, Raju K, Teitler I, Li H, Alekseyenko AV, Blaser MJ (2012) Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 488:621–626
Pingitore A, Chambers ES, Hill T, Maldonado IR, Liu B, Bewick G, Morrison DJ, Preston T, Wallis GA, Tedford C, Castañera González R, Huang GC, Choudhary P, Frost G, Persaud SJ (2017) The diet-derived short chain fatty acid propionate improves beta-cell function in humans and stimulates insulin secretion from human islets in vitro. Diabetes Obes Metab 19:257–265
Chambers ES, Viardot A, Psichas A, Morrison DJ, Murphy KG, Zac-Varghese SE, MacDougall K, Preston T, Tedford C, Finlayson GS, Blundell JE, Bell JD, Thomas EL, Mt-Isa S, Ashby D, Gibson GR, Kolida S, Dhillo WS, Bloom SR, Morley W, Clegg S, Frost G (2015) Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 64:1744–1754
Psichas A, Sleeth ML, Murphy KG, Brooks L, Bewick GA, Hanyaloglu AC, Ghatei MA, Bloom SR, Frost G (2015) The short chain fatty acid propionate stimulates GLP-1 and PYY secretion via free fatty acid receptor 2 in rodents. Int J Obes (Lond) 39:424–429
Canfora EE, van der Beek CM, Jocken JWE, Goossens GH, Holst JJ, Olde Damink SWM, Lenaerts K, Dejong CHC, Blaak EE (2017) Colonic infusions of short-chain fatty acid mixtures promote energy metabolism in overweight/obese men: a randomized crossover trial. Sci Rep 7:2360
Ridaura VK, Faith JJ, Rey FE, Cheng J, Duncan AE, Kau AL, Griffin NW, Lombard V, Henrissat B, Bain JR, Muehlbauer MJ, Ilkayeva O, Semenkovich CF, Funai K, Hayashi DK, Lyle BJ, Martini MC, Ursell LK, Clemente JC, Van Treuren W, Walters WA, Knight R, Newgard CB, Heath AC, Gordon JI (2013) Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 341:1241214
Gao Z, Yin J, Zhang J, Ward RE, Martin RJ, Lefevre M, Cefalu WT, Ye J (2009) Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 58:1509–1517
Henagan TM, Stefanska B, Fang Z, Navard AM, Ye J, Lenard NR, Devarshi PP (2015) Sodium butyrate epigenetically modulates high-fat diet-induced skeletal muscle mitochondrial adaptation, obesity and insulin resistance through nucleosome positioning. Br J Pharmacol 172:2782–2798
Lin HV, Frassetto A, Kowalik EJ, Nawrocki AR, Lu MM, Kosinski JR, Hubert JA, Szeto D, Yao X, Forrest G, Marsh DJ (2012) Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS One 7:e35240
Yamashita H, Fujisawa K, Ito E, Idei S, Kawaguchi N, Kimoto M, Hiemori M, Tsuji H (2007) Improvement of obesity and glucose tolerance by acetate in Type 2 diabetic Otsuka Long-Evans Tokushima Fatty (OLETF) rats. Biosci Biotechnol Biochem 71:1236–1243
Whelton SP, Hyre AD, Pedersen B, Yi Y, Whelton PK, He J (2005) Effect of dietary fiber intake on blood pressure: a meta-analysis of randomized, controlled clinical trials. J Hypertens 23:475–481
Khalesi S, Sun J, Buys N, Jayasinghe R (2014) Effect of probiotics on blood pressure: a systematic review and meta-analysis of randomized, controlled trials. Hypertension 64:897–903
Holmes E, Loo RL, Stamler J, Bictash M, Yap IK, Chan Q, Ebbels T, De Iorio M, Brown IJ, Veselkov KA, Daviglus ML, Kesteloot H, Ueshima H, Zhao L, Nicholson JK, Elliott P (2008) Human metabolic phenotype diversity and its association with diet and blood pressure. Nature 453:396–400
Pluznick JL, Protzko RJ, Gevorgyan H, Peterlin Z, Sipos A, Han J, Brunet I, Wan LX, Rey F, Wang T, Firestein SJ, Yanagisawa M, Gordon JI, Eichmann A, Peti-Peterdi J, Caplan MJ (2013) Olfactory receptor responding to gut microbiota-derived signals plays a role in renin secretion and blood pressure regulation. Proc Natl Acad Sci USA 110:4410–4415
