Abstract
The CB2 receptor is the peripheral receptor for cannabinoids. It is mainly expressed in immune tissues, highlighting the possibility that the endocannabinoid system has an immunomodulatory role. In this respect, the CB2 receptor was shown to modulate immune cell functions, both in cellulo and in animal models of inflammatory diseases. In this regard, numerous studies have reported that mice lacking the CB2 receptor have an exacerbated inflammatory phenotype. This suggests that therapeutic strategies aiming at modulating CB2 signaling could be promising for the treatment of various inflammatory conditions. Herein, we review the pharmacology of the CB2 receptor, its expression pattern, and the signaling pathways induced by its activation. We next examine the regulation of immune cell functions by the CB2 receptor and the evidence obtained from primary human cells, immortalized cell lines, and animal models of inflammation. Finally, we discuss the possible therapies targeting the CB2 receptor and the questions that remain to be addressed to determine whether this receptor could be a potential target to treat inflammatory disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The psychotropic effects induced by cannabis promoted its widespread use among the population. These effects are mediated by a cannabinoid receptor that is mainly expressed in the central nervous system, namely CB1. The identification of a receptor that is selectively activated by cannabinoids suggested that the human body synthesizes at least one natural ligand for this receptor. This hypothesis was confirmed by the discovery of two high-affinity ligands for the CB1 receptor: arachidonoyl-ethanolamide (AEA) [1] and 2-arachidonoyl-glycerol (2-AG) [2]. As these novel lipid mediators were uncovered, a second cannabinoid receptor (CB2) was being cloned and characterized. Its expression profile among tissues was found to be distinct from that of CB1. It was primarily found in immune cells and was initially not detected in the brain, although this was later proven incorrect by several studies. In light of these findings, the CB2 receptor was postulated to be responsible for the immunomodulatory effects of cannabinoids and endocannabinoids. In the past two decades, this hypothesis was tested in a wide array of cellular and animal models. This article offers a comprehensive review of the evidence that was gathered in these studies, with a focus on peripheral inflammation. The CB2 receptor’s potential as a therapeutic target in inflammatory disease is also discussed.
Cloning of the CB2 receptor
The non-psychoactive effects of cannabinoids were initially believed to be mediated either centrally or through their interaction with non-receptor proteins. Although there are phytocannabinoids that exert non-psychoactive effects without binding to CB2 receptor [e.g., cannabidiol (CBD), cannabigerol (CBG)], discovering the latter explained many of the peripheral effects of cannabinoids. Munro et al. cloned the human CB2 receptor in 1993 from the promyelocytic leukaemic cell line HL-60 [3]. To achieve this, cells were treated with dimethylformamide to induce granulocyte differentiation, a cDNA library was prepared, polymerase chain reaction (PCR) was performed using degenerated primers, and the amplification products were cloned and sequenced. One of the clones showed homology to the G-protein-coupled-receptor (GPCR) family and was related to the CB1 receptor. The protein encoded by this sequence was found to have 44 % homology with the CB1 receptor. This homology increased to 68 % when only the transmembrane portion was considered. Binding assays showed that this receptor had high affinity for the cannabinoid receptor ligands WIN 55,212-2 and CP 55,940, as well as the endocannabinoid AEA and the phytocannabinoid ∆9-THC. The authors suggested that the previously described central receptor be named CB1 and that this novel, peripheral receptor be named CB2.
A few years later, Shire et al. [4] cloned the murine CB2 receptor from a mouse splenocyte cDNA library. They found it to be 82 % homologous to the human CB2 receptor and to have similar affinity for the ligands AEA, CP 55,940, and ∆9-THC. WIN 55,212-2, however, bound the mouse CB2 receptor with an affinity six-fold lower than that documented for human CB2. This was followed by the cloning of the rat CB2 receptor by Brown et al. [5]. The authors also compared the sequence of their clone with those of the mouse and human CB2 receptor and found significant differences in protein length, although these were mainly the consequence of disparities in carboxyl termini. Amino acid conservation was highest in the transmembrane regions of the three receptors.
In addition to binding the endocannabinoids AEA and 2-AG, the CB2 receptor binds many phytocannabinoids. The pharmacology of endocannabinoids and that of the CB2 receptor were rigorously reviewed in the past [6, 7]. Table 1 provides a summary of the various endocannabinoids and phytocannabinoids and their affinity for the human CB2 receptor.
Available tools to study CB2 receptor functions
Pharmacological compounds
Synthetic cannabinoids, such as CP 55,940 and WIN 55,212-2, were already available when the CB2 receptor was cloned. They were subsequently shown to be potent CB2 ligands, but also to lack selectivity, as they activate CB1 with comparable efficiency. In this respect, several agonists and antagonists were rapidly developed and made available to the scientific community. The most widely used compounds are the agonist JWH 133, and the antagonists SR144528 and AM630. Still, many compounds display good potency and selectivity towards CB2. Table 2 contains a comprehensive list of those compounds, as well as their binding potency towards human CB2, and in some cases, the other receptors they target.
Knockout mice
The first CB2 receptor-deficient mouse was generated by Buckley et al. in 2000 [32]. The CNR2 gene was inactivated by homologous recombination, by replacing a 341 bp fragment of its coding sequence with the neomycin gene. This mutation eliminated part of intracellular loop 3, transmembrane domains 6 and 7, and the carboxyl extremity of the receptor. Autoradiography experiments confirmed the absence of specific binding of [3H]CP 55,940 in the spleen of CB −/− 2 mice. No significant difference in the binding of [3H]CP 55,940 between wild-type and knockout animals was found in the brain, supporting that CB1-receptor expression was not altered in CB2 −/− animals. The authors confirmed this by demonstrating that knockout mice were as responsive to the psychotropic effects of Δ9-THC as wild-type animals.
CB −/− 2 mice display no morphological differences when compared to their wild-type counterparts. They are normal size and weight, are fertile, have normal litter sizes and care for their young. However, subsequent studies by other groups show that CB −/− 2 mice develop differences at the cellular level. In this regard, Ofeck et al. have demonstrated that CB −/− 2 mice have lower counts of osteoblast precursors and increased numbers and activity of osteoclasts [33]. In consequence, these mice have a low bone mass phenotype that worsens with age. They also present abnormalities in the development of several T and B cell subsets [34]. While this might impair immune homeostasis, CB −/− 2 mice fail to spontaneously develop any observable immune disease. Therefore, they are suitable to study CB2 function and have, since, become invaluable tools in cannabinoid research. In this respect, they have been used to define the impact of CB2 deficiency in a variety of inflammatory disease models, and the results of these studies will be discussed in the section entitled CB2 activation by endocannabinoids in vivo
Antibodies
As it is the case with numerous GPCRs, CB2 protein detection is difficult due to the lack of specificity of primary antibodies. This concept was underscored in a recent study by Marchalant et al. [35], who showed that a commercially available and widely used CB2 polyclonal antibody is heavily cross-reactive towards other proteins. Noteworthy, they demonstrated that some of the proteins detected by the antibody were not membrane-bound, ruling out the previously suggested hypothesis that the additional bands represent glycosylation variants of the CB2 receptor. Moreover, Graham et al. [36] compared several CB2 primary antibodies in flow cytometry experiments on human primary leukocytes. The antibodies which they compared generated different expression patterns between cell types. Therefore, data regarding CB2 protein detection must be interpreted with caution.
The detection of the CB2 receptor using antibodies can be substituted, to some extent, by the alternate methods. For example, Schmöle et al. [37], recently, generated a bacterial artificial chromosome (BAC) transgenic mouse model that expresses a green fluorescent protein (GFP) under the CB2 promoter. This mouse can be used to determine CB2 expression in mouse tissues in vitro and in situ, by several techniques, including RT-PCR, qPCR, immunoblot, flow cytometry, and immunofluorescence. This system, based on GFP detection, is an alternative to the use of CB2 antibodies on mouse tissues. It is more reliable in the sense that most antibodies directed against GFP are specific and yield reproducible data. However, this kind of approach cannot be used for CB2 detection in human primary cells and tissues, which remain problematic. A different strategy that was evaluated by Petrov et al. involves the synthesis of fluorescent CB2 agonists [38]. The synthesized compound showed marked selectivity for CB2 over the CB1, 5-HT2A, and 5-HT2C receptors. This agonist was validated as a flow cytometry probe to detect the CB2 receptor in cells, and also to evaluate CB2-receptor binding using fluorescence microscopy. Other methods of detection could also be added to CB2 ligands to use them as probes, such as biotinylation [39].
CB2 expression profiles in human and animal tissues
Expression profile of CB2 among tissues
Upon cloning the human CB2 receptor from HL-60 cells, Munro et al. isolated a portion of a rat homologue by PCR [3]. They used this homologue to probe various rat tissues and detected high CB2 receptor mRNA levels in the spleen, but not in the liver, nasal epithelium, thymus, brain, lung, or kidney. Cell sorting allowed the authors to associate CB2 receptor expression to the monocyte/macrophage population of the spleen rather than T cells. Two years later, Galiègue et al. published the first study describing CB2 receptor expression in various human tissues and isolated leukocyte populations [40]. The authors found high CB2 mRNA levels in tonsils, spleen, PBMC, and thymus, and were able to detect the CB2 protein in tonsils by immunohistochemistry using an anti-CB2 polyclonal antibody. They also evaluated CB2 receptor mRNA expression in numerous human organs and found it to be absent from most non-immune tissues, with the exception of pancreas, lung, and uterus, which had relatively low mRNA levels. Several reports have, since, shown that the CB2 receptor is expressed in both male [41] and female [42, 43] reproductive tissues. In this regard, the CB2 receptor exerts an important role in the fertility of both sexes, which has already been extensively reviewed [44–47].
The pattern of CB2 receptor expression among human tissues is consistent between studies. More groups have reported the presence of the CB2 receptor mRNA and protein in the human spleen [48] and tonsils [49]. Moreover, the high level of CB2 expression in human immune tissues was also reported in murine and rodent spleen [37, 50–56] and thymus [37, 54].
The presence and role of the CB2 receptor in the central nervous system have yet to be fully elucidated, and the issue was discussed in a review article recently published by Atwood and Mackie [57]. It was initially believed that it was not expressed in non-immune cells of the central nervous system, because Munro et al. did not detect CB2 receptor mRNA in any brain part when they cloned the receptor [3], which is supported by many studies [40, 54, 58, 59]. However, we now know that the CB2 receptor is not completely absent from the brain, since it is expressed in microglia [60]. Still, the concept of the CB2 receptor being a second central cannabinoid receptor is up to debate for three main reasons: (1) a study showed that the CB2 receptor agonists JWH-015 and JWH-133 modulate peripheral neuron functions [61] and (2) the CB2 receptor was detected in the uninjured brain by immunochemistry on numerous occasions [62–64], and (3) a recent study found that hippocampal principal neurons express CB2 mRNA, and that CB2-selective agonist HU-308 modulated the activity of these cells [65]. Conversely, a study that relied on GFP detection to determine the expression of the CB2 receptor in the murine brain showed that the signal is located in microglia [37]. Therefore, the lack of reliability of the antibodies that were used in immunochemistry experiments stresses the need for more research to expand our knowledge on the involvement of the CB2 receptor in the central nervous system and neuroinflammation.
In 2009, Liu et al. showed that two distinct isoforms of the CB2 receptor exist [66]. The novel CB2 isoform was a splicing variant of the earlier cloned receptor, and was identified from a human neuroblastoma cDNA library. Splicing variants were also discovered in mice and rats, although their genomic structures and transcripts were different from those found in humans. Furthermore, the two human variants were found to display tissue-specific expression patterns. While the classical CB2 isoform was predominantly found in spleen and other immune tissues, the novel isoform was detected in higher levels in testis and brain regions of the reward system. The identification of this new CB2 variant could shed some light on the confusing expression patterns that were previously reported. Finally, it underscores the possibility of a role for CB2 in reproductive and central nervous systems that are distinct from the immunomodulatory role of the classical CB2 isoform.
CB2 expression in immune cells
It is well known that the CB2 receptor is widespread among cells of the immune system. Table 3 provides the literature associated with the expression of the CB2 receptor in human leukocytes. Every cell type that has been investigated was found to express both mRNA and protein in at least one report. However, there is conflicting data associated with a few cell types. For example, there is no consensus in the literature regarding the presence of the CB2 receptor in human neutrophils. Of note, not every study was conducted on purified, eosinophil-depleted neutrophils. Given that eosinophils have very abundant amounts of CB2 receptor mRNA, a small number of eosinophils among the neutrophil sample could result in a false positive. This is consistent with the observation that CB2 levels are lower in neutrophils than in eosinophils.
As discussed in the previous section, the scientific community should always be critical when interpreting protein data, especially of GPCRs. A large number of researchers have now reported expression data obtained with commercially available antibodies, and most of them relied on a positive control to validate their results. It was later underscored that in the case of the CB2 receptor, a reliable negative control is absolutely necessary to confirm that the signal is not generated by non-specific binding of the antibody [35, 67].
