Abstract
Primary aromatic amines (PAAs) may be found in food contact materials and can potentially migrate from packaging material into the packed foodstuffs. Certain members of this substance class are of particular concern due to their toxicological properties. Legislators and authorities require a risk assessment regarding the transfer of PAAs into food. The evaluation of PAAs’ migration into food relies on migration or extraction tests using aqueous and acidic food simulants or solvents. However, PAAs exhibit stability issues in acidic media, which could lead to an underestimation of migration. Therefore, a systematic stability study was conducted for 28 PAAs in 3% acetic acid and water at different storage conditions. Standard solutions of 2 and 10 ng/ml of the amines in these media were prepared and stored under the following conditions: 2 h at 40 °C, 70 °C, and 100 °C, and 10 days at − 20 °C, 4 °C, 20 °C, 40° C, and 60 °C. The test solutions were analyzed using liquid chromatography coupled to mass spectrometry, and recovery was calculated against freshly prepared standard solutions. It was found that several PAAs undergo a loss of recovery under the investigated conditions; 7 of the substances were identified to be the most unstable: 3,3ʹ-dimethoxybenzidine, 3,3ʹ-dimethylbenzidine, 6-methoxy-m-toluidine, 4-methoxy-m-phenylendiamine, 2,6-toluenediamine, benzidine and 2,4-toluenediamine. It was concluded that both time and storage temperature play a pivotal role in the stability of PAAs. The experiments also demonstrated that PAAs exhibit greater stability in water than in 3% acetic acid.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
PAAs may unintentionally exist in food contact materials and articles as residues or by-products from the synthesis of azo dyes and other pigments or may be released through reductive cleavage under specific conditions (Aznar et al. 2009). The reduction of the azo bond can occur through various mechanisms, catalyzed either by chemicals or enzymes. Additionally, PAAs can result from the hydrolysis of unreacted isocyanates in aqueous food or food simulants from polyurethane (PU) adhesives or other PUs that haven’t undergone complete curing (Campanella et al. 2015). When utilized in food contact applications, PAAs have the potential to migrate from the packaging materials into food, making these materials a potential source of consumer exposure to PAAs.
PAAs have drawn significant attention from legislators and authorities because of their toxicological properties. Certain members of this group are classified as human carcinogens (EC 2008; BfR 2013). The transfer of PAAs from consumer goods into food items must, therefore, undergo a risk assessment to safeguard human health. In terms of food contact legislation, restrictions for PAA migration have been outlined in Regulation (EU) No 10/2011 (EU 2011). This regulation applies to plastic materials and articles intended for contact with food. Prior to the 15th amendment to this regulation in 2020, Annex II specified that plastic materials and articles should not release PAAs in detectable quantities into food or food simulants, with a detection limit of 0.01 mg per kg of food or food simulant. This detection limit applied to the sum of PAAs not listed in Table 1 of Annex I. However, with the amendment Regulation (EU) No 2020/1245 (EU 2020) these rules were strengthened. It was stipulated that the migration of the PAAs listed in Annex XVII Annex 8 to entry 43 of Regulation (EC) No 1907/2006 (EC 2006) should not be detectable for each individual amine, with a detection limit of 0.002 mg/kg food (simulant). The provided list of PAAs corresponds to the most problematic substances regarding carcinogenicity. The lower detection limit can be attributed to advancements in instrumental analytics, which now enable more sensitive and selective detection.
From an analytical compliance testing perspective, the potential transfer of PAAs from a food contact material to food is assessed through migration tests using food simulants or through extraction experiments. The possible migration is calculated based on the levels determined in the material, assuming total transfer (worst case scenario), or by employing mathematical migration modelling. The specific migration conditions applied for laboratory testing depend on the intended application of the respective materials, and are outlined for plastic materials in Regulation (EU) No 10/2011. Generally, the official food simulant 3% acetic acid is considered the worst-case simulant for PAA migration (JRC 2016). It is known that PAAs are not stable in fat simulants, as they may react with fatty acid components to form to the corresponding amides (Paseiro-Cerrato et al. 2014). Consequently, the testing is typically conducted in aqueous or acidic food simulants.