Marques FZ, Nelson E, Chu PY, Horlock D, Fiedler A, Ziemann M, Tan JK, Kuruppu S, Rajapakse NW, El-Osta A, Mackay CR, Kaye DM (2017) High-fiber diet and acetate supplementation change the gut microbiota and prevent the development of hypertension and heart failure in hypertensive mice. Circulation 135:964–977
Kim S, Goel R, Kumar A, Qi Y, Lobaton G, Hosaka K, Mohammed M, Handberg EM, Richards EM, Pepine CJ, Raizada MK (2018) Imbalance of gut microbiome and intestinal epithelial barrier dysfunction in patients with high blood pressure. Clin Sci (Lond) 132:701–718
Mortensen FV, Nielsen H, Mulvany MJ, Hessov I (1990) Short chain fatty acids dilate isolated human colonic resistance arteries. Gut 31:1391–1394
Nutting CW, Islam S, Daugirdas JT (1991) Vasorelaxant effects of short chain fatty acid salts in rat caudal artery. Am J Physiol 261:H561–567
Brown AJ, Goldsworthy SM, Barnes AA, Eilert MM, Tcheang L, Daniels D, Muir AI, Wigglesworth MJ, Kinghorn I, Fraser NJ, Pike NB, Strum JC, Steplewski KM, Murdock PR, Holder JC, Marshall FH, Szekeres PG, Wilson S, Ignar DM, Foord SM, Wise A, Dowell SJ (2003) The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J Biol Chem 278:11312–11319
Thangaraju M, Cresci GA, Liu K, Ananth S, Gnanaprakasam JP, Browning DD, Mellinger JD, Smith SB, Digby GJ, Lambert NA, Prasad PD, Ganapathy V (2009) GPR109A is a G-protein-coupled receptor for the bacterial fermentation product butyrate and functions as a tumor suppressor in colon. Cancer Res 69:2826–2832
Kimura I, Ozawa K, Inoue D, Imamura T, Kimura K, Maeda T, Terasawa K, Kashihara D, Hirano K, Tani T, Takahashi T, Miyauchi S, Shioi G, Inoue H, Tsujimoto G (2013) The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat Commun 4:1829
Natarajan N, Hori D, Flavahan S, Steppan J, Flavahan NA, Berkowitz DE, Pluznick JL (2016) Microbial short chain fatty acid metabolites lower blood pressure via endothelial G protein-coupled receptor 41. Physiol Genom 48:826–834
Bennett BJ, de Aguiar Vallim TQ, Wang Z, Shih DM, Meng Y, Gregory J, Allayee H, Lee R, Graham M, Crooke R, Edwards PA, Hazen SL, Lusis AJ (2013) Trimethylamine-N-oxide, a metabolite associated with atherosclerosis, exhibits complex genetic and dietary regulation. Cell Metab 17:49–60
Hoyles L, Jiménez-Pranteda ML, Chilloux J, Brial F, Myridakis A, Aranias T, Magnan C, Gibson GR, Sanderson JD, Nicholson JK, Gauguier D, McCartney AL, Dumas ME (2018) Metabolic retroconversion of trimethylamine N-oxide and the gut microbiota. Microbiome 6:73
Velasquez MT, Ramezani A, Manal A, Raj DS (2016) Trimethylamine N-oxide: the good, the bad and the unknown. Toxins (Basel) 8:326
Cho CE, Caudill MA (2017) Trimethylamine-N-oxide: friend, foe, or simply caught in the cross-fire? Trends Endocrinol Metab 28:121–130
Mueller DM, Allenspach M, Othman A, Saely CH, Muendlein A, Vonbank A, Drexel H, von Eckardstein A (2015) Plasma levels of trimethylamine-N-oxide are confounded by impaired kidney function and poor metabolic control. Atherosclerosis 243:638–644
Collins HL, Drazul-Schrader D, Sulpizio AC, Koster PD, Williamson Y, Adelman SJ, Owen K, Sanli T, Bellamine A (2016) l-Carnitine intake and high trimethylamine N-oxide plasma levels correlate with low aortic lesions in ApoE(−/−) transgenic mice expressing CETP. Atherosclerosis 244:29–37