CB2 receptor signaling
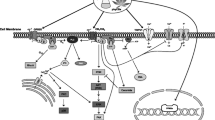
The CB2 receptor was associated to the GPCR family when it was cloned. However, the signal transduction pathways induced by CB2 receptor activation are far less characterized than those of CB1. CB1 is known to inhibit adenylyl cyclase, to modulate ion channels, and to activate numerous downstream signaling events, including p38 and p42/44 MAPK (ERK-1/2), PI3K, calcium mobilization (phospholipase C/IP3), the arachidonic acid cascade, and nitric oxide production (reviewed in [83]). A few studies have aimed to compare the signaling events of CB1 and CB2 in a given cell system and found some divergences between the two receptors. This section recapitulates the evidence regarding the signaling events downstream of the CB2 receptor.
Gi/o protein coupling and adenylyl cyclase inhibition
Like the CB1, the CB2 receptor couples with Gi/o proteins. This was established by Slipetz et al. who found that in CB2-transfected Chinese Hamster Ovary (CHO) cells, pretreatment with pertussis toxin (PTX) abolished the effect of cannabinoids on forskolin-induced cAMP production [84]. Other groups using CB2-transfected cell models found signaling events to be PTX-sensitive, supporting the involvement of Gi/o proteins [85, 86]. This interaction was later confirmed in murine microglial cells [87], the murine macrophage cell line J774-1 [88], the human promyelocytic cell line HL-60 [89–91], and human bronchial epithelial cells [92]. Since it has proven to couple to Gi/o proteins, the impact of CB2 activation on adenylyl cyclase activity was also investigated. As expected, adenylyl cyclase was inhibited upon treatment of cells with CB2 receptor agonists and/or synthetic cannabinoids, resulting in a decrease in intracellular cAMP levels [84, 85, 93, 94].
Potassium channels
As opposed to the CB1 receptor, the CB2 receptor does not appear to couple to potassium channels. A study by Felder et al. [9] investigated the possible modulation of inwardly rectifying potassium current (K ir) channels in CB2-transfected AtT-20 cells. In these cells, activation of the CB2 receptor with WIN 55,212-2 failed to have an impact on K ir. Another study showed that in Xenopus laevis oocytes co-expressing the CB2 receptor and G-protein-gated inwardly rectifying potassium (GIRK) channels, WIN 55,212-2 failed to induce consistent coupling of the CB2 receptor to GIRK channels [95]. Of note, the CB1 receptor was able to couple with GIRK channels and to modulate agonist-induced currents in the same cellular model. This important difference between CB1 and CB2 receptors established CB2 as a functionally distinct receptor.
Mitogen-activated protein kinases (MAPK)
Signal transduction pathways induced by CB2 receptor activation were first investigated in CB2-CHO cells by Bouaboula et al. [86]. They found that upon CP 55,940 addition, adenylyl cyclase inhibition was followed by ERK-1/2 phosphorylation. This effect was significantly diminished by the protein kinase C (PKC) inhibitor GF 109203X, suggesting that PKC was involved in MAPK activation. Moreover, they were able to confirm their findings in HL-60 cells, which express the CB2 receptor. Another group investigated MAPK activation by various CB2 ligands in HL-60 cells and found that CP 55,940, 2-AG, and AEA increased ERK-1/2 phosphorylation [89]. This effect was blocked by the CB2 receptor antagonist SR144528 and was stronger in cells stimulated by 2-AG and CP 55,940 than in those treated with AEA. MAPK activation downstream of CB2 activation was also demonstrated in vitro in murine osteoblasts [96], in DAUDI leukemia cells [94], murine microglia [97], and human primary monocytes [78]. Finally, this pathway was showed to be activated in vivo, in a mouse model of acute experimental pancreatitis. In this model, a CB2 receptor agonist reduced inflammation through the p38-MK2 pathway [98].
Intracellular calcium concentrations and phospholipase C activity
A study conducted in calf pulmonary endothelial cells showed that CB2 activation modulates intracellular calcium concentrations [99]. In this model, AEA initiated phospholipase C (PLC) activation and inositol 1,4,5-triphosphate (IP3) production, which led to intracellular Ca2+ release from the endoplasmic reticulum, as well as an increase in mitochondrial Ca2+. This effect of AEA was not mimicked by arachidonic acid (AA), was blocked by SR144528, and was unchanged by treatment with SR141716A, confirming the involvement of the CB2, but not the CB1 receptor. Another group later confirmed this in HEK-293 cells co-expressing the CB2 receptor with chimeric Gi and Go proteins [100]. In this model, treatment with CP 55,940 or other CB receptor agonists was found to increase intracellular Ca2+ levels. The phospholipase C inhibitor U73122 abrogated the effect of CP 55,940 on calcium mobilization, as did thapsigargin. This evidence shows that in these cells, CB2 receptor activation induces calcium mobilization via the PLC-IP3 signaling pathway.
In vitro studies of CB2 receptor functions
CB2 activation by endocannabinoids in vitro
The endocannabinoids 2-AG and AEA both act on various immune cell types through CB2 receptor activation (summarized in Table 4). Interestingly, there is a sharp contrast between the anti-inflammatory effects that are triggered by the two lipids. 2-AG was most often found to modulate functions related to leukocyte recruitment, such as chemokine release, adhesion to fibronectin, and migration. This positive regulation of immune cell recruitment by 2-AG is the main pro-inflammatory effect of endocannabinoids or cannabinoids in vitro that has been reported. AEA, on the other hand, was found to downregulate leukocyte functions, such as pro-inflammatory cytokine release and nitric oxide production. A few reports also show increased production of the anti-inflammatory cytokine IL-10 by cells treated with AEA. In all cases, the involvement of the CB2 receptor was confirmed by the use of a selective antagonist. However, it is still possible that endocannabinoid metabolites are involved in the reported effects. Noteworthy, this hypothesis was tested in human eosinophils which were shown to migrate in response to 2-AG [101]. In this model, the effect of 2-AG on eosinophil transmigration was blocked by the pre-incubation of cells with a CB2 receptor antagonist. However, a CB2-selective agonist failed to mimic the impact of 2-AG, and its 15-LO-derived metabolites were suggested to be necessary for eosinophils to migrate. Therefore, the successful blockade of endocannabinoid-induced effects with a CB2 antagonist does not always rule out the possibility that other mediators, notably endocannabinoid metabolites, are involved as well [102]. This concept could explain why endocannabinoids can induce both pro- and anti-inflammatory effects.
CB2 activation by exogenous agonists in vitro
In contrast to endocannabinoids, CB2 receptor agonists have only been shown to exert anti-inflammatory effects on leukocytes, which are detailed in Table 5. Some of the studies were performed using a non-selective cannabinoid, but the involvement of the CB2 receptor was always confirmed with an antagonist. In addition to downregulating leukocyte functions, such as cytokine release, reactive oxygen species production and migration, CB2 agonists limited HIV-1 expression, and replication in human macrophages and microglia [75, 125].
In vivo studies of CB2 receptor functions
Impact of CB2 knockout in inflammation models
Transgenic mice have greatly contributed to our understanding of this receptor’s role in human disease, including inflammatory conditions. In this regard, several models have shown that mice that are lacking the CB2 receptor have exacerbated inflammation (summarized in Table 6). The effects that were usually observed in CB −/− 2 animals included increased leukocyte recruitment (often neutrophils) and pro-inflammatory cytokine production, which often caused tissue damage. Conversely, one study found CB2-deficient mice to be in better condition than the wild-type group [142]. However, the model was cecal ligation-induced sepsis, a condition in which efficient bacterial clearance by the immune system is vital. The authors’ observations that the CB −/− 2 group had less mortality and less bacterial invasion was explained by the lower levels of IL-10 in these mice, which might have led to a better phagocytic response. Overall, these findings are consistent with the other reports of increased immune cell functions in the absence of the CB2 receptor.
CB2 activation by exogenous agonists in vivo
The potential of activating CB2 in vivo to treat inflammation has been investigated in numerous studies. Two main strategies are employed: (1) the administration of a CB2 receptor agonist; and (2) the administration of an endocannabinoid hydrolysis inhibitor to augment endocannabinoid signaling.
The administration of CB2 receptor agonists has been performed in several inflammation models. Table 7 summarizes the data that were generated with this approach. In many instances, the chosen agonist was not CB2-selective and targeted both cannabinoid receptors, in which case, the involvement of CB2 was confirmed by showing that the treatment of animals with a CB2 antagonist abrogated the effects of the cannabinoid receptor agonist. Altogether, the results of those studies point to the conclusion that CB2 activation improves inflammation in mice. The recruitment of leukocytes to tissues and the production of pro-inflammatory cytokines and reactive oxygen species were downregulated in various inflammation models. In the case of atherosclerosis, two studies showed not only a decrease in inflammatory cells and mediators upon cannabinoid treatment, but also a slower progression of the disease [148, 149]. Indeed, oral ∆9-THC administration, at doses that are suboptimal for inducing psychotropic effects, resulted in reduced atherosclerotic lesion development. Since these effects of ∆9-THC were shown to be mediated by the CB2 receptor, this supports that a selective CB2 receptor agonist might be a valuable tool for the treatment of atherosclerosis.
CB2 activation by endocannabinoids in vivo
The most widely used approach to investigate the impact of endocannabinoids in vivo is the blockade of their hydrolysis, as it is an efficient way to increase their levels in tissues. Despite the numerous studies that have used this method in animal models, it is still unclear whether the effects of endocannabinoids are pro- or anti-inflammatory. This is due, in part, to the presence of numerous enzymes that can metabolize them into other bioactive lipids. The main pathway is hydrolysis into AA by lipases, such as MAG lipase for 2-AG [164] and FAAH for AEA [165]. AA is a precursor for the biosynthesis of leukotrienes, prostaglandins, and other lipid mediators of inflammation. Alternatively, endocannabinoids can undergo oxidation and the biological effects of the metabolites that originate from these pathways are not very well characterized [166]. Therefore, it is not possible to conclude that endocannabinoids exert their effects through CB2 in an inflammation model unless this is confirmed by the genetic or pharmacological blockade of the receptor. In this respect, Table 8 only presents studies that have thoroughly confirmed the involvement of the CB2 receptor in the effects they observed.
A limited number of studies reported pro-inflammatory effects of endocannabinoids in vivo, and only three of those (listed in Table 9) were confirmed to involve the CB2 receptor. In two models of dermatitis in mice, treatment with the CB2 antagonist SR144528 improved inflammation by inhibiting granulocyte recruitment and pro-inflammatory mediator production [174, 175]. In both cases, this translated in a measurable decrease in swelling. As presented above in Table 6, 2-AG has been implicated in the recruitment and migration of B and T cells, dendritic cells, eosinophils, monocytes, and natural killer cells in a CB2-dependent manner, which could very well translate to in vivo studies. However, to this day, there is no published data demonstrating that exogenous cannabinoids and selective CB2 receptor agonists have pro-inflammatory effects. Therefore, it is possible that the pro-inflammatory effects of endocannabinoids that are presented in Table 9 are a result of CB2 activation and/or the action of one or more endocannabinoid metabolites [102].
Of note, many disorders cause a change in CB2 receptor protein levels, due to pre-existing pro-inflammatory conditions. In multiple sclerosis and amyotrophic lateral sclerosis, for instance, the expression of CB2 in microglia is increased, both in human tissues and mouse models [176, 177]. A similar effect was reported in a rodent model of neuropathic pain [178]. This certainly facilitates the impact of CB2 receptor activation by exogenous agonists of endocannabinoids in these inflammation models.
The CB2 receptor as a potential therapeutic target
While there is a large body of evidence supporting that CB2 receptor activation has anti-inflammatory effects, it has yet to be targeted to treat human disease. In the two previous sections, we presented in vitro and in vivo studies that suggested a role for the CB2 receptor in numerous inflammatory conditions. In this section, we discuss the potential of the CB2 receptor as a target in the treatment of chronic inflammatory diseases, such as rheumatoid arthritis, atherosclerosis, and inflammatory bowel disease.
Potential in rheumatoid arthritis
Rheumatoid arthritis (RA) is an inflammatory disease that affects approximately 1 % of the adult population worldwide. RA is characterized by chronic inflammation of the synovium, cartilage destruction, and bone loss. Patients with RA exhibit an influx of innate (neutrophils, macrophages) and adaptive (lymphocytes) immune cells in the synovial cavity. These cells promote inflammation and connective tissue damage by producing cytokines (TNF-α, IL-6, IL-1β), pro-inflammatory lipids, and metalloproteinases (MMPs). The synovial lining becomes hyperplastic and an invasive structure (the pannus) is formed. Osteoclasts become exaggeratedly activated and cause bone resorption [180].
2-AG and AEA are present in the synovial fluid of patients with RA, but not healthy volunteers, suggesting an involvement of the endocannabinoid system in the disease. CB1 and CB2 mRNA and proteins were also found in the synovial tissues of RA patients [181]. CB2 activation can inhibit the production of pro-inflammatory cytokines and MMP release from fibroblast-like synoviocytes (FLSs) [182, 183]. It can also promote osteoblast differentiation in vitro [33, 184] and inhibit FLS proliferation [182]. These observations indicate that CB2 receptor activation in RA joints could improve multiple aspects of the disease, including inflammation, FLS hyperplasia, and bone loss.
