Abstract
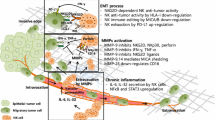
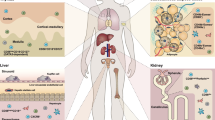
Natural killer (NK) cells are large granular lymphocytes of the innate immune system, responsible for direct targeting and killing of both virally infected and transformed cells. Under pathological conditions and during inflammation, NK cells extravasate into the lymph nodes and accumulate at inflammatory or tumor sites. The activation of NK cells depends on an intricate balance between activating and inhibitory signals that determines if a target will be susceptible to NK-mediated lysis. Many experimental evidences indicate that NK cells are also involved in several immunoregulatory processes and have the ability to modulate the adaptive immune responses. Many other important aspects about NK cell biology are emerging in these last years. The aim of this review is to elucidate the role of NK cells in tumor angiogenesis and their interaction with mast cells. In fact, it has been observed that NK cells produce pro-angiogenic factors and participate alone or in cooperation with mast cells to the regulation of angiogenesis in both physiological and pathological conditions including tumors.
Similar content being viewed by others
References
Miller JS, Verfaillie C, McGlave P. The generation of human natural killer cells from CD34+/DR− primitive progenitors in long-term bone marrow culture. Blood. 1992;80(9):2182–7.
Bennett IM, Zatsepina O, Zamai L, Azzoni L, Mikheeva T, Perussia B. Definition of a natural killer NKR-P1A+/CD56-/CD16- functionally immature human NK cell subset that differentiates in vitro in the presence of interleukin 12. J Exp Med. 1996;184(5):1845–56.
Moretta A, Bottino C, Vitale M, Pende D, Cantoni C, Mingari MC, Biassoni R, Moretta L. Activating receptors and coreceptors involved in human natural killer cell-mediated cytolysis. Annu Rev Immunol. 2001;19:197–223.
Trinchieri G. Biology of natural killer cells, advances in immunology. Amsterdam: Elsevier; 1989. pp. 187–376.
Freud AG, Caligiuri MA. Human natural killer cell development. Immunol Rev. 2006;214:56–72.
Timonen T, Saksela E. Isolation of human NK cells by density gradient centrifugation. J Immunol Methods. 1980;36(3–4):285–91.
Young JD-E, Cohn ZA. Cellular and humoral mechanisms of cytotoxicity: structural and functional analogies. Adv Immunol. 1987;41:269–332.
Carrega P, Ferlazzo G. Natural killer cell distribution and trafficking in human tissues, Front Immunol 3 (2012).
Cooper MA. Human natural killer cells: a unique innate immunoregulatory role for the CD56bright subset. Blood. 2001;97(10):3146–51.
Moretta L, Moretta A. Unravelling natural killer cell function: triggering and inhibitory human NK receptors. EMBO J. 2003;23(2):255–9.
Hayakawa Y, Smyth MJ. Innate immune recognition and suppression of tumors, advances in cancer research, Amsterdam: Elsevier; 2006. pp. 293–322.
Smyth MJ, Hayakawa Y, Takeda K, Yagita H. New aspects of natural-killer-cell surveillance and therapy of cancer. Nat Rev Cancer. 2002;2(11):850–61.
Sullivan JL, Woda BA. X-linked lymphoproliferative syndrome. Immunodefic Rev. 1989;1(4):325–47.
Hanna J, Goldman-Wohl D, Hamani Y, Avraham I, Greenfield C, Natanson-Yaron S, Prus D, Cohen-Daniel L, Arnon TI, Manaster I, Gazit R, Yutkin V, Benharroch D, Porgador A, Keshet E, Yagel S, Mandelboim O. Decidual NK cells regulate key developmental processes at the human fetal-maternal interface. Nat Med. 2006;12(9):1065–74.
Hanna J, Mandelboim O. When killers become helpers. Trends Immunol. 2007;28(5):201–6.
Ratsep MT, Felker AM, Kay VR, Tolusso L, Hofmann AP, Croy BA. Uterine natural killer cells: supervisors of vasculature construction in early decidua basalis. Reproduction. 2014;149(2):R91–102.