Buchman AL, Dubin MD, Moukarzel AA, Jenden DJ, Roch M, Rice KM, Gornbein J, Ament ME (1995) Choline deficiency: a cause of hepatic steatosis during parenteral nutrition that can be reversed with intravenous choline supplementation. Hepatology 22:1399–1403
Dumas ME, Barton RH, Toye A, Cloarec O, Blancher C, Rothwell A, Fearnside J, Tatoud R, Blanc V, Lindon JC, Mitchell SC, Holmes E, McCarthy MI, Scott J, Gauguier D, Nicholson JK (2006) Metabolic profiling reveals a contribution of gut microbiota to fatty liver phenotype in insulin-resistant mice. Proc Natl Acad Sci USA 103:12511–12516
Schugar RC, Shih DM, Warrier M, Helsley RN, Burrows A, Ferguson D, Brown AL, Gromovsky AD, Heine M, Chatterjee A, Li L, Li XS, Wang Z, Willard B, Meng Y, Kim H, Che N, Pan C, Lee RG, Crooke RM, Graham MJ, Morton RE, Langefeld CD, Das SK, Rudel LL, Zein N, McCullough AJ, Dasarathy S, Tang WHW, Erokwu BO, Flask CA, Laakso M, Civelek M, Naga Prasad SV, Heeren J, Lusis AJ, Hazen SL, Brown JM (2017) The TMAO-producing enzyme flavin-containing monooxygenase 3 regulates obesity and the beiging of white adipose tissue. Cell Rep 19:2451–2461
Tang WH, Wang Z, Levison BS, Koeth RA, Britt EB, Fu X, Wu Y, Hazen SL (2013) Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N Engl J Med 368:1575–1584
Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, Feldstein AE, Britt EB, Fu X, Chung YM, Wu Y, Schauer P, Smith JD, Allayee H, Tang WH, DiDonato JA, Lusis AJ, Hazen SL (2011) Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 472:57–63
Koeth RA, Wang Z, Levison BS, Buffa JA, Org E, Sheehy BT, Britt EB, Fu X, Wu Y, Li L, Smith JD, DiDonato JA, Chen J, Li H, Wu GD, Lewis JD, Warrier M, Brown JM, Krauss RM, Tang WH, Bushman FD, Lusis AJ, Hazen SL (2013) Intestinal microbiota metabolism of l-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med 19:576–585
Zhu W, Gregory JC, Org E, Buffa JA, Gupta N, Wang Z, Li L, Fu X, Wu Y, Mehrabian M, Sartor RB, McIntyre TM, Silverstein RL, Tang WHW, DiDonato JA, Brown JM, Lusis AJ, Hazen SL (2016) Gut microbial metabolite TMAO enhances platelet hyperreactivity and thrombosis risk. Cell 165:111–124
Randrianarisoa E, Lehn-Stefan A, Wang X, Hoene M, Peter A, Heinzmann SS, Zhao X, Königsrainer I, Königsrainer A, Balletshofer B, Machann J, Schick F, Fritsche A, Häring HU, Xu G, Lehmann R, Stefan N (2016) Relationship of serum trimethylamine N-oxide (TMAO) levels with early atherosclerosis in humans. Sci Rep 6:26745
Dumas ME, Rothwell AR, Hoyles L, Aranias T, Chilloux J, Calderari S, Noll EM, Péan N, Boulangé CL, Blancher C, Barton RH, Gu Q, Fearnside JF, Deshayes C, Hue C, Scott J, Nicholson JK, Gauguier D (2017) Microbial–host co-metabolites are prodromal markers predicting phenotypic heterogeneity in behavior, obesity, and impaired glucose tolerance. Cell Rep 20:136–148
Burcelin R, Crivelli V, Dacosta A, Roy-Tirelli A, Thorens B (2002) Heterogeneous metabolic adaptation of C57BL/6J mice to high-fat diet. Am J Physiol Endocrinol Metab 282:E834–842
Koza RA, Nikonova L, Hogan J, Rim JS, Mendoza T, Faulk C, Skaf J, Kozak LP (2006) Changes in gene expression foreshadow diet-induced obesity in genetically identical mice. PLoS Genet 2:e81
Romano KA, Martinez-Del Campo A, Kasahara K, Chittim CL, Vivas EI, Amador-Noguez D, Balskus EP, Rey FE (2017) Metabolic, epigenetic, and transgenerational effects of gut bacterial choline consumption. Cell Host Microbe 22:279–290.e277
Wallrabenstein I, Kuklan J, Weber L, Zborala S, Werner M, Altmüller J, Becker C, Schmidt A, Hatt H, Hummel T, Gisselmann G (2013) Human trace amine-associated receptor TAAR5 can be activated by trimethylamine. PLoS One 8:e54950