In vivo, CB2 agonists have proven to be beneficial in a murine model of rheumatoid arthritis, collagen-induced arthritis (CIA). One study showed treatment with the CB2 receptor agonist JWH 133 to improve arthritis severity and to reduce bone destruction and leukocyte infiltration in the joints [183]. Another group investigated the impact of a different CB2-selective agonist, HU-308. They found that the agonist decreased swelling, synovial inflammation, and joint destruction, in addition to lowering circulating antibodies against collagen II [185]. Finally, the agonist HU-320 ameliorated established CIA [186]. Of note, CB2 agonists did not prevent the onset of RA in any of those reports, as there were no differences in disease incidence between groups.
This growing body of evidence establishes the CB2 receptor as a promising target for the treatment of RA. In all three of the above-mentioned studies, the CIA model was used to test CB2 agonists. Given that there is no animal model of RA that perfectly duplicates all aspects the human condition, these findings should be confirmed in different models.
Potential in atherosclerosis
Atherosclerosis is an inflammatory disease that is characterized by the presence of arterial plaques. These lesions contain immune cells, lipid-laden macrophages (foam cells), cholesterol, smooth muscle cells, and collagen fibres [187]. The physical rupture of the plaques causes the occlusion of arteries, which can lead to tissue infarction. Plaque development is influenced by inflammatory mediators, such as cytokines and chemokines, which are crucial to the recruitment of immune cells to the intima. In this respect, therapies that would downregulate the production of these mediators could reduce the progression of atherosclerotic lesion development. Since the CB2 receptor is known to decrease the production of numerous chemokines and to inhibit leukocyte migration in vitro and in vivo, it emerged as a potential target to treat atherosclerosis.
A recent study specifically aimed to characterize the endocannabinoid system in human foam cells [188]. The authors found that the CB2 agonist JHW-015 significantly decreased oxLDL accumulation in these macrophages. Moreover, it reduced the production of TNF-α, IL-6, and IL-10 and the expression of CD36, a scavenger receptor that is responsible for the uptake of modified lipoproteins by macrophages and the induction of foam cell formation. The endocannabinoids 2-AG and AEA mimicked these effects, which were block by the CB2 antagonist SR144528. These findings are in accordance with a previous study which showed that CB2 activation by WIN 55,212-2 reduces the oxLDL-induced inflammatory response in rat macrophages [131].
As briefly discussed in the section entitled In vivo studies of CB2 receptor functions, the role of the CB2 receptor was investigated in mouse models of atherosclerosis. The first study to demonstrate the benefits of CB2 activation in atherosclerosis was performed in ApoE −/− mice using low doses of the cannabinoid ∆9-THC, which diminished inflammation and blocked the progression of the disease [149]. These effects were prevented by SR144528, confirming the involvement of the CB2 receptor. The anti-atherosclerotic effects of CB2 in the ApoE −/− model were later confirmed with WIN 55,212-2 as an agonist, and the antagonist AM630 confirmed the mechanism to be CB2-dependent [148, 189]. In Ldlr −/− CB −/− 2 double knockout mice, lesional macrophage and smooth muscle cell contents were higher than in Ldlr −/− CB +/+ 2 animals [190]. In Ldlr −/− mice deficient for CB2 in hematopoietic cells only, plaque area after 12 weeks on an atherogenic diet was larger than in mice with no CB2 deficiency [191].
In summary, a large body of evidence strongly suggests that CB2 receptor activation is an appropriate target for atherosclerosis treatment. CB2 agonists have the potential to be beneficial on many levels, as they were shown to improve inflammatory cell recruitment and activation, lipid uptake by macrophages, and the size of atherosclerotic plaques. However, a few reports show conflicting data, especially in the Ldlr −/− model. A report shows unaltered lesion size following WIN 55,212-2 treatment in this model, although CB2 receptor activation did decrease lesional macrophage accumulation [192]. Another group treated Ldlr −/− mice with JWH-133 and found no significant effect on lesion size or on their content in macrophages, lipids, smooth muscle cells, collagen, and T cells [193]. More investigation is required to determine the causes of these discrepancies before moving forward in the development of therapies targeting CB2 for atherosclerosis.
Potential in inflammatory bowel disease
Inflammatory bowel disease (IBD) includes two main conditions: ulcerative colitis and Crohn’s disease. They are caused by an excessive immune response and can affect any part of the gastrointestinal tract [194]. The endocannabinoid system first gained interest in IBD pathophysiology in light of a study that described a protective effect of CB1 in DNBS-induced colitis [195]. Cannabinoids were then shown to enhance epithelial wound healing in a CB1-dependent fashion [76]. The authors of the latter study also evaluated the expression of cannabinoid receptors in human IBD tissue by immunochemistry. They found that the CB1 receptor was expressed in the normal human colon, but that CB2 expression was higher in IBD tissues and that its presence was concentrated in plasma cells and macrophages. These findings raised the hypothesis that the CB2 receptor was also involved in the inflammatory component of IBD.
A subsequent study reported that a FAAH inhibitor decreased inflammation in the TNBS-induced colitis model, and that the deletion of either CB1 or CB2 abrogated this effect [196]. In the same colitis model, the use of the MAG lipase inhibitor JZL184 to increase 2-AG levels also inhibited the development of colitis [173]. Mice treated with JZL184 had less colon alteration and lower expression of pro-inflammatory cytokines, and these effects were abolished by the antagonists AM251 (CB1) and AM630 (CB2).
Several groups tested the impact of a CB2 receptor agonist in the IBD models. The CB2-selective agonists JWH-133 and AM1241 both protected against TNBS-induced colitis, whereas AM630 worsened it [197]. The non-psychotropic cannabinoid cannabigerol (CBG) was tested in DNBS-induced colitis and was found to reduce the colon weight/colon length ratio (an indirect marker of inflammation), MPO activity, and iNOS expression by a CB2-dependent mechanism [198]. Finally, the plant metabolite and unconventional CB2 agonist (E)-β-caryophyllene (BCP) was also evaluated in a model of DSS-induced colitis. Oral administration of BCP decreased micro- and macro-scopic colon damage, MPO activity, NF-κB activation, and pro-inflammatory cytokine production [199].
This wide array of CB2 receptor agonists being able to improve IBD in animal models prompted the development of highly selective compounds that could be used to treat the disease in humans. In this regard, a research group synthesized a series of CB2-selective agonists and tested the resulting lead compounds in models of experimental colitis [200, 201]. Intra-peritoneal injection of the agonists was effective at protecting mice against colitis. Of note, a selective compound that is orally effective in experimental colitis was later synthesized [202].
Conclusion
In light of the evidence that was generated over the past two decades by the scientific community, we can draw a few general conclusions regarding the role of the CB2 receptor. First, it is mainly found in immune tissues and is expressed in most immune cell types. Second, its deletion in animals usually causes an exacerbated inflammatory phenotype in several models, due to an upregulation of immune cell functions. Third, CB2 activation by cannabinoids, either in vitro or in vivo, usually decreases inflammatory cell activation. Finally, the administration of CB2 agonists in animal models of inflammatory disease can slow the progression of some diseases, in addition to reducing inflammation.
Several questions still need to be investigated. For example, there is no consensus regarding the expression of the CB2 receptor in non-immune brain cells, and the role that CB2 might play in brain functions is unknown. Moreover, the impact of endocannabinoids on immune cells is still unclear. While most animal studies show that the blockade of endocannabinoid hydrolysis results in less inflammation, it is not possible to tell whether these effects are caused only by CB2 activation and whether the opposite would occur in humans. In this respect, endocannabinoids can induce human leukocyte migration (Table 4). However, the impact of endocannabinoid metabolites on leukocyte functions is not well defined, and this should be addressed before endocannabinoid hydrolysis inhibitors that can be considered as a valid strategy to enhance CB2 receptor signaling [102]. Finally, the few CB2 agonists that are currently being developed aim at treating inflammatory pain [203–205]. Perhaps, these novel compounds are worthy of sparking new studies to define their putative beneficial role in inflammatory diseases.
Abbreviations
- 2-AG:
-
2-Arachidonoyl-glycerol
- AA:
-
Arachidonic acid
- AEA:
-
N-Arachidonoyl-ethanolamide
- AM1241:
-
(2-Iodo-5-nitrophenyl)-(1-(1-methylpiperidin-2-ylmethyl)-1H-indol-3-yl)methanone
- AM630:
-
6-Iodo-2-methyl-1-[2-(4-morpholinyl)ethyl]-1H-indol-3-yl](4-methoxyphenyl)methanone
- CB65:
-
N-Cyclohexyl-7-chloro-1-[2-(4-morpholinyl)ethyl]quinolin-4(1H)-one-3-carboxamide
- cAMP:
-
Cyclic adenosine monophosphate
- CBD:
-
Cannabidiol
- CBG:
-
Cannabigerol
- CBN:
-
Cannabinol
- COX:
-
Cyclooxygenase
- CP 55,940:
-
(–)-Cis-3-[2-hydroxy-4-(1,1-dimethylheptyl)phenyl]-trans-4-(3-hydroxypropyl)cyclohexanol
- Δ9-THC:
-
(–)-Δ9-Tetrahydrocannabinol
- ERK-1/2:
-
Extracellular signal-regulated kinases-1/2
- FAAH:
-
Fatty acid amide hydrolase
- GFP:
-
Green fluorescent protein
- GIRK:
-
G-protein-coupled inwardly rectifying potassium (channel)
- GP 1a:
-
N-(Piperidin-1-yl)-1-(2,4-dichlorophenyl)-1,4-dihydro-6-methylindeno[1,2-c]pyrazole-3-carboxamide
- GP 2a:
-
N-Cyclohexyl-1-(2,4-dichlorophenyl)-1,4-dihydro-6-methylindeno[1,2-c]pyrazole-3-carboxamide
- GPCR:
-
G-protein-coupled-receptor
- HU-210:
-
3-(1,1′-Dimethylheptyl)-6aR,7,10,10aR-tetrahydro-1-hydroxy-6,6-dimethyl-6H-dibenzo[b,d]pyran-9-methanol
- HU-308:
-
4-[4-(1,1-Dimethylheptyl)-2,6-dimethoxyphenyl]-6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-methanol
- IP3 :
-
Inositol 1,4,5-trisphosphate
- JTE 907:
-
N-(1,3-Benzodioxol-5-ylmethyl)-1,2-dihydro-7-methoxy-2-oxo-8-(pentyloxy)-3-quinolinecarboxamide
- JWH 015:
-
(2-Methyl-1-propyl-1H-indol-3-yl)-1-naphthalenyl-methanone