Krzywinska E, Kantari-Mimoun C, Kerdiles Y, Sobecki M, Isagawa T, Gotthardt D, Castells M, Haubold J, Millien C, Viel T, Tavitian B, Takeda N, Fandrey J, Vivier E, Sexl V, Stockmann C. Loss of HIF-1α in natural killer cells inhibits tumour growth by stimulating non-productive angiogenesis, Nat Commun 8(1) (2017).
Lee H, Schlereth SL, Park EY, Emami-Naeini P, Chauhan SK, Dana R. A novel pro-angiogenic function for interferon-γ–secreting natural killer cells. Investig Opthalmol Vis Sci. 2014;55(5):2885.
Hoeres T, Wilhelm M, Smetak M, Holzmann E, Schulze-Tanzil G, Birkmann J. Immune cells regulate VEGF signalling via release of VEGF and antagonistic soluble VEGF receptor-1. Clin Exp Immunol. 2018;192(1):54–67.
Lee JY, Lee M, Lee SK. Role of endometrial immune cells in implantation. Clin Exp Reprod Med. 2011;38(3):119.
Carmeliet P, Tessier-Lavigne M. Common mechanisms of nerve and blood vessel wiring. Nature. 2005;436(7048):193–200.
Zhang J, Adams MA, Croy BA. Alterations in maternal and fetal heart functions accompany failed spiral arterial remodeling in pregnant mice. Am J Obstet Gynecol. 2011;205(5):485 e1–16.
Blois SM, Klapp BF, Barrientos G. Decidualization and angiogenesis in early pregnancy: unravelling the functions of DC and NK cells. J Reprod Immunol. 2011;88(2):86–92.
Leonard S, Murrant C, Tayade C, Vandenheuvel M, Watering R, Croy B. Mechanisms regulating immune cell contributions to spiral artery modification—facts and hypotheses—a review. Placenta. 2006;27:40–6.
Bruno A, Ferlazzo G, Albini A, Noonan DM. A think tank of TINK/TANKs: Tumor-infiltrating/tumor-associated natural killer cells in tumor progression and angiogenesis. J Natl Cancer Inst. 2014;106(8):1–13.
Bruno A, Focaccetti C, Pagani A, Imperatori AS, Spagnoletti M, Rotolo N, Cantelmo AR, Franzi F, Capella C, Ferlazzo G, Mortara L, Albini A, Noonan DM. The proangiogenic phenotype of natural killer cells in patients with non-small cell lung cancer. Neoplasia. 2013;15(2):133-IN7.
Bruno A, Bassani B, D’Urso DG, Pitaku I, Cassinotti E, Pelosi G, Boni L, Dominioni L, Noonan DM, Mortara L, Albini A. Angiogenin and the MMP9–TIMP2 axis are up-regulated in proangiogenic, decidual NK-like cells from patients with colorectal cancer. FASEB J. 2018:fj.201701103R. https://doi.org/10.1096/fj.201701103R.
Yao L, Sgadari C, Furuke K, Bloom ET, Teruya-Feldstein J, Tosato G. Contribution of natural killer cells to inhibition of angiogenesis by interleukin-12. Blood. 1999;93(5):1612–21.
Trinchieri G, Rengaraju M, D’Andrea A, Valiante NM, Kubin M, Aste M, Chehimi J. Producer cells of interleukin 12. Parasitol Today. 1993;9(3):97.
Cavallo F, Quaglino E, Cifaldi L, Di Carlo E, Andre A, Bernabei P, Musiani P, Forni G, Calogero RA. Interleukin 12-activated lymphocytes influence tumor genetic programs. Cancer Res. 2001;61(8):3518–23.
Gotthardt D, Putz EM, Grundschober E, Prchal-Murphy M, Straka E, Kudweis P, Heller G, Bago-Horvath Z, Witalisz-Siepracka A, Cumaraswamy AA, Gunning PT, Strobl B, Mu ller M, Moriggl R, Stockmann C, Sexl V. STAT5 is a key regulator in NK cells and acts as a molecular switch from tumor surveillance to tumor promotion. Cancer Discov. 2016;6(4):414–29.
Ribatti D. Mast cells and macrophages exert beneficial and detrimental effects on tumor progression and angiogenesis. Immunol Lett. 2013;152(2):83–8.
Gruber BL, Marchese MJ, Kiely J, Schwartz LB, Schecter NM. 520 Human mast cell products degrade connective tissue matrix. J Allergy Clin Immunol. 1988;81(1):298.