Srinivasa S, Fitch KV, Lo J, Kadar H, Knight R, Wong K, Abbara S, Gauguier D, Capeau J, Boccara F, Grinspoon SK (2015) Plaque burden in HIV-infected patients is associated with serum intestinal microbiota-generated trimethylamine. AIDS 29:443–452
Elliott P, Posma JM, Chan Q, Garcia-Perez I, Wijeyesekera A, Bictash M, Ebbels TM, Ueshima H, Zhao L, van Horn L, Daviglus M, Stamler J, Holmes E, Nicholson JK (2015) Urinary metabolic signatures of human adiposity. Sci Transl Med 7:285ra262
Amorim Franco TM, Blanchard JS (2017) Bacterial branched-chain amino acid biosynthesis: structures, mechanisms, and drugability. Biochemistry 56:5849–5865
Pedersen HK, Gudmundsdottir V, Nielsen HB, Hyotylainen T, Nielsen T, Jensen BA, Forslund K, Hildebrand F, Prifti E, Falony G, Le Chatelier E, Levenez F, Doré J, Mattila I, Plichta DR, Pöhö P, Hellgren LI, Arumugam M, Sunagawa S, Vieira-Silva S, Jørgensen T, Holm JB, Trošt K, M. Consortium, Kristiansen K, Brix S, Raes J, Wang J, Hansen T, Bork P, Brunak S, Oresic M, Ehrlich SD, Pedersen O (2016) Human gut microbes impact host serum metabolome and insulin sensitivity. Nature 535:376–381
Floyd JC, Fajans SS, Conn JW, Knopf RF, Rull J (1966) Stimulation of insulin secretion by amino acids. J Clin Investig 45:1487–1502
Felig P, Marliss E, Cahill GF (1969) Plasma amino acid levels and insulin secretion in obesity. N Engl J Med 281:811–816
Newgard CB, An J, Bain JR, Muehlbauer MJ, Stevens RD, Lien LF, Haqq AM, Shah SH, Arlotto M, Slentz CA, Rochon J, Gallup D, Ilkayeva O, Wenner BR, Yancy WS, Eisenson H, Musante G, Surwit RS, Millington DS, Butler MD, Svetkey LP (2009) A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab 9:311–326
Tai ES, Tan ML, Stevens RD, Low YL, Muehlbauer MJ, Goh DL, Ilkayeva OR, Wenner BR, Bain JR, Lee JJ, Lim SC, Khoo CM, Shah SH, Newgard CB (2010) Insulin resistance is associated with a metabolic profile of altered protein metabolism in Chinese and Asian-Indian men. Diabetologia 53:757–767
Lee CC, Watkins SM, Lorenzo C, Wagenknecht LE, Il’yasova D, Chen YD, Haffner SM, Hanley AJ (2016) Branched-chain amino acids and insulin metabolism: the Insulin Resistance Atherosclerosis Study (IRAS). Diabetes Care 39:582–588
Katagiri R, Goto A, Budhathoki S, Yamaji T, Yamamoto H, Kato Y, Iwasaki M, Tsugane S (2018) Association between plasma concentrations of branched-chain amino acids and adipokines in Japanese adults without diabetes. Sci Rep 8:1043
Jennings A, MacGregor A, Pallister T, Spector T, Cassidy A (2016) Associations between branched chain amino acid intake and biomarkers of adiposity and cardiometabolic health independent of genetic factors: a twin study. Int J Cardiol 223:992–998
Nakamura H, Jinzu H, Nagao K, Noguchi Y, Shimba N, Miyano H, Watanabe T, Iseki K (2014) Plasma amino acid profiles are associated with insulin, C-peptide and adiponectin levels in type 2 diabetic patients. Nutr Diabetes 4:e133
Suhre K, Meisinger C, Döring A, Altmaier E, Belcredi P, Gieger C, Chang D, Milburn MV, Gall WE, Weinberger KM, Mewes HW, Hrabé de Angelis M, Wichmann HE, Kronenberg F, Adamski J, Illig T (2010) Metabolic footprint of diabetes: a multiplatform metabolomics study in an epidemiological setting. PLoS One 5:e13953
Menni C, Fauman E, Erte I, Perry JR, Kastenmüller G, Shin SY, Petersen AK, Hyde C, Psatha M, Ward KJ, Yuan W, Milburn M, Palmer CN, Frayling TM, Trimmer J, Bell JT, Gieger C, Mohney RP, Brosnan MJ, Suhre K, Soranzo N, Spector TD (2013) Biomarkers for type 2 diabetes and impaired fasting glucose using a nontargeted metabolomics approach. Diabetes 62:4270–4276