- JWH 133:
-
(6aR,10aR)-3-(1,1-Dimethylbutyl)-6a,7,10,10a-tetrahydro-6,6,9-trimethyl-6H-dibenzo[b,d]pyran
- L-759,633:
-
(6aR,10aR)-3-(1,1-Dimethylheptyl)-6a,7,10,10a-tetrahydro-1-methoxy-6,6,9-trimethyl-6H-dibenzo[b,d]pyran
- L-759,656:
-
(6aR,10aR)-3-(1,1-Dimethylheptyl)-6a,7,8,9,10,10a-hexahydro-1-methoxy-6,6-dimethyl-9-methylene-6H-dibenzo[b,d]pyran
- LOX:
-
Lipoxygenase
- MAG:
-
Monoacylglycerol
- MAPK:
-
Mitogen-activated protein kinases
- NADA:
-
N-Arachidonoyl-dopamine
- PI3K:
-
Phosphoinositide 3-kinase
- PKC:
-
Protein kinase C
- PLC:
-
Phospholipase C
- PTX:
-
Pertussis toxin
- SER 601:
-
N-(Adamant-1-yl)-6-isopropyl-4-oxo-1-pentyl-1,4-dihydroquinoline-3-carboxamide
- WIN 55,212-2:
-
[(3R)-2,3-Dihydro-5-methyl-3-(4-morpholinylmethyl)pyrrolo[1,2,3-de]-1,4-benzoxazin-6-yl]-1-naphthalenyl-methanone, monomethanesulfonate
- SR141716A:
-
N-(Piperidin-1-yl)-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide hydrochloride
- SR144528:
-
5-(4-Chloro-3-methylphenyl)-1-[(4-methylphenyl)methyl]-N-[(1S,2S,4R)-1,3,3-trimethylbicyclo[2.2.1]hept-2-yl]-1H-pyrazole-3-carboxamide
References
Devane WA, Hanus L, Breuer A, Pertwee RG, Stevenson LA, Griffin G, Gibson D, Mandelbaum A, Etinger A, Mechoulam R (1992) Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258(5090):1946–1949
Mechoulam R, Ben-Shabat S, Hanus L, Ligumsky M, Kaminski NE, Schatz AR, Gopher A, Almog S, Martin BR, Compton DR et al (1995) Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem Pharmacol 50(1):83–90
Munro S, Thomas KL, Abu-Shaar M (1993) Molecular characterization of a peripheral receptor for cannabinoids. Nature 365(6441):61–65. doi:10.1038/365061a0
Shire D, Calandra B, Rinaldi-Carmona M, Oustric D, Pessegue B, Bonnin-Cabanne O, Le Fur G, Caput D, Ferrara P (1996) Molecular cloning, expression and function of the murine CB2 peripheral cannabinoid receptor. Biochim Biophys Acta 1307(2):132–136
Brown SM, Wager-Miller J, Mackie K (2002) Cloning and molecular characterization of the rat CB2 cannabinoid receptor. Biochim Biophys Acta 1576(3):255–264
Pertwee RG (1997) Pharmacology of cannabinoid CB1 and CB2 receptors. Pharmacol Ther 74(2):129–180
Pertwee RG (2015) Endocannabinoids and their pharmacological actions. Handb Exp Pharmacol 231:1–37. doi:10.1007/978-3-319-20825-1_1
Showalter VM, Compton DR, Martin BR, Abood ME (1996) Evaluation of binding in a transfected cell line expressing a peripheral cannabinoid receptor (CB2): identification of cannabinoid receptor subtype selective ligands. J Pharmacol Exp Ther 278(3):989–999
Felder CC, Joyce KE, Briley EM, Mansouri J, Mackie K, Blond O, Lai Y, Ma AL, Mitchell RL (1995) Comparison of the pharmacology and signal transduction of the human cannabinoid CB1 and CB2 receptors. Mol Pharmacol 48(3):443–450
Gonsiorek W, Lunn C, Fan X, Narula S, Lundell D, Hipkin RW (2000) The Endocannabinoid 2-arachidonyl glycerol is a full agonist through human type 2 cannabinoid receptor: antagonism by anandamide. Mol Pharmacol 57(5):1045–1050
Leggett JD, Aspley S, Beckett SR, D’Antona AM, Kendall DA, Kendall DA (2004) Oleamide is a selective endogenous agonist of rat and human CB1 cannabinoid receptors. Br J Pharmacol 141(2):253–262. doi:10.1038/sj.bjp.0705607
Bisogno T, Melck D, Bobrov M, Gretskaya NM, Bezuglov VV, De Petrocellis L, Di Marzo V (2000) N-Acyl-dopamines: novel synthetic CB1 cannabinoid-receptor ligands and inhibitors of anandamide inactivation with cannabimimetic activity in vitro and in vivo. Biochem J 351(Pt 3):817–824
Hanus L, Abu-Lafi S, Fride E, Breuer A, Vogel Z, Shalev DE, Kustanovich I, Mechoulam R (2001) 2-Arachidonyl glyceryl ether, an endogenous agonist of the cannabinoid CB1 receptor. Proc Natl Acad Sci USA 98(7):3662–3665. doi:10.1073/pnas.061029898
Pertwee RG (1999) Pharmacology of cannabinoid receptor ligands. Curr Med Chem 6(8):635–664
Gertsch J, Leonti M, Raduner S, Racz I, Chen JZ, Xie XQ, Altmann KH, Karsak M, Zimmer A (2008) Beta-caryophyllene is a dietary cannabinoid. Proc Natl Acad Sci USA 105(26):9099–9104. doi:10.1073/pnas.0803601105
Ibrahim MM, Deng H, Zvonok A, Cockayne DA, Kwan J, Mata HP, Vanderah TW, Lai J, Porreca F, Makriyannis A, Malan TP Jr (2003) Activation of CB2 cannabinoid receptors by AM1241 inhibits experimental neuropathic pain: pain inhibition by receptors not present in the CNS. Proc Natl Acad Sci USA 100(18):10529–10533. doi:10.1073/pnas.1834309100
Akopian AN, Ruparel NB, Patwardhan A, Hargreaves KM (2008) Cannabinoids desensitize capsaicin and mustard oil responses in sensory neurons via TRPA1 activation. J Neurosci 28(5):1064–1075. doi:10.1523/JNEUROSCI.1565-06.2008
Huffman JW, Liddle J, Yu S, Aung MM, Abood ME, Wiley JL, Martin BR (1999) 3-(1′,1′-Dimethylbutyl)-1-deoxy-delta8-THC and related compounds: synthesis of selective ligands for the CB2 receptor. Bioorg Med Chem 7(12):2905–2914
McDougall JJ, Yu V, Thomson J (2008) In vivo effects of CB2 receptor-selective cannabinoids on the vasculature of normal and arthritic rat knee joints. Br J Pharmacol 153(2):358–366. doi:10.1038/sj.bjp.0707565
Valenzano KJ, Tafesse L, Lee G, Harrison JE, Boulet JM, Gottshall SL, Mark L, Pearson MS, Miller W, Shan S, Rabadi L, Rotshteyn Y, Chaffer SM, Turchin PI, Elsemore DA, Toth M, Koetzner L, Whiteside GT (2005) Pharmacological and pharmacokinetic characterization of the cannabinoid receptor 2 agonist, GW405833, utilizing rodent models of acute and chronic pain, anxiety, ataxia and catalepsy. Neuropharmacology 48(5):658–672. doi:10.1016/j.neuropharm.2004.12.008
Hanus L, Breuer A, Tchilibon S, Shiloah S, Goldenberg D, Horowitz M, Pertwee RG, Ross RA, Mechoulam R, Fride E (1999) HU-308: a specific agonist for CB2, a peripheral cannabinoid receptor. Proc Natl Acad Sci USA 96(25):14228–14233
Ross RA, Brockie HC, Stevenson LA, Murphy VL, Templeton F, Makriyannis A, Pertwee RG (1999) Agonist-inverse agonist characterization at CB1 and CB2 cannabinoid receptors of L759633, L759656, and AM630. Br J Pharmacol 126(3):665–672. doi:10.1038/sj.bjp.0702351
Pasquini S, Botta L, Semeraro T, Mugnaini C, Ligresti A, Palazzo E, Maione S, Di Marzo V, Corelli F (2008) Investigations on the 4-quinolone-3-carboxylic acid motif. 2. Synthesis and structure-activity relationship of potent and selective cannabinoid-2 receptor agonists endowed with analgesic activity in vivo. J Med Chem 51(16):5075–5084. doi:10.1021/jm800552f
Murineddu G, Lazzari P, Ruiu S, Sanna A, Loriga G, Manca I, Falzoi M, Dessi C, Curzu MM, Chelucci G, Pani L, Pinna GA (2006) Tricyclic pyrazoles. 4. Synthesis and biological evaluation of analogues of the robust and selective CB2 cannabinoid ligand 1-(2′,4′-dichlorophenyl)-6-methyl-N-piperidin-1-yl-1,4-dihydroindeno[1,2-c]pyrazo le-3-carboxamide. J Med Chem 49(25):7502–7512. doi:10.1021/jm060920d
Manera C, Benetti V, Castelli MP, Cavallini T, Lazzarotti S, Pibiri F, Saccomanni G, Tuccinardi T, Vannacci A, Martinelli A, Ferrarini PL (2006) Design, synthesis, and biological evaluation of new 1,8-naphthyridin-4(1H)-on-3-carboxamide and quinolin-4(1H)-on-3-carboxamide derivatives as CB2 selective agonists. J Med Chem 49(20):5947–5957. doi:10.1021/jm0603466
Ryberg E, Larsson N, Sjogren S, Hjorth S, Hermansson NO, Leonova J, Elebring T, Nilsson K, Drmota T, Greasley PJ (2007) The orphan receptor GPR55 is a novel cannabinoid receptor. Br J Pharmacol 152(7):1092–1101. doi:10.1038/sj.bjp.0707460
Cheer JF, Cadogan AK, Marsden CA, Fone KC, Kendall DA (1999) Modification of 5-HT2 receptor mediated behaviour in the rat by oleamide and the role of cannabinoid receptors. Neuropharmacology 38(4):533–541
Thomas BF, Gilliam AF, Burch DF, Roche MJ, Seltzman HH (1998) Comparative receptor binding analyses of cannabinoid agonists and antagonists. J Pharmacol Exp Ther 285(1):285–292
Rinaldi-Carmona M, Barth F, Millan J, Derocq JM, Casellas P, Congy C, Oustric D, Sarran M, Bouaboula M, Calandra B, Portier M, Shire D, Breliere JC, Le Fur GL (1998) SR 144528, the first potent and selective antagonist of the CB2 cannabinoid receptor. J Pharmacol Exp Ther 284(2):644–650
Patil M, Patwardhan A, Salas MM, Hargreaves KM, Akopian AN (2011) Cannabinoid receptor antagonists AM251 and AM630 activate TRPA1 in sensory neurons. Neuropharmacology 61(4):778–788. doi:10.1016/j.neuropharm.2011.05.024
Iwamura H, Suzuki H, Ueda Y, Kaya T, Inaba T (2001) In vitro and in vivo pharmacological characterization of JTE-907, a novel selective ligand for cannabinoid CB2 receptor. J Pharmacol Exp Ther 296(2):420–425
Buckley NE, McCoy KL, Mezey E, Bonner T, Zimmer A, Felder CC, Glass M, Zimmer A (2000) Immunomodulation by cannabinoids is absent in mice deficient for the cannabinoid CB(2) receptor. Eur J Pharmacol 396(2–3):141–149
Ofek O, Karsak M, Leclerc N, Fogel M, Frenkel B, Wright K, Tam J, Attar-Namdar M, Kram V, Shohami E, Mechoulam R, Zimmer A, Bab I (2006) Peripheral cannabinoid receptor, CB2, regulates bone mass. Proc Natl Acad Sci USA 103(3):696–701. doi:10.1073/pnas.0504187103
Ziring D, Wei B, Velazquez P, Schrage M, Buckley NE, Braun J (2006) Formation of B and T cell subsets require the cannabinoid receptor CB2. Immunogenetics 58(9):714–725. doi:10.1007/s00251-006-0138-x
Marchalant Y, Brownjohn PW, Bonnet A, Kleffmann T, Ashton JC (2014) Validating antibodies to the cannabinoid CB2 receptor: antibody sensitivity is not evidence of antibody specificity. J Histochem Cytochem 62(6):395–404. doi:10.1369/0022155414530995
Graham ES, Angel CE, Schwarcz LE, Dunbar PR, Glass M (2010) Detailed characterisation of CB2 receptor protein expression in peripheral blood immune cells from healthy human volunteers using flow cytometry. Int J Immunopathol Pharmacol 23(1):25–34
Schmole AC, Lundt R, Gennequin B, Schrage H, Beins E, Kramer A, Zimmer T, Limmer A, Zimmer A, Otte DM (2015) Expression analysis of CB2-GFP BAC transgenic mice. PLoS One 10(9):e0138986. doi:10.1371/journal.pone.0138986
Petrov RR, Ferrini ME, Jaffar Z, Thompson CM, Roberts K, Diaz P (2011) Design and evaluation of a novel fluorescent CB2 ligand as probe for receptor visualization in immune cells. Bioorg Med Chem Lett 21(19):5859–5862. doi:10.1016/j.bmcl.2011.07.099
Fezza F, Oddi S, Di Tommaso M, De Simone C, Rapino C, Pasquariello N, Dainese E, Finazzi-Agro A, Maccarrone M (2008) Characterization of biotin-anandamide, a novel tool for the visualization of anandamide accumulation. J Lipid Res 49(6):1216–1223. doi:10.1194/jlr.M700486-JLR200
Galiegue S, Mary S, Marchand J, Dussossoy D, Carriere D, Carayon P, Bouaboula M, Shire D, Le Fur G, Casellas P (1995) Expression of central and peripheral cannabinoid receptors in human immune tissues and leukocyte subpopulations. Eur J Biochem 232(1):54–61
Grimaldi P, Orlando P, Di Siena S, Lolicato F, Petrosino S, Bisogno T, Geremia R, De Petrocellis L, Di Marzo V (2009) The endocannabinoid system and pivotal role of the CB2 receptor in mouse spermatogenesis. Proc Natl Acad Sci USA 106(27):11131–11136. doi:10.1073/pnas.0812789106