Detoraki A, Staiano RI, Granata F, Giannattasio G, Prevete N, de Paulis A, Ribatti D, Genovese A, Triggiani M, Marone G. Vascular endothelial growth factors synthesized by human lung mast cells exert angiogenic effects. J Allergy Clin Immunol. 2009;123(5):1142–9.e5.
Ribatti D, Crivellato E, Candussio L, Nico B, Vacca A, Roncali L, Dammacco F. Mast cells and their secretory granules are angiogenic in the chick embryo chorioallantoic membrane. Clin Exp Allergy. 2001;31(4):602–8.
Norrby K, Jakobsson A, Sörbo J. Mast-cell-mediated angiogenesis: a novel experimental model using the rat mesentery. Virchows Arch B Cell Pathol Incl Mol Pathol. 1986;52(1):195–206.
Norrby K, Jakobsson A, Sörbo J. Mast-cell secretion and angiogenesis, a quantitative study in rats and mice. Virchows Arch B Cell Pathol Incl Mol Pathol. 1989;57(1):251–6.
Ribatti D, Crivellato E. Mast cells, angiogenesis, and tumour growth. Biochim Biophys Acta. 2012;1822(1):2–8.
Williams NS, Moore TA, Schatzle JD, Puzanov IJ, Sivakumar PV, Zlotnik A, Bennett M, Kumar V. Generation of lytic natural killer 1.1+, Ly-49-cells from multipotential murine bone marrow progenitors in a stroma-free culture: definition of cytokine requirements and developmental intermediates. J Exp Med. 1997;186(9):1609–14.
Kusaka Y, Sato K, Zhang Q, Morita A, Kasahara T, Yanagihara Y. Association of natural killer cell activity with serum IgE. Int Arch Allergy Immunol. 1997;112(4):331–5.
Arase N, Arase H, Hirano S, Yokosuka T, Sakurai D, Saito T. IgE-mediated activation of NK cells through Fc RIII. J Immunol. 2003;170(6):3054–8.
Meyer N, Woidacki K, Knöfler M, Meinhardt G, Nowak D, Velicky P, Pollheimer J, Zenclussen AC. Chymase-producing cells of the innate immune system are required for decidual vascular remodeling and fetal growth. Sci Rep. 2017;22(7):45106.
St. John AL, Rathore APS, Yap H, Ng ML, Metcalfe DD, Vasudevan SG, Abraham SN. Immune surveillance by mast cells during dengue infection promotes natural killer (NK) and NKT-cell recruitment and viral clearance. 2011;Proc Natl Acad Sci 108(22):9190–9195.
Burke SM, Issekutz TB, Mohan K, Lee PWK, Shmulevitz M, Marshall JS. Human mast cell activation with virus-associated stimuli leads to the selective chemotaxis of natural killer cells by a CXCL8-dependent mechanism. Blood. 2008;111(12):5467–76.
Portales-Cervantes L, Haidl ID, Lee PW, Marshall JS. Virus-infected human mast cells enhance natural killer cell functions. J Innate Immunity. 2016;9(1):94–108.
Vosskuhl K, Greten TF, Manns MP, Korangy F, Wedemeyer J. Lipopolysaccharide-mediated mast cell activation induces IFN- secretion by NK cells. J Immunol. 2010;185(1):119–25.
Huang B, Lei Z, Zhang GM, Li D, Song C, Li B, Liu Y, Yuan Y, Unkeless J, Xiong H, Feng ZH. SCF-mediated mast cell infiltration and activation exacerbate the inflammation and immunosuppression in tumor microenvironment. Blood. 2008;112(4):1269–79.
Oldford SA, Haidl ID, Howatt MA, Leiva CA, Johnston B, Marshall JS. A critical role for mast cells and mast cell-derived IL-6 in TLR2-mediated inhibition of tumor growth. J Immunol. 2010;185(11):7067–76.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Bernhard Gibbs.
Rights and permissions
About this article
Cite this article
Ribatti, D., Tamma, R. & Crivellato, E. Cross talk between natural killer cells and mast cells in tumor angiogenesis. Inflamm. Res. 68, 19–23 (2019). https://doi.org/10.1007/s00011-018-1181-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-018-1181-4