Shi L, Brunius C, Lehtonen M, Auriola S, Bergdahl IA, Rolandsson O, Hanhineva K, Landberg R (2018) Plasma metabolites associated with type 2 diabetes in a Swedish population: a case–control study nested in a prospective cohort. Diabetologia 61:849–861
Fontana L, Cummings NE, Arriola Apelo SI, Neuman JC, Kasza I, Schmidt BA, Cava E, Spelta F, Tosti V, Syed FA, Baar EL, Veronese N, Cottrell SE, Fenske RJ, Bertozzi B, Brar HK, Pietka T, Bullock AD, Figenshau RS, Andriole GL, Merrins MJ, Alexander CM, Kimple ME, Lamming DW (2016) Decreased consumption of branched-chain amino acids improves metabolic health. Cell Rep 16:520–530
Cummings NE, Williams EM, Kasza I, Konon EN, Schaid MD, Schmidt BA, Poudel C, Sherman DS, Yu D, Arriola Apelo SI, Cottrell SE, Geiger G, Barnes ME, Wisinski JA, Fenske RJ, Matkowskyj KA, Kimple ME, Alexander CM, Merrins MJ, Lamming DW (2018) Restoration of metabolic health by decreased consumption of branched-chain amino acids. J Physiol 596:623–645
Wang TJ, Larson MG, Vasan RS, Cheng S, Rhee EP, McCabe E, Lewis GD, Fox CS, Jacques PF, Fernandez C, O’Donnell CJ, Carr SA, Mootha VK, Florez JC, Souza A, Melander O, Clish CB, Gerszten RE (2011) Metabolite profiles and the risk of developing diabetes. Nat Med 17:448–453
Mahendran Y, Jonsson A, Have CT, Allin KH, Witte DR, Jørgensen ME, Grarup N, Pedersen O, Kilpeläinen TO, Hansen T (2017) Genetic evidence of a causal effect of insulin resistance on branched-chain amino acid levels. Diabetologia 60:873–878
Lotta LA, Scott RA, Sharp SJ, Burgess S, Luan J, Tillin T, Schmidt AF, Imamura F, Stewart ID, Perry JR, Marney L, Koulman A, Karoly ED, Forouhi NG, Sjögren RJ, Näslund E, Zierath JR, Krook A, Savage DB, Griffin JL, Chaturvedi N, Hingorani AD, Khaw KT, Barroso I, McCarthy MI, O’Rahilly S, Wareham NJ, Langenberg C (2016) Genetic predisposition to an impaired metabolism of the branched-chain amino acids and Risk of type 2 diabetes: a Mendelian randomisation analysis. PLoS Med 13:e1002179
Kellogg TF, Wostmann BS (1969) Fecal neutral steroids and bile acids from germfree rats. J Lipid Res 10:495–503
Miyata M, Takamatsu Y, Kuribayashi H, Yamazoe Y (2009) Administration of ampicillin elevates hepatic primary bile acid synthesis through suppression of ileal fibroblast growth factor 15 expression. J Pharmacol Exp Ther 331:1079–1085
Myronovych A, Kirby M, Ryan KK, Zhang W, Jha P, Setchell KD, Dexheimer PJ, Aronow B, Seeley RJ, Kohli R (2014) Vertical sleeve gastrectomy reduces hepatic steatosis while increasing serum bile acids in a weight-loss-independent manner. Obesity (Silver Spring) 22:390–400
Shapiro H, Kolodziejczyk AA, Halstuch D, Elinav E (2018) Bile acids in glucose metabolism in health and disease. J Exp Med 215:383–396
Molinaro A, Wahlström A, Marschall HU (2018) Role of bile acids in metabolic control. Trends Endocrinol Metab 29:31–41
Chávez-Talavera O, Tailleux A, Lefebvre P, Staels B (2017) Bile acid control of metabolism and inflammation in obesity, type 2 diabetes, dyslipidemia, and nonalcoholic fatty liver disease. Gastroenterology 152:1679–1694.e1673
Ryan KK, Tremaroli V, Clemmensen C, Kovatcheva-Datchary P, Myronovych A, Karns R, Wilson-Pérez HE, Sandoval DA, Kohli R, Bäckhed F, Seeley RJ (2014) FXR is a molecular target for the effects of vertical sleeve gastrectomy. Nature 509:183–188