El-Talatini MR, Taylor AH, Elson JC, Brown L, Davidson AC, Konje JC (2009) Localisation and function of the endocannabinoid system in the human ovary. PLoS One 4(2):e4579. doi:10.1371/journal.pone.0004579
Taylor AH, Abbas MS, Habiba MA, Konje JC (2010) Histomorphometric evaluation of cannabinoid receptor and anandamide modulating enzyme expression in the human endometrium through the menstrual cycle. Histochem Cell Biol 133(5):557–565. doi:10.1007/s00418-010-0695-9
Battista N, Meccariello R, Cobellis G, Fasano S, Di Tommaso M, Pirazzi V, Konje JC, Pierantoni R, Maccarrone M (2012) The role of endocannabinoids in gonadal function and fertility along the evolutionary axis. Mol Cell Endocrinol 355(1):1–14. doi:10.1016/j.mce.2012.01.014
Meccariello R, Battista N, Bradshaw HB, Wang H (2014) Updates in reproduction coming from the endocannabinoid system. Int J Endocrinol 2014:412354. doi:10.1155/2014/412354
Wang H, Dey SK, Maccarrone M (2006) Jekyll and hyde: two faces of cannabinoid signaling in male and female fertility. Endocr Rev 27(5):427–448. doi:10.1210/er.2006-0006
Taylor AH, Amoako AA, Bambang K, Karasu T, Gebeh A, Lam PM, Marzcylo TH, Konje JC (2010) Endocannabinoids and pregnancy. Clin Chim Acta 411(13–14):921–930. doi:10.1016/j.cca.2010.03.012
Rayman N, Lam KH, van der Holt B, Koss C, van Leeuwen J, Budel LM, Mulder AH, Sonneveld P, Delwel R (2011) The expression of the peripheral cannabinoid receptor CB2 has no effect on clinical outcome in diffuse large B-cell lymphomas. Eur J Haematol 86(6):466–476. doi:10.1111/j.1600-0609.2011.01596.x
Carayon P, Marchand J, Dussossoy D, Derocq JM, Jbilo O, Bord A, Bouaboula M, Galiegue S, Mondiere P, Penarier G, Fur GL, Defrance T, Casellas P (1998) Modulation and functional involvement of CB2 peripheral cannabinoid receptors during B-cell differentiation. Blood 92(10):3605–3615
Carlisle SJ, Marciano-Cabral F, Staab A, Ludwick C, Cabral GA (2002) Differential expression of the CB2 cannabinoid receptor by rodent macrophages and macrophage-like cells in relation to cell activation. Int Immunopharmacol 2(1):69–82
Sherwood TA, Nong L, Agudelo M, Newton C, Widen R, Klein TW (2009) Identification of transcription start sites and preferential expression of select CB2 transcripts in mouse and human B lymphocytes. J Neuroimmune Pharmacol 4(4):476–488. doi:10.1007/s11481-009-9169-z
Lu Q, Straiker A, Lu Q, Maguire G (2000) Expression of CB2 cannabinoid receptor mRNA in adult rat retina. Vis Neurosci 17(1):91–95
Lee SF, Newton C, Widen R, Friedman H, Klein TW (2001) Downregulation of cannabinoid receptor 2 (CB2) messenger RNA expression during in vitro stimulation of murine splenocytes with lipopolysaccharide. Adv Exp Med Biol 493:223–228. doi:10.1007/0-306-47611-8_26
Schatz AR, Lee M, Condie RB, Pulaski JT, Kaminski NE (1997) Cannabinoid receptors CB1 and CB2: a characterization of expression and adenylate cyclase modulation within the immune system. Toxicol Appl Pharmacol 142(2):278–287. doi:10.1006/taap.1996.8034
Ashton JC, Friberg D, Darlington CL, Smith PF (2006) Expression of the cannabinoid CB2 receptor in the rat cerebellum: an immunohistochemical study. Neurosci Lett 396(2):113–116. doi:10.1016/j.neulet.2005.11.038
Matias I, Pochard P, Orlando P, Salzet M, Pestel J, Di Marzo V (2002) Presence and regulation of the endocannabinoid system in human dendritic cells. Eur J Biochem 269(15):3771–3778
Atwood BK, Mackie K (2010) CB2: a cannabinoid receptor with an identity crisis. Br J Pharmacol 160(3):467–479. doi:10.1111/j.1476-5381.2010.00729.x
Derbenev AV, Stuart TC, Smith BN (2004) Cannabinoids suppress synaptic input to neurones of the rat dorsal motor nucleus of the vagus nerve. J Physiol 559(Pt 3):923–938. doi:10.1113/jphysiol.2004.067470
McCoy KL, Matveyeva M, Carlisle SJ, Cabral GA (1999) Cannabinoid inhibition of the processing of intact lysozyme by macrophages: evidence for CB2 receptor participation. J Pharmacol Exp Ther 289(3):1620–1625
Klegeris A, Bissonnette CJ, McGeer PL (2003) Reduction of human monocytic cell neurotoxicity and cytokine secretion by ligands of the cannabinoid-type CB2 receptor. Br J Pharmacol 139(4):775–786. doi:10.1038/sj.bjp.0705304
Griffin G, Fernando SR, Ross RA, McKay NG, Ashford ML, Shire D, Huffman JW, Yu S, Lainton JA, Pertwee RG (1997) Evidence for the presence of CB2-like cannabinoid receptors on peripheral nerve terminals. Eur J Pharmacol 339(1):53–61
Gong JP, Onaivi ES, Ishiguro H, Liu QR, Tagliaferro PA, Brusco A, Uhl GR (2006) Cannabinoid CB2 receptors: immunohistochemical localization in rat brain. Brain Res 1071(1):10–23. doi:10.1016/j.brainres.2005.11.035
Suarez J, Llorente R, Romero-Zerbo SY, Mateos B, Bermudez-Silva FJ, de Fonseca FR, Viveros MP (2009) Early maternal deprivation induces gender-dependent changes on the expression of hippocampal CB1 and CB2 cannabinoid receptors of neonatal rats. Hippocampus 19(7):623–632. doi:10.1002/hipo.20537
Suarez J, Bermudez-Silva FJ, Mackie K, Ledent C, Zimmer A, Cravatt BF, de Fonseca FR (2008) Immunohistochemical description of the endogenous cannabinoid system in the rat cerebellum and functionally related nuclei. J Comp Neurol 509(4):400–421. doi:10.1002/cne.21774
Stempel AV, Stumpf A, Zhang HY, Ozdogan T, Pannasch U, Theis AK, Otte DM, Wojtalla A, Racz I, Ponomarenko A, Xi ZX, Zimmer A, Schmitz D (2016) Cannabinoid type 2 receptors mediate a cell type-specific plasticity in the hippocampus. Neuron 90(4):795–809. doi:10.1016/j.neuron.2016.03.034
Liu QR, Pan CH, Hishimoto A, Li CY, Xi ZX, Llorente-Berzal A, Viveros MP, Ishiguro H, Arinami T, Onaivi ES, Uhl GR (2009) Species differences in cannabinoid receptor 2 (CNR2 gene): identification of novel human and rodent CB2 isoforms, differential tissue expression and regulation by cannabinoid receptor ligands. Genes Brain Behav 8(5):519–530. doi:10.1111/j.1601-183X.2009.00498.x
Baek JH, Darlington CL, Smith PF, Ashton JC (2013) Antibody testing for brain immunohistochemistry: brain immunolabeling for the cannabinoid CB2 receptor. J Neurosci Methods 216(2):87–95. doi:10.1016/j.jneumeth.2013.03.021
Castaneda JT, Harui A, Kiertscher SM, Roth JD, Roth MD (2013) Differential expression of intracellular and extracellular CB2 cannabinoid receptor protein by human peripheral blood leukocytes. J Neuroimmune Pharmacol 8(1):323–332. doi:10.1007/s11481-012-9430-8
Sanchez Lopez AJ, Roman-Vega L, Ramil Tojeiro E, Giuffrida A, Garcia-Merino A (2015) Regulation of cannabinoid receptor gene expression and endocannabinoid levels in lymphocyte subsets by interferon-beta: a longitudinal study in multiple sclerosis patients. Clin Exp Immunol 179(1):119–127. doi:10.1111/cei.12443
Agudelo M, Newton C, Widen R, Sherwood T, Nong L, Friedman H, Klein TW (2008) Cannabinoid receptor 2 (CB2) mediates immunoglobulin class switching from IgM to IgE in cultures of murine-purified B lymphocytes. J Neuroimmune Pharmacol 3(1):35–42. doi:10.1007/s11481-007-9088-9
Small-Howard AL, Shimoda LM, Adra CN, Turner H (2005) Anti-inflammatory potential of CB1-mediated cAMP elevation in mast cells. Biochem J 388(Pt 2):465–473. doi:10.1042/BJ20041682
Chouinard F, Turcotte C, Guan X, Larose MC, Poirier S, Bouchard L, Provost V, Flamand L, Grandvaux N, Flamand N (2013) 2-Arachidonoyl-glycerol- and arachidonic acid-stimulated neutrophils release antimicrobial effectors against E. coli, S. aureus, HSV-1, and RSV. J Leukoc Biol 93(2):267–276. doi:10.1189/jlb.0412200
Chouinard F, Lefebvre JS, Navarro P, Bouchard L, Ferland C, Lalancette-Hebert M, Marsolais D, Laviolette M, Flamand N (2011) The endocannabinoid 2-arachidonoyl-glycerol activates human neutrophils: critical role of its hydrolysis and de novo leukotriene B4 biosynthesis. J Immunol 186(5):3188–3196. doi:10.4049/jimmunol.1002853
Oka S, Ikeda S, Kishimoto S, Gokoh M, Yanagimoto S, Waku K, Sugiura T (2004) 2-Arachidonoylglycerol, an endogenous cannabinoid receptor ligand, induces the migration of EoL-1 human eosinophilic leukemia cells and human peripheral blood eosinophils. J Leukoc Biol 76(5):1002–1009. doi:10.1189/jlb.0404252
Ramirez SH, Reichenbach NL, Fan S, Rom S, Merkel SF, Wang X, Ho WZ, Persidsky Y (2013) Attenuation of HIV-1 replication in macrophages by cannabinoid receptor 2 agonists. J Leukoc Biol 93(5):801–810. doi:10.1189/jlb.1012523
Wright K, Rooney N, Feeney M, Tate J, Robertson D, Welham M, Ward S (2005) Differential expression of cannabinoid receptors in the human colon: cannabinoids promote epithelial wound healing. Gastroenterology 129(2):437–453. doi:10.1016/j.gastro.2005.05.026
Reichenbach V, Munoz-Luque J, Ros J, Casals G, Navasa M, Fernandez-Varo G, Morales-Ruiz M, Jimenez W (2013) Bacterial lipopolyshaccaride inhibits CB2 receptor expression in human monocytic cells. Gut 62(7):1089–1091. doi:10.1136/gutjnl-2012-303662
Montecucco F, Burger F, Mach F, Steffens S (2008) CB2 cannabinoid receptor agonist JWH-015 modulates human monocyte migration through defined intracellular signaling pathways. Am J Physiol Heart Circ Physiol 294(3):H1145–H1155. doi:10.1152/ajpheart.01328.2007
Kurihara R, Tohyama Y, Matsusaka S, Naruse H, Kinoshita E, Tsujioka T, Katsumata Y, Yamamura H (2006) Effects of peripheral cannabinoid receptor ligands on motility and polarization in neutrophil-like HL60 cells and human neutrophils. J Biol Chem 281(18):12908–12918. doi:10.1074/jbc.M510871200
Catani MV, Gasperi V, Catanzaro G, Baldassarri S, Bertoni A, Sinigaglia F, Avigliano L, Maccarrone M (2010) Human platelets express authentic CB(1) and CB(2) receptors. Curr Neurovasc Res 7(4):311–318
Signorello MG, Giacobbe E, Passalacqua M, Leoncini G (2013) The 2-arachidonoylglycerol effect on myosin light chain phosphorylation in human platelets. Biochimie 95(8):1620–1628. doi:10.1016/j.biochi.2013.05.003
Gardner B, Zu LX, Sharma S, Liu Q, Makriyannis A, Tashkin DP, Dubinett SM (2002) Autocrine and paracrine regulation of lymphocyte CB2 receptor expression by TGF-beta. Biochem Biophys Res Commun 290(1):91–96. doi:10.1006/bbrc.2001.6179
Demuth DG, Molleman A (2006) Cannabinoid signalling. Life Sci 78(6):549–563. doi:10.1016/j.lfs.2005.05.055
Chapman RS, Whetton AD, Chresta CM, Dive C (1995) Characterization of drug resistance mediated via the suppression of apoptosis by Abelson protein tyrosine kinase. Mol Pharmacol 48(2):334–343
Bayewitch M, Avidor-Reiss T, Levy R, Barg J, Mechoulam R, Vogel Z (1995) The peripheral cannabinoid receptor: adenylate cyclase inhibition and G protein coupling. FEBS Lett 375(1–2):143–147
Bouaboula M, Poinot-Chazel C, Marchand J, Canat X, Bourrie B, Rinaldi-Carmona M, Calandra B, Le Fur G, Casellas P (1996) Signaling pathway associated with stimulation of CB2 peripheral cannabinoid receptor. Involvement of both mitogen-activated protein kinase and induction of Krox-24 expression. Eur J Biochem 237(3):704–711