Tremaroli V, Karlsson F, Werling M, Ståhlman M, Kovatcheva-Datchary P, Olbers T, Fändriks L, le Roux CW, Nielsen J, Bäckhed F (2015) Roux-en-Y gastric bypass and vertical banded gastroplasty induce long-term changes on the human gut microbiome contributing to fat mass regulation. Cell Metab 22:228–238
Albaugh VL, Banan B, Ajouz H, Abumrad NN, Flynn CR (2017) Bile acids and bariatric surgery. Mol Asp Med 56:75–89
Neuschwander-Tetri BA, Loomba R, Sanyal AJ, Lavine JE, Van Natta ML, Abdelmalek MF, Chalasani N, Dasarathy S, Diehl AM, Hameed B, Kowdley KV, McCullough A, Terrault N, Clark JM, Tonascia J, Brunt EM, Kleiner DE, Doo E, Network NCR (2015) Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): a multicentre, randomised, placebo-controlled trial. Lancet 385:956–965
Mueller M, Thorell A, Claudel T, Jha P, Koefeler H, Lackner C, Hoesel B, Fauler G, Stojakovic T, Einarsson C, Marschall HU, Trauner M (2015) Ursodeoxycholic acid exerts farnesoid X receptor-antagonistic effects on bile acid and lipid metabolism in morbid obesity. J Hepatol 62:1398–1404
Mudaliar S, Henry RR, Sanyal AJ, Morrow L, Marschall HU, Kipnes M, Adorini L, Sciacca CI, Clopton P, Castelloe E, Dillon P, Pruzanski M, Shapiro D (2013) Efficacy and safety of the farnesoid X receptor agonist obeticholic acid in patients with type 2 diabetes and nonalcoholic fatty liver disease. Gastroenterology 145:574–582.e571
Ryan PM, Stanton C, Caplice NM (2017) Bile acids at the cross-roads of gut microbiome–host cardiometabolic interactions. Diabetol Metab Syndr 9:102
Dall’Asta M, Calani L, Tedeschi M, Jechiu L, Brighenti F, Del Rio D (2012) Identification of microbial metabolites derived from in vitro fecal fermentation of different polyphenolic food sources. Nutrition 28:197–203
Vetrani C, Rivellese AA, Annuzzi G, Adiels M, Borén J, Mattila I, Orešič M, Aura AM (2016) Metabolic transformations of dietary polyphenols: comparison between in vitro colonic and hepatic models and in vivo urinary metabolites. J Nutr Biochem 33:111–118
Parkar SG, Trower TM, Stevenson DE (2013) Fecal microbial metabolism of polyphenols and its effects on human gut microbiota. Anaerobe 23:12–19
Redan BW, Buhman KK, Novotny JA, Ferruzzi MG (2016) Altered transport and metabolism of phenolic compounds in obesity and diabetes: implications for functional food development and assessment. Adv Nutr 7:1090–1104
Gryp T, Vanholder R, Vaneechoutte M, Glorieux G (2017) p-Cresyl sulfate. Toxins (Basel) 9:52
Russell WR, Duncan SH, Scobbie L, Duncan G, Cantlay L, Calder AG, Anderson SE, Flint HJ (2013) Major phenylpropanoid-derived metabolites in the human gut can arise from microbial fermentation of protein. Mol Nutr Food Res 57:523–535
Barnes KJ, Rowland A, Polasek TM, Miners JO (2014) Inhibition of human drug-metabolising cytochrome P450 and UDP-glucuronosyltransferase enzyme activities in vitro by uremic toxins. Eur J Clin Pharmacol 70:1097–1106
Tanaka S, Yano S, Sheikh AM, Nagai A, Sugimoto T (2014) Effects of uremic toxin p-cresol on proliferation, apoptosis, differentiation, and glucose uptake in 3T3-L1 cells. Artif Organs 38:566–571
Chang WC, Wu JS, Chen CW, Kuo PL, Chien HM, Wang YT, Shen SC (2015) Protective effect of vanillic acid against hyperinsulinemia, hyperglycemia and hyperlipidemia via alleviating hepatic insulin resistance and inflammation in high-fat diet (HFD)-fed rats. Nutrients 7:9946–9959
Li G, Young KD (2013) Indole production by the tryptophanase TnaA in Escherichia coli is determined by the amount of exogenous tryptophan. Microbiology 159:402–410
Barreto FC, Barreto DV, Liabeuf S, Meert N, Glorieux G, Temmar M, Choukroun G, Vanholder R, Massy ZA, E.U.T.W.G. (EUTox) (2009) Serum indoxyl sulfate is associated with vascular disease and mortality in chronic kidney disease patients. Clin J Am Soc Nephrol 4:1551–1558
Chitalia VC, Shivanna S, Martorell J, Balcells M, Bosch I, Kolandaivelu K, Edelman ER (2013) Uremic serum and solutes increase post-vascular interventional thrombotic risk through altered stability of smooth muscle cell tissue factor. Circulation 127:365–376
Shivanna S, Kolandaivelu K, Shashar M, Belghasim M, Al-Rabadi L, Balcells M, Zhang A, Weinberg J, Francis J, Pollastri MP, Edelman ER, Sherr DH, Chitalia VC (2016) The aryl hydrocarbon receptor is a critical regulator of tissue factor stability and an antithrombotic target in uremia. J Am Soc Nephrol 27:189–201
de Mello VD, Paananen J, Lindström J, Lankinen MA, Shi L, Kuusisto J, Pihlajamäki J, Auriola S, Lehtonen M, Rolandsson O, Bergdahl IA, Nordin E, Ilanne-Parikka P, Keinänen-Kiukaanniemi S, Landberg R, Eriksson JG, Tuomilehto J, Hanhineva K, Uusitupa M (2017) Indolepropionic acid and novel lipid metabolites are associated with a lower risk of type 2 diabetes in the Finnish Diabetes Prevention Study. Sci Rep 7:46337
Dumas ME, Wilder SP, Bihoreau MT, Barton RH, Fearnside JF, Argoud K, D’Amato L, Wallis RH, Blancher C, Keun HC, Baunsgaard D, Scott J, Sidelmann UG, Nicholson JK, Gauguier D (2007) Direct quantitative trait locus mapping of mammalian metabolic phenotypes in diabetic and normoglycemic rat models. Nat Genet 39:666–672
Yousri NA, Mook-Kanamori DO, Selim MM, Takiddin AH, Al-Homsi H, Al-Mahmoud KA, Karoly ED, Krumsiek J, Do KT, Neumaier U, Mook-Kanamori MJ, Rowe J, Chidiac OM, McKeon C, Al Muftah WA, Kader SA, Kastenmüller G, Suhre K (2015) A systems view of type 2 diabetes-associated metabolic perturbations in saliva, blood and urine at different timescales of glycaemic control. Diabetologia 58:1855–1867
Fiehn O, Garvey WT, Newman JW, Lok KH, Hoppel CL, Adams SH (2010) Plasma metabolomic profiles reflective of glucose homeostasis in non-diabetic and type 2 diabetic obese African–American women. PLoS One 5:e15234
Lennerz BS, Vafai SB, Delaney NF, Clish CB, Deik AA, Pierce KA, Ludwig DS, Mootha VK (2015) Effects of sodium benzoate, a widely used food preservative, on glucose homeostasis and metabolic profiles in humans. Mol Genet Metab 114:73–79
Lees HJ, Swann JR, Wilson ID, Nicholson JK, Holmes E (2013) Hippurate: the natural history of a mammalian-microbial cometabolite. J Proteome Res 12:1527–1546
Calvani R, Miccheli A, Capuani G, Tomassini Miccheli A, Puccetti C, Delfini M, Iaconelli A, Nanni G, Mingrone G (2010) Gut microbiome-derived metabolites characterize a peculiar obese urinary metabotype. Int J Obes (Lond) 34:1095–1098
Salek RM, Maguire ML, Bentley E, Rubtsov DV, Hough T, Cheeseman M, Nunez D, Sweatman BC, Haselden JN, Cox RD, Connor SC, Griffin JL (2007) A metabolomic comparison of urinary changes in type 2 diabetes in mouse, rat, and human. Physiol Genom 29:99–108
Luo P, Yu H, Zhao X, Bao Y, Hong CS, Zhang P, Tu Y, Yin P, Gao P, Wei L, Zhuang Z, Jia W, Xu G (2016) Metabolomics study of roux-en-Y gastric bypass surgery (RYGB) to treat type 2 diabetes patients based on ultraperformance liquid chromatography–mass spectrometry. J Proteome Res 15:1288–1299