Merighi S, Gessi S, Varani K, Simioni C, Fazzi D, Mirandola P, Borea PA (2012) Cannabinoid CB2 receptors modulate ERK-1/2 kinase signalling and NO release in microglial cells stimulated with bacterial lipopolysaccharide. Br J Pharmacol 165(6):1773–1788. doi:10.1111/j.1476-5381.2011.01673.x
Yamaori S, Ishii H, Chiba K, Yamamoto I, Watanabe K (2013) Delta-tetrahydrocannabinol induces cytotoxicity in macrophage J774-1 cells: involvement of cannabinoid receptor 2 and p38 MAPK. Toxicology 314(2–3):254–261. doi:10.1016/j.tox.2013.10.007
Kobayashi Y, Arai S, Waku K, Sugiura T (2001) Activation by 2-arachidonoylglycerol, an endogenous cannabinoid receptor ligand, of p42/44 mitogen-activated protein kinase in HL-60 cells. J Biochem 129(5):665–669
He F, Qiao ZH, Cai J, Pierce W, He DC, Song ZH (2007) Involvement of the 90-kDa heat shock protein (Hsp-90) in CB2 cannabinoid receptor-mediated cell migration: a new role of Hsp-90 in migration signaling of a G protein-coupled receptor. Mol Pharmacol 72(5):1289–1300. doi:10.1124/mol.107.036566
Gokoh M, Kishimoto S, Oka S, Metani Y, Sugiura T (2005) 2-Arachidonoylglycerol, an endogenous cannabinoid receptor ligand, enhances the adhesion of HL-60 cells differentiated into macrophage-like cells and human peripheral blood monocytes. FEBS Lett 579(28):6473–6478. doi:10.1016/j.febslet.2005.10.030
Gkoumassi E, Dekkers BG, Droge MJ, Elzinga CR, Schmidt M, Meurs H, Zaagsma J, Nelemans SA (2007) Virodhamine and CP55,940 modulate cAMP production and IL-8 release in human bronchial epithelial cells. Br J Pharmacol 151(7):1041–1048. doi:10.1038/sj.bjp.0707320
Rhee MH, Nevo I, Levy R, Vogel Z (2000) Role of the highly conserved Asp-Arg-Tyr motif in signal transduction of the CB2 cannabinoid receptor. FEBS Lett 466(2–3):300–304
Bari M, Spagnuolo P, Fezza F, Oddi S, Pasquariello N, Finazzi-Agro A, Maccarrone M (2006) Effect of lipid rafts on CB2 receptor signaling and 2-arachidonoyl-glycerol metabolism in human immune cells. J Immunol 177(8):4971–4980
McAllister SD, Griffin G, Satin LS, Abood ME (1999) Cannabinoid receptors can activate and inhibit G protein-coupled inwardly rectifying potassium channels in a xenopus oocyte expression system. J Pharmacol Exp Ther 291(2):618–626
Ofek O, Attar-Namdar M, Kram V, Dvir-Ginzberg M, Mechoulam R, Zimmer A, Frenkel B, Shohami E, Bab I (2011) CB2 cannabinoid receptor targets mitogenic Gi protein-cyclin D1 axis in osteoblasts. J Bone Miner Res 26(2):308–316. doi:10.1002/jbmr.228
Zhou LL, Lin ZX, Fung KP, Cheng CH, Che CT, Zhao M, Wu SH, Zuo Z (2011) Celastrol-induced apoptosis in human HaCaT keratinocytes involves the inhibition of NF-kappaB activity. Eur J Pharmacol 670(2–3):399–408. doi:10.1016/j.ejphar.2011.09.014
Michler T, Storr M, Kramer J, Ochs S, Malo A, Reu S, Goke B, Schafer C (2013) Activation of cannabinoid receptor 2 reduces inflammation in acute experimental pancreatitis via intra-acinar activation of p38 and MK2-dependent mechanisms. Am J Physiol Gastrointest Liver Physiol 304(2):G181–G192. doi:10.1152/ajpgi.00133.2012
Zoratti C, Kipmen-Korgun D, Osibow K, Malli R, Graier WF (2003) Anandamide initiates Ca2+ signaling via CB2 receptor linked to phospholipase C in calf pulmonary endothelial cells. Br J Pharmacol 140(8):1351–1362. doi:10.1038/sj.bjp.0705529
Malysz J, Daza AV, Kage K, Grayson GK, Yao BB, Meyer MD, Gopalakrishnan M (2009) Characterization of human cannabinoid CB2 receptor coupled to chimeric Galpha(qi5) and Galpha(qo5) proteins. Eur J Pharmacol 603(1–3):12–21. doi:10.1016/j.ejphar.2008.11.047
Larose MC, Turcotte C, Chouinard F, Ferland C, Martin C, Provost V, Laviolette M, Flamand N (2014) Mechanisms of human eosinophil migration induced by the combination of IL-5 and the endocannabinoid 2-arachidonoyl-glycerol. J Allergy Clin Immunol 133(5):1480–1482, 1482 e1481–1483. doi:10.1016/j.jaci.2013.12.1081
Turcotte C, Chouinard F, Lefebvre JS, Flamand N (2015) Regulation of inflammation by cannabinoids, the endocannabinoids 2-arachidonoyl-glycerol and arachidonoyl-ethanolamide, and their metabolites. J Leukoc Biol 97(6):1049–1070. doi:10.1189/jlb.3RU0115-021R
Ortega-Gutierrez S, Molina-Holgado E, Guaza C (2005) Effect of anandamide uptake inhibition in the production of nitric oxide and in the release of cytokines in astrocyte cultures. Glia 52(2):163–168. doi:10.1002/glia.20229
Chiurchiu V, Cencioni MT, Bisicchia E, De Bardi M, Gasperini C, Borsellino G, Centonze D, Battistini L, Maccarrone M (2013) Distinct modulation of human myeloid and plasmacytoid dendritic cells by anandamide in multiple sclerosis. Ann Neurol 73(5):626–636. doi:10.1002/ana.23875
Eljaschewitsch E, Witting A, Mawrin C, Lee T, Schmidt PM, Wolf S, Hoertnagl H, Raine CS, Schneider-Stock R, Nitsch R, Ullrich O (2006) The endocannabinoid anandamide protects neurons during CNS inflammation by induction of MKP-1 in microglial cells. Neuron 49(1):67–79. doi:10.1016/j.neuron.2005.11.027
Correa F, Hernangomez M, Mestre L, Loria F, Spagnolo A, Docagne F, Di Marzo V, Guaza C (2010) Anandamide enhances IL-10 production in activated microglia by targeting CB2 receptors: roles of ERK1/2, JNK, and NF-kappaB. Glia 58(2):135–147. doi:10.1002/glia.20907
Correa F, Hernangomez-Herrero M, Mestre L, Loria F, Docagne F, Guaza C (2011) The endocannabinoid anandamide downregulates IL-23 and IL-12 subunits in a viral model of multiple sclerosis: evidence for a cross-talk between IL-12p70/IL-23 axis and IL-10 in microglial cells. Brain Behav Immun 25(4):736–749. doi:10.1016/j.bbi.2011.01.020
Malek N, Popiolek-Barczyk K, Mika J, Przewlocka B, Starowicz K (2015) Anandamide, acting via CB2 Receptors, alleviates LPS-induced neuroinflammation in rat primary microglial cultures. Neural Plast 2015:130639. doi:10.1155/2015/130639
Eisenstein TK, Meissler JJ, Wilson Q, Gaughan JP, Adler MW (2007) Anandamide and Δ9-tetrahydrocannabinol directly inhibit cells of the immune system via CB2 receptors. J Neuroimmunol 189(1–2):17–22. doi:10.1016/j.jneuroim.2007.06.001
Cencioni MT, Chiurchiu V, Catanzaro G, Borsellino G, Bernardi G, Battistini L, Maccarrone M (2010) Anandamide suppresses proliferation and cytokine release from primary human T-lymphocytes mainly via CB2 receptors. PLoS One 5(1):e8688. doi:10.1371/journal.pone.0008688
Coopman K, Smith LD, Wright KL, Ward SG (2007) Temporal variation in CB2R levels following T lymphocyte activation: evidence that cannabinoids modulate CXCL12-induced chemotaxis. Int Immunopharmacol 7(3):360–371. doi:10.1016/j.intimp.2006.11.008
Joseph J, Niggemann B, Zaenker KS, Entschladen F (2004) Anandamide is an endogenous inhibitor for the migration of tumor cells and T lymphocytes. Cancer Immunol Immunother 53(8):723–728. doi:10.1007/s00262-004-0509-9
Rayman N, Lam KH, Laman JD, Simons PJ, Lowenberg B, Sonneveld P, Delwel R (2004) Distinct expression profiles of the peripheral cannabinoid receptor in lymphoid tissues depending on receptor activation status. J Immunol 172(4):2111–2117
Jorda MA, Verbakel SE, Valk PJ, Vankan-Berkhoudt YV, Maccarrone M, Finazzi-Agro A, Lowenberg B, Delwel R (2002) Hematopoietic cells expressing the peripheral cannabinoid receptor migrate in response to the endocannabinoid 2-arachidonoylglycerol. Blood 99(8):2786–2793
Tanikawa T, Kurohane K, Imai Y (2007) Induction of preferential chemotaxis of unstimulated B-lymphocytes by 2-arachidonoylglycerol in immunized mice. Microbiol Immunol 51(10):1013–1019
Maestroni GJ (2004) The endogenous cannabinoid 2-arachidonoyl glycerol as in vivo chemoattractant for dendritic cells and adjuvant for Th1 response to a soluble protein. FASEB J 18(15):1914–1916. doi:10.1096/fj.04-2190fje
Kishimoto S, Oka S, Gokoh M, Sugiura T (2006) Chemotaxis of human peripheral blood eosinophils to 2-arachidonoylglycerol: comparison with other eosinophil chemoattractants. Int Arch Allergy Immunol 140(Suppl 1):3–7. doi:10.1159/000092704
Shiratsuchi A, Watanabe I, Yoshida H, Nakanishi Y (2008) Involvement of cannabinoid receptor CB2 in dectin-1-mediated macrophage phagocytosis. Immunol Cell Biol 86(2):179–184. doi:10.1038/sj.icb.7100121
Kishimoto S, Gokoh M, Oka S, Muramatsu M, Kajiwara T, Waku K, Sugiura T (2003) 2-arachidonoylglycerol induces the migration of HL-60 cells differentiated into macrophage-like cells and human peripheral blood monocytes through the cannabinoid CB2 receptor-dependent mechanism. J Biol Chem 278(27):24469–24475. doi:10.1074/jbc.M301359200
Kishimoto S, Kobayashi Y, Oka S, Gokoh M, Waku K, Sugiura T (2004) 2-Arachidonoylglycerol, an endogenous cannabinoid receptor ligand, induces accelerated production of chemokines in HL-60 cells. J Biochem 135(4):517–524
Walter L, Franklin A, Witting A, Wade C, Xie Y, Kunos G, Mackie K, Stella N (2003) Nonpsychotropic cannabinoid receptors regulate microglial cell migration. J Neurosci 23(4):1398–1405
Gokoh M, Kishimoto S, Oka S, Mori M, Waku K, Ishima Y, Sugiura T (2005) 2-Arachidonoylglycerol, an endogenous cannabinoid receptor ligand, induces rapid actin polymerization in HL-60 cells differentiated into macrophage-like cells. Biochem J 386(Pt 3):583–589. doi:10.1042/BJ20041163
Kishimoto S, Muramatsu M, Gokoh M, Oka S, Waku K, Sugiura T (2005) Endogenous cannabinoid receptor ligand induces the migration of human natural killer cells. J Biochem 137(2):217–223. doi:10.1093/jb/mvi021
Gasperi V, Evangelista D, Chiurchiu V, Florenzano F, Savini I, Oddi S, Avigliano L, Catani MV, Maccarrone M (2014) 2-Arachidonoylglycerol modulates human endothelial cell/leukocyte interactions by controlling selectin expression through CB1 and CB2 receptors. Int J Biochem Cell Biol 51:79–88. doi:10.1016/j.biocel.2014.03.028
Rock RB, Gekker G, Hu S, Sheng WS, Cabral GA, Martin BR, Peterson PK (2007) WIN55,212-2-mediated inhibition of HIV-1 expression in microglial cells: involvement of cannabinoid receptors. J Neuroimmune Pharmacol 2(2):178–183. doi:10.1007/s11481-006-9040-4
Sheng WS, Hu S, Min X, Cabral GA, Lokensgard JR, Peterson PK (2005) Synthetic cannabinoid WIN55,212-2 inhibits generation of inflammatory mediators by IL-1β-stimulated human astrocytes. Glia 49(2):211–219. doi:10.1002/glia.20108
Do Y, McKallip RJ, Nagarkatti M, Nagarkatti PS (2004) Activation through cannabinoid receptors 1 and 2 on dendritic cells triggers NF-κB-dependent apoptosis: novel role for endogenous and exogenous cannabinoids in immunoregulation. J Immunol 173(4):2373–2382
Adhikary S, Kocieda VP, Yen JH, Tuma RF, Ganea D (2012) Signaling through cannabinoid receptor 2 suppresses murine dendritic cell migration by inhibiting matrix metalloproteinase 9 expression. Blood 120(18):3741–3749. doi:10.1182/blood-2012-06-435362