Akira K, Masu S, Imachi M, Mitome H, Hashimoto M, Hashimoto T (2008) 1H NMR-based metabonomic analysis of urine from young spontaneously hypertensive rats. J Pharm Biomed Anal 46:550–556
Pallister T, Jackson MA, Martin TC, Zierer J, Jennings A, Mohney RP, MacGregor A, Steves CJ, Cassidy A, Spector TD, Menni C (2017) Hippurate as a metabolomic marker of gut microbiome diversity: modulation by diet and relationship to metabolic syndrome. Sci Rep 7:13670
De Vadder F, Kovatcheva-Datchary P, Zitoun C, Duchampt A, Bäckhed F, Mithieux G (2016) Microbiota-produced succinate improves glucose homeostasis via intestinal gluconeogenesis. Cell Metab 24:151–157
Beckonert O, Keun HC, Ebbels TM, Bundy J, Holmes E, Lindon JC, Nicholson JK (2007) Metabolic profiling, metabolomic and metabonomic procedures for NMR spectroscopy of urine, plasma, serum and tissue extracts. Nat Protoc 2:2692–2703
Want EJ, Wilson ID, Gika H, Theodoridis G, Plumb RS, Shockcor J, Holmes E, Nicholson JK (2010) Global metabolic profiling procedures for urine using UPLC–MS. Nat Protoc 5:1005–1018
Holmes E, Wilson ID, Nicholson JK (2008) Metabolic phenotyping in health and disease. Cell 134:714–717
Nicholson JK, Connelly J, Lindon JC, Holmes E (2002) Metabonomics: a platform for studying drug toxicity and gene function. Nat Rev Drug Discov 1:153–161
Clayton TA, Lindon JC, Cloarec O, Antti H, Charuel C, Hanton G, Provost JP, Le Net JL, Baker D, Walley RJ, Everett JR, Nicholson JK (2006) Pharmaco-metabonomic phenotyping and personalized drug treatment. Nature 440:1073–1077
Clayton TA, Baker D, Lindon JC, Everett JR, Nicholson JK (2009) Pharmacometabonomic identification of a significant host-microbiome metabolic interaction affecting human drug metabolism. Proc Natl Acad Sci USA 106:14728–14733
Nicholson JK, Holmes E, Wilson ID (2005) Gut microorganisms, mammalian metabolism and personalized health care. Nat Rev Microbiol 3:431–438
Suez J, Korem T, Zeevi D, Zilberman-Schapira G, Thaiss CA, Maza O, Israeli D, Zmora N, Gilad S, Weinberger A, Kuperman Y, Harmelin A, Kolodkin-Gal I, Shapiro H, Halpern Z, Segal E, Elinav E (2014) Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature 514:181–186
Chassaing B, Koren O, Goodrich JK, Poole AC, Srinivasan S, Ley RE, Gewirtz AT (2015) Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 519:92–96
Robinette SL, Holmes E, Nicholson JK, Dumas ME (2012) Genetic determinants of metabolism in health and disease: from biochemical genetics to genome-wide associations. Genome Med 4:30
Sabari BR, Zhang D, Allis CD, Zhao Y (2017) Metabolic regulation of gene expression through histone acylations. Nat Rev Mol Cell Biol 18:90–101
Goudarzi A, Zhang D, Huang H, Barral S, Kwon OK, Qi S, Tang Z, Buchou T, Vitte AL, He T, Cheng Z, Montellier E, Gaucher J, Curtet S, Debernardi A, Charbonnier G, Puthier D, Petosa C, Panne D, Rousseaux S, Roeder RG, Zhao Y, Khochbin S (2016) Dynamic competing histone H4 K5K8 acetylation and butyrylation are hallmarks of highly active gene promoters. Mol Cell 62:169–180
Acknowledgements
Work in the authors’ group is supported by the European Community’s Seventh Framework Programme under Grant agreement HEALTH-F4-2012-305312 (METACARDIS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Brial, F., Le Lay, A., Dumas, ME. et al. Implication of gut microbiota metabolites in cardiovascular and metabolic diseases. Cell. Mol. Life Sci. 75, 3977–3990 (2018). https://doi.org/10.1007/s00018-018-2901-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-018-2901-1