Rajesh M, Mukhopadhyay P, Batkai S, Hasko G, Liaudet L, Huffman JW, Csiszar A, Ungvari Z, Mackie K, Chatterjee S, Pacher P (2007) CB2-receptor stimulation attenuates TNF-α-induced human endothelial cell activation, transendothelial migration of monocytes, and monocyte-endothelial adhesion. Am J Physiol Heart Circ Physiol 293(4):H2210–H2218. doi:10.1152/ajpheart.00688.2007
Persidsky Y, Fan S, Dykstra H, Reichenbach NL, Rom S, Ramirez SH (2015) Activation of cannabinoid type two receptors (CB2) diminish inflammatory responses in macrophages and brain endothelium. J Neuroimmune Pharmacol 10(2):302–308. doi:10.1007/s11481-015-9591-3
Hao MX, Jiang LS, Fang NY, Pu J, Hu LH, Shen LH, Song W, He B (2010) The cannabinoid WIN55,212-2 protects against oxidized LDL-induced inflammatory response in murine macrophages. J Lipid Res 51(8):2181–2190. doi:10.1194/jlr.M001511
Ross RA, Brockie HC, Pertwee RG (2000) Inhibition of nitric oxide production in RAW264.7 macrophages by cannabinoids and palmitoylethanolamide. Eur J Pharmacol 401(2):121–130
Raborn ES, Marciano-Cabral F, Buckley NE, Martin BR, Cabral GA (2008) The cannabinoid Δ-9-tetrahydrocannabinol mediates inhibition of macrophage chemotaxis to RANTES/CCL5: linkage to the CB2 receptor. J Neuroimmune Pharmacol 3(2):117–129. doi:10.1007/s11481-007-9077-z
Correa F, Mestre L, Docagne F, Guaza C (2005) Activation of cannabinoid CB2 receptor negatively regulates IL-12p40 production in murine macrophages: role of IL-10 and ERK1/2 kinase signaling. Br J Pharmacol 145(4):441–448. doi:10.1038/sj.bjp.0706215
Giudice ED, Rinaldi L, Passarotto M, Facchinetti F, D’Arrigo A, Guiotto A, Carbonare MD, Battistin L, Leon A (2007) Cannabidiol, unlike synthetic cannabinoids, triggers activation of RBL-2H3 mast cells. J Leukoc Biol 81(6):1512–1522. doi:10.1189/jlb.1206738
Romero-Sandoval EA, Horvath R, Landry RP, DeLeo JA (2009) Cannabinoid receptor type 2 activation induces a microglial anti-inflammatory phenotype and reduces migration via MKP induction and ERK dephosphorylation. Mol Pain 5:25. doi:10.1186/1744-8069-5-25
Murikinati S, Juttler E, Keinert T, Ridder DA, Muhammad S, Waibler Z, Ledent C, Zimmer A, Kalinke U, Schwaninger M (2010) Activation of cannabinoid 2 receptors protects against cerebral ischemia by inhibiting neutrophil recruitment. FASEB J 24(3):788–798. doi:10.1096/fj.09-141275
Montecucco F, Di Marzo V, da Silva RF, Vuilleumier N, Capettini L, Lenglet S, Pagano S, Piscitelli F, Quintao S, Bertolotto M, Pelli G, Galan K, Pilet L, Kuzmanovic K, Burger F, Pane B, Spinella G, Braunersreuther V, Gayet-Ageron A, Pende A, Viviani GL, Palombo D, Dallegri F, Roux-Lombard P, Santos RA, Stergiopulos N, Steffens S, Mach F (2012) The activation of the cannabinoid receptor type 2 reduces neutrophilic protease-mediated vulnerability in atherosclerotic plaques. Eur Heart J 33(7):846–856. doi:10.1093/eurheartj/ehr449
Yuan M, Kiertscher SM, Cheng Q, Zoumalan R, Tashkin DP, Roth MD (2002) Δ9-tetrahydrocannabinol regulates Th1/Th2 cytokine balance in activated human T cells. J Neuroimmunol 133(1–2):124–131
Ghosh S, Preet A, Groopman JE, Ganju RK (2006) Cannabinoid receptor CB2 modulates the CXCL12/CXCR4-mediated chemotaxis of T lymphocytes. Mol Immunol 43(14):2169–2179. doi:10.1016/j.molimm.2006.01.005
Robinson RH, Meissler JJ, Fan X, Yu D, Adler MW, Eisenstein TK (2015) A CB2-selective cannabinoid suppresses T-cell activities and increases tregs and IL-10. J Neuroimmune Pharmacol 10(2):318–332. doi:10.1007/s11481-015-9611-3
Csoka B, Nemeth ZH, Mukhopadhyay P, Spolarics Z, Rajesh M, Federici S, Deitch EA, Batkai S, Pacher P, Hasko G (2009) CB2 cannabinoid receptors contribute to bacterial invasion and mortality in polymicrobial sepsis. PLoS One 4(7):e6409. doi:10.1371/journal.pone.0006409
Karsak M, Gaffal E, Date R, Wang-Eckhardt L, Rehnelt J, Petrosino S, Starowicz K, Steuder R, Schlicker E, Cravatt B, Mechoulam R, Buettner R, Werner S, Di Marzo V, Tuting T, Zimmer A (2007) Attenuation of allergic contact dermatitis through the endocannabinoid system. Science 316(5830):1494–1497. doi:10.1126/science.1142265
Hegde VL, Hegde S, Cravatt BF, Hofseth LJ, Nagarkatti M, Nagarkatti PS (2008) Attenuation of experimental autoimmune hepatitis by exogenous and endogenous cannabinoids: involvement of regulatory T cells. Mol Pharmacol 74(1):20–33. doi:10.1124/mol.108.047035
Engel MA, Kellermann CA, Burnat G, Hahn EG, Rau T, Konturek PC (2010) Mice lacking cannabinoid CB1-, CB2-receptors or both receptors show increased susceptibility to trinitrobenzene sulfonic acid (TNBS)-induced colitis. J Physiol Pharmacol 61(1):89–97
Duerr GD, Heinemann JC, Gestrich C, Heuft T, Klaas T, Keppel K, Roell W, Klein A, Zimmer A, Velten M, Kilic A, Bindila L, Lutz B, Dewald O (2015) Impaired border zone formation and adverse remodeling after reperfused myocardial infarction in cannabinoid CB2 receptor deficient mice. Life Sci 138:8–17. doi:10.1016/j.lfs.2014.11.005
Amenta PS, Jallo JI, Tuma RF, Hooper DC, Elliott MB (2014) Cannabinoid receptor type-2 stimulation, blockade, and deletion alter the vascular inflammatory responses to traumatic brain injury. J Neuroinflammation 11:191. doi:10.1186/s12974-014-0191-6
Zhao Y, Liu Y, Zhang W, Xue J, Wu YZ, Xu W, Liang X, Chen T, Kishimoto C, Yuan Z (2010) WIN55212-2 ameliorates atherosclerosis associated with suppression of pro-inflammatory responses in ApoE-knockout mice. Eur J Pharmacol 649(1–3):285–292. doi:10.1016/j.ejphar.2010.09.027
Steffens S, Veillard NR, Arnaud C, Pelli G, Burger F, Staub C, Karsak M, Zimmer A, Frossard JL, Mach F (2005) Low dose oral cannabinoid therapy reduces progression of atherosclerosis in mice. Nature 434(7034):782–786. doi:10.1038/nature03389
McKallip RJ, Nagarkatti M, Nagarkatti PS (2005) Δ-9-tetrahydrocannabinol enhances breast cancer growth and metastasis by suppression of the antitumor immune response. J Immunol 174(6):3281–3289
Zarruk JG, Fernandez-Lopez D, Garcia-Yebenes I, Garcia-Gutierrez MS, Vivancos J, Nombela F, Torres M, Burguete MC, Manzanares J, Lizasoain I, Moro MA (2012) Cannabinoid type 2 receptor activation downregulates stroke-induced classic and alternative brain macrophage/microglial activation concomitant to neuroprotection. Stroke 43(1):211–219. doi:10.1161/STROKEAHA.111.631044
Maresz K, Pryce G, Ponomarev ED, Marsicano G, Croxford JL, Shriver LP, Ledent C, Cheng X, Carrier EJ, Mann MK, Giovannoni G, Pertwee RG, Yamamura T, Buckley NE, Hillard CJ, Lutz B, Baker D, Dittel BN (2007) Direct suppression of CNS autoimmune inflammation via the cannabinoid receptor CB1 on neurons and CB2 on autoreactive T cells. Nat Med 13(4):492–497. doi:10.1038/nm1561
Batkai S, Mukhopadhyay P, Horvath B, Rajesh M, Gao RY, Mahadevan A, Amere M, Battista N, Lichtman AH, Gauson LA, Maccarrone M, Pertwee RG, Pacher P (2012) Δ8-Tetrahydrocannabivarin prevents hepatic ischaemia/reperfusion injury by decreasing oxidative stress and inflammatory responses through cannabinoid CB2 receptors. Br J Pharmacol 165(8):2450–2461. doi:10.1111/j.1476-5381.2011.01410.x
Tang J, Chen Q, Guo J, Yang L, Tao Y, Li L, Miao H, Feng H, Chen Z, Zhu G (2015) Minocycline attenuates neonatal germinal-matrix-hemorrhage-induced neuroinflammation and brain edema by activating cannabinoid receptor 2. Mol Neurobiol. doi:10.1007/s12035-015-9154-x
Newton CA, Chou PJ, Perkins I, Klein TW (2009) CB1 and CB2 cannabinoid receptors mediate different aspects of Δ-9-tetrahydrocannabinol (THC)-induced T helper cell shift following immune activation by Legionella pneumophila infection. J Neuroimmune Pharmacol 4(1):92–102. doi:10.1007/s11481-008-9126-2
Buchweitz JP, Karmaus PW, Williams KJ, Harkema JR, Kaminski NE (2008) Targeted deletion of cannabinoid receptors CB1 and CB2 produced enhanced inflammatory responses to influenza A/PR/8/34 in the absence and presence of Δ9-tetrahydrocannabinol. J Leukoc Biol 83(3):785–796. doi:10.1189/jlb.0907618
Di Filippo C, Rossi F, Rossi S, D’Amico M (2004) Cannabinoid CB2 receptor activation reduces mouse myocardial ischemia-reperfusion injury: involvement of cytokine/chemokines and PMN. J Leukoc Biol 75(3):453–459. doi:10.1189/jlb.0703303
Giannini L, Nistri S, Mastroianni R, Cinci L, Vannacci A, Mariottini C, Passani MB, Mannaioni PF, Bani D, Masini E (2008) Activation of cannabinoid receptors prevents antigen-induced asthma-like reaction in guinea pigs. J Cell Mol Med 12(6A):2381–2394. doi:10.1111/j.1582-4934.2008.00258.x
Tambaro S, Casu MA, Mastinu A, Lazzari P (2014) Evaluation of selective cannabinoid CB1 and CB2 receptor agonists in a mouse model of lipopolysaccharide-induced interstitial cystitis. Eur J Pharmacol 729:67–74. doi:10.1016/j.ejphar.2014.02.013
Sardinha J, Kelly ME, Zhou J, Lehmann C (2014) Experimental cannabinoid 2 receptor-mediated immune modulation in sepsis. Mediators Inflamm 2014:978678. doi:10.1155/2014/978678
Adhikary S, Li H, Heller J, Skarica M, Zhang M, Ganea D, Tuma RF (2011) Modulation of inflammatory responses by a cannabinoid-2-selective agonist after spinal cord injury. J Neurotrauma 28(12):2417–2427. doi:10.1089/neu.2011.1853
Zoppi S, Madrigal JL, Caso JR, Garcia-Gutierrez MS, Manzanares J, Leza JC, Garcia-Bueno B (2014) Regulatory role of the cannabinoid CB2 receptor in stress-induced neuroinflammation in mice. Br J Pharmacol 171(11):2814–2826. doi:10.1111/bph.12607
Amenta PS, Jallo JI, Tuma RF, Elliott MB (2012) A cannabinoid type 2 receptor agonist attenuates blood-brain barrier damage and neurodegeneration in a murine model of traumatic brain injury. J Neurosci Res 90(12):2293–2305. doi:10.1002/jnr.23114
Dinh TP, Carpenter D, Leslie FM, Freund TF, Katona I, Sensi SL, Kathuria S, Piomelli D (2002) Brain monoglyceride lipase participating in endocannabinoid inactivation. Proc Natl Acad Sci USA 99(16):10819–10824. doi:10.1073/pnas.152334899
Goparaju SK, Ueda N, Yamaguchi H, Yamamoto S (1998) Anandamide amidohydrolase reacting with 2-arachidonoylglycerol, another cannabinoid receptor ligand. FEBS Lett 422(1):69–73
Rouzer CA, Marnett LJ (2011) Endocannabinoid oxygenation by cyclooxygenases, lipoxygenases, and cytochromes P450: cross-talk between the eicosanoid and endocannabinoid signaling pathways. Chem Rev 111(10):5899–5921. doi:10.1021/cr2002799
Comelli F, Giagnoni G, Bettoni I, Colleoni M, Costa B (2007) The inhibition of monoacylglycerol lipase by URB602 showed an anti-inflammatory and anti-nociceptive effect in a murine model of acute inflammation. Br J Pharmacol 152(5):787–794. doi:10.1038/sj.bjp.0707425
Wen J, Ribeiro R, Tanaka M, Zhang Y (2015) Activation of CB2 receptor is required for the therapeutic effect of ABHD6 inhibition in experimental autoimmune encephalomyelitis. Neuropharmacology 99:196–209. doi:10.1016/j.neuropharm.2015.07.010
Costola-de-Souza C, Ribeiro A, Ferraz-de-Paula V, Calefi AS, Aloia TP, Gimenes-Junior JA, de Almeida VI, Pinheiro ML, Palermo-Neto J (2013) Monoacylglycerol lipase (MAGL) inhibition attenuates acute lung injury in mice. PLoS One 8(10):e77706. doi:10.1371/journal.pone.0077706
Naidu PS, Kinsey SG, Guo TL, Cravatt BF, Lichtman AH (2010) Regulation of inflammatory pain by inhibition of fatty acid amide hydrolase. J Pharmacol Exp Ther 334(1):182–190. doi:10.1124/jpet.109.164806
Booker L, Kinsey SG, Abdullah RA, Blankman JL, Long JZ, Ezzili C, Boger DL, Cravatt BF, Lichtman AH (2012) The fatty acid amide hydrolase (FAAH) inhibitor PF-3845 acts in the nervous system to reverse LPS-induced tactile allodynia in mice. Br J Pharmacol 165(8):2485–2496. doi:10.1111/j.1476-5381.2011.01445.x
Krustev E, Reid A, McDougall JJ (2014) Tapping into the endocannabinoid system to ameliorate acute inflammatory flares and associated pain in mouse knee joints. Arthritis Res Ther 16(5):437. doi:10.1186/s13075-014-0437-9
Alhouayek M, Lambert DM, Delzenne NM, Cani PD, Muccioli GG (2011) Increasing endogenous 2-arachidonoylglycerol levels counteracts colitis and related systemic inflammation. FASEB J 25(8):2711–2721. doi:10.1096/fj.10-176602
Galanakis DK (1992) Anticoagulant albumin fragments that bind to fibrinogen/fibrin: possible implications. Semin Thromb Hemost 18(1):44–52. doi:10.1055/s-2007-1002409
Oka S, Wakui J, Ikeda S, Yanagimoto S, Kishimoto S, Gokoh M, Nasui M, Sugiura T (2006) Involvement of the cannabinoid CB2 receptor and its endogenous ligand 2-arachidonoylglycerol in oxazolone-induced contact dermatitis in mice. J Immunol 177(12):8796–8805
Maresz K, Carrier EJ, Ponomarev ED, Hillard CJ, Dittel BN (2005) Modulation of the cannabinoid CB2 receptor in microglial cells in response to inflammatory stimuli. J Neurochem 95(2):437–445. doi:10.1111/j.1471-4159.2005.03380.x
Yiangou Y, Facer P, Durrenberger P, Chessell IP, Naylor A, Bountra C, Banati RR, Anand P (2006) COX-2, CB2 and P2X7-immunoreactivities are increased in activated microglial cells/macrophages of multiple sclerosis and amyotrophic lateral sclerosis spinal cord. BMC Neurol 6:12. doi:10.1186/1471-2377-6-12
Walczak JS, Pichette V, Leblond F, Desbiens K, Beaulieu P (2005) Behavioral, pharmacological and molecular characterization of the saphenous nerve partial ligation: a new model of neuropathic pain. Neuroscience 132(4):1093–1102. doi:10.1016/j.neuroscience.2005.02.010
Oka S, Yanagimoto S, Ikeda S, Gokoh M, Kishimoto S, Waku K, Ishima Y, Sugiura T (2005) Evidence for the involvement of the cannabinoid CB2 receptor and its endogenous ligand 2-arachidonoylglycerol in 12-O-tetradecanoylphorbol-13-acetate-induced acute inflammation in mouse ear. J Biol Chem 280(18):18488–18497. doi:10.1074/jbc.M413260200
Harris ED Jr (1990) Rheumatoid arthritis. Pathophysiology and implications for therapy. N Engl J Med 322(18):1277–1289. doi:10.1056/NEJM199005033221805
Richardson D, Pearson RG, Kurian N, Latif ML, Garle MJ, Barrett DA, Kendall DA, Scammell BE, Reeve AJ, Chapman V (2008) Characterisation of the cannabinoid receptor system in synovial tissue and fluid in patients with osteoarthritis and rheumatoid arthritis. Arthritis Res Ther 10(2):R43. doi:10.1186/ar2401
Gui H, Liu X, Wang ZW, He DY, Su DF, Dai SM (2014) Expression of cannabinoid receptor 2 and its inhibitory effects on synovial fibroblasts in rheumatoid arthritis. Rheumatology (Oxford) 53(5):802–809. doi:10.1093/rheumatology/ket447
Fukuda S, Kohsaka H, Takayasu A, Yokoyama W, Miyabe C, Miyabe Y, Harigai M, Miyasaka N, Nanki T (2014) Cannabinoid receptor 2 as a potential therapeutic target in rheumatoid arthritis. BMC Musculoskelet Disord 15:275. doi:10.1186/1471-2474-15-275
Sophocleous A, Landao-Bassonga E, Van’t Hof RJ, Idris AI, Ralston SH (2011) The type 2 cannabinoid receptor regulates bone mass and ovariectomy-induced bone loss by affecting osteoblast differentiation and bone formation. Endocrinology 152(6):2141–2149. doi:10.1210/en.2010-0930
Gui H, Liu X, Liu LR, Su DF, Dai SM (2015) Activation of cannabinoid receptor 2 attenuates synovitis and joint distruction in collagen-induced arthritis. Immunobiology 220(6):817–822. doi:10.1016/j.imbio.2014.12.012
Sumariwalla PF, Gallily R, Tchilibon S, Fride E, Mechoulam R, Feldmann M (2004) A novel synthetic, nonpsychoactive cannabinoid acid (HU-320) with antiinflammatory properties in murine collagen-induced arthritis. Arthritis Rheum 50(3):985–998. doi:10.1002/art.20050
Insull W Jr (2009) The pathology of atherosclerosis: plaque development and plaque responses to medical treatment. Am J Med 122(1 Suppl):S3–S14. doi:10.1016/j.amjmed.2008.10.013
Chiurchiu V, Lanuti M, Catanzaro G, Fezza F, Rapino C, Maccarrone M (2014) Detailed characterization of the endocannabinoid system in human macrophages and foam cells, and anti-inflammatory role of type-2 cannabinoid receptor. Atherosclerosis 233(1):55–63. doi:10.1016/j.atherosclerosis.2013.12.042
Zhao Y, Yuan Z, Liu Y, Xue J, Tian Y, Liu W, Zhang W, Shen Y, Xu W, Liang X, Chen T (2010) Activation of cannabinoid CB2 receptor ameliorates atherosclerosis associated with suppression of adhesion molecules. J Cardiovasc Pharmacol 55(3):292–298. doi:10.1097/FJC.0b013e3181d2644d
Netherland CD, Pickle TG, Bales A, Thewke DP (2010) Cannabinoid receptor type 2 (CB2) deficiency alters atherosclerotic lesion formation in hyperlipidemic Ldlr-null mice. Atherosclerosis 213(1):102–108. doi:10.1016/j.atherosclerosis.2010.07.060
Delsing DJ, Leijten FP, Arts K, van Eenennaam H, Garritsen A, Gijbels MJ, de Winther MP, van Elsas A (2011) Cannabinoid receptor 2 deficiency in haematopoietic cells aggravates early atherosclerosis in LDL receptor deficient mice. Open Cardiovasc Med J 5:15–21. doi:10.2174/1874192401105010015
Netherland-Van Dyke C, Rodgers W, Fulmer M, Lahr Z, Thewke D (2015) Cannabinoid receptor type 2 (CB2) dependent and independent effects of WIN55,212-2 on atherosclerosis in Ldlr-null mice. J Cardiol Ther 3(2):53–63. doi:10.12970/2311-052X.2015.03.02.2
Willecke F, Zeschky K, Ortiz Rodriguez A, Colberg C, Auwarter V, Kneisel S, Hutter M, Lozhkin A, Hoppe N, Wolf D, von zur Muhlen C, Moser M, Hilgendorf I, Bode C, Zirlik A (2011) Cannabinoid receptor 2 signaling does not modulate atherogenesis in mice. PLoS One 6(4):e19405. doi:10.1371/journal.pone.0019405
Hanauer SB (2006) Inflammatory bowel disease: epidemiology, pathogenesis, and therapeutic opportunities. Inflamm Bowel Dis 12(Suppl 1):S3–S9
Massa F, Marsicano G, Hermann H, Cannich A, Monory K, Cravatt BF, Ferri GL, Sibaev A, Storr M, Lutz B (2004) The endogenous cannabinoid system protects against colonic inflammation. J Clin Invest 113(8):1202–1209. doi:10.1172/JCI19465
Storr MA, Keenan CM, Emmerdinger D, Zhang H, Yuce B, Sibaev A, Massa F, Buckley NE, Lutz B, Goke B, Brand S, Patel KD, Sharkey KA (2008) Targeting endocannabinoid degradation protects against experimental colitis in mice: involvement of CB1 and CB2 receptors. J Mol Med (Berl) 86(8):925–936. doi:10.1007/s00109-008-0359-6
Storr MA, Keenan CM, Zhang H, Patel KD, Makriyannis A, Sharkey KA (2009) Activation of the cannabinoid 2 receptor (CB2) protects against experimental colitis. Inflamm Bowel Dis 15(11):1678–1685. doi:10.1002/ibd.20960
Borrelli F, Fasolino I, Romano B, Capasso R, Maiello F, Coppola D, Orlando P, Battista G, Pagano E, Di Marzo V, Izzo AA (2013) Beneficial effect of the non-psychotropic plant cannabinoid cannabigerol on experimental inflammatory bowel disease. Biochem Pharmacol 85(9):1306–1316. doi:10.1016/j.bcp.2013.01.017
Bento AF, Marcon R, Dutra RC, Claudino RF, Cola M, Leite DF, Calixto JB (2011) β-caryophyllene inhibits dextran sulfate sodium-induced colitis in mice through CB2 receptor activation and PPARγ pathway. Am J Pathol 178(3):1153–1166. doi:10.1016/j.ajpath.2010.11.052
El Bakali J, Gilleron P, Body-Malapel M, Mansouri R, Muccioli GG, Djouina M, Barczyk A, Klupsch F, Andrzejak V, Lipka E, Furman C, Lambert DM, Chavatte P, Desreumaux P, Millet R (2012) 4-Oxo-1,4-Dihydropyridines as selective CB(2) cannabinoid receptor ligands. Part 2: discovery of new agonists endowed with protective effect against experimental colitis. J Med Chem 55(20):8948–8952. doi:10.1021/jm3008568
Tourteau A, Andrzejak V, Body-Malapel M, Lemaire L, Lemoine A, Mansouri R, Djouina M, Renault N, El Bakali J, Desreumaux P, Muccioli GG, Lambert DM, Chavatte P, Rigo B, Leleu-Chavain N, Millet R (2013) 3-Carboxamido-5-aryl-isoxazoles as new CB2 agonists for the treatment of colitis. Bioorg Med Chem 21(17):5383–5394. doi:10.1016/j.bmc.2013.06.010
El Bakali J, Muccioli GG, Body-Malapel M, Djouina M, Klupsch F, Ghinet A, Barczyk A, Renault N, Chavatte P, Desreumaux P, Lambert DM, Millet R (2015) Conformational restriction leading to a selective CB2 cannabinoid receptor agonist orally active against colitis. ACS Med Chem Lett 6(2):198–203. doi:10.1021/ml500439x
Giblin GM, O’Shaughnessy CT, Naylor A, Mitchell WL, Eatherton AJ, Slingsby BP, Rawlings DA, Goldsmith P, Brown AJ, Haslam CP, Clayton NM, Wilson AW, Chessell IP, Wittington AR, Green R (2007) Discovery of 2-[(2,4-dichlorophenyl)amino]-N-[(tetrahydro- 2H-pyran-4-yl)methyl]-4-(trifluoromethyl)-5-pyrimidinecarboxamide, a selective CB2 receptor agonist for the treatment of inflammatory pain. J Med Chem 50(11):2597–2600. doi:10.1021/jm061195+
Mitchell WL, Giblin GM, Naylor A, Eatherton AJ, Slingsby BP, Rawlings AD, Jandu KS, Haslam CP, Brown AJ, Goldsmith P, Clayton NM, Wilson AW, Chessell IP, Green RH, Whittington AR, Wall ID (2009) Pyridine-3-carboxamides as novel CB2 agonists for analgesia. Bioorg Med Chem Lett 19(1):259–263. doi:10.1016/j.bmcl.2008.10.118
Giblin GM, Billinton A, Briggs M, Brown AJ, Chessell IP, Clayton NM, Eatherton AJ, Goldsmith P, Haslam C, Johnson MR, Mitchell WL, Naylor A, Perboni A, Slingsby BP, Wilson AW (2009) Discovery of 1-[4-(3-chlorophenylamino)-1-methyl-1H-pyrrolo[3,2-c]pyridin-7-yl]-1-morpholin-4-ylmethanone (GSK554418A), a brain penetrant 5-azaindole CB2 agonist for the treatment of chronic pain. J Med Chem 52(19):5785–5788. doi:10.1021/jm9009857
Acknowledgments
This work is/was supported by Grants to NF from the Canadian Institutes of Health Research (MOP-97930) and The Natural Sciences and Engineering Research Council of Canada. NF also received operating grants from le Fonds sur les maladies respiratoires J.-D. Bégin—P.-H. Lavoie. CT was the recipient of a doctoral award from the Canadian Consortium for the Investigation of Cannabinoids and is currently supported by the Canadian Institutes of Health Research. MRB, ML, and NF are members of the inflammation group of the Respiratory Health Network of the Fonds de recherche en Santé-Québec.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Turcotte, C., Blanchet, MR., Laviolette, M. et al. The CB2 receptor and its role as a regulator of inflammation. Cell. Mol. Life Sci. 73, 4449–4470 (2016). https://doi.org/10.1007/s00018-016-2300-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-016-2300-4