Advancements in selective separation technologies such as liquid chromatography (LC) and sensitive mass spectrometry (MS) have made it possible to analyze various PAAs using a single LC–MS method (Mortensen et al. 2005). Many researchers have successfully optimized multi-analyte methods for PAAs in the last decade, using reversed phase material columns and MS in single ion mode or multiple reaction mode (MRM) (Aznar et al. 2009; Yavuz et al. 2016; Pezo et al. 2012; Szabó et al. 2021; Devreux et al. 2020). However, internal studies (Hoppe et al. 2022; IVLV 2022) have shown that certain PAAs are not stable in the food simulant 3% acetic acid, also recently reported by (Szabó et al. 2022). Szabó's study included stability testing in various food simulants such as 3% acetic acid, 10% ethanol, 20% ethanol and 50% ethanol and additionally in 3 mmol/L hydrochloric acid.
The stability aspect holds significant importance in food regulatory compliance assessment. Instability in the simulant under migration contact conditions may lead to an underestimation of migration, potentially resulting in food contact articles being evaluated compliant even though migration of PAAs into real food cannot be ruled out. Therefore, there is a need to collect data on the stability of PAAs under migration and extraction conditions, as well as during storage in food simulants or solvents. In this study, the stability of the 22 PAAs listed in Regulation (EC) No 1907/2006 was systematically investigated, including aniline and isomers of toluene diamine (TDA) and methylenedianiline (MDA). These experiments were conducted using 3% acetic acid and water.
2 Material and methods
2.1 Chemicals and reagents
Aniline (CAS No 62–53-3; ≥ 99.9% purity) and 2,6-diaminotoluene (2,6-TDA, CAS No 823–40-5; > 99,9% purity) were purchased from Sigma-Aldrich (Taufkirchen, Germany), 2,2ʹ-methylenedianiline (2,2ʹ-MDA, CAS No 6582–52-1; > 95% purity) was purchased from Enamine Ltd. (Kiev, Ukraine) and 2,4ʹ-methylenedianiline (2,4ʹ-MDA, CAS No 1208–52-2; > 99.2% purity) was purchased from Ambeed, Inc (Arlington Heights, IL, USA). A PAA standard mix ‘Aryl Amine Mix’ (Catalog No: AE-00049-R1; 10 µg/ml in ethyl acetate) was purchased from Techlab (Metz, France). This commercially available mixture already contains the following PAAs that were analyzed in the present study: 4-aminobiphenyl (CAS No 92–67-2), benzidine (CAS No 92–87-5), 4-chloro-o-toluidine (CAS No 95–69-2), 2-naphthylamine (CAS No 91–59-8), o-aminoazotoluene (CAS No 97–56-3), 4-chloroaniline (CAS No 106–47-8), 4,4ʹ-Methylenedianiline (4,4ʹ-MDA, CAS No 101–77-9), 3,3ʹ-dichlorobenzidine (CAS No 91–94-1), 3,3ʹ-dimethoxybenzidine (CAS No 119–90-4), 3,3ʹ-dimethylbenzidine (CAS No 119–93-7), 4,4ʹ-methylenedi-o-toluidine (CAS No 838–88-0), 6-methoxy-m-toluidine (CAS 120–71-8), 4,4ʹ-methylene-bis-(2-chloro-aniline) (CAS No 101–14-4), 4,4ʹ-oxydianiline (CAS No 101–80-4), 4,4ʹ-thiodianiline (CAS No 139–65-1), o-toluidine (CAS No 95–53-4), 2,4-toluenediamine (2,4-TDA, CAS No 95–80-7), 2,4,5-trimethylaniline (CAS No 137–17-7), o-anisidine (CAS No 90–04-0), 4-amino-azobenzene (CAS No 60–09-3), 3-chloro-o-toluidine (CAS No 87–60-5), 2-aminobiphenyl (CAS No 90–41-5). Acetonitrile (ACN) LC–MS grade, methanol LC–MS grade, acetic acid per analysis and formic acid per analysis were purchased from Merck (Darmstadt, Germany). Highly purified water from a TKA GenPure water purification system from Wasseraufbereitungssysteme GmbH (Niederelbert, Germany) was used in all procedures.
Standard stock solutions of the 4 individual PAAs, including aniline, 2,6-TDA, 2,2ʹ-MDA and 2,4ʹ-MDA, were prepared in methanol and combined with the ‘aryl amine mix’ at a concentration of 1 µg/ml in methanol. This internal ‘PAA mix standard’ contained 28 PAAs and was stored at – 20 °C. Dilutions in 3% acetic acid and water for stability tests and calibration solutions were freshly prepared each day.
2.2 Stability testing
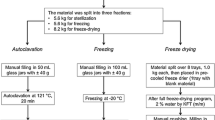
Freshly diluted standard solutions with concentrations of 2 and 10 ng/ml per amine were stored in glass vials (1.5 ml) at different time–temperature conditions (Table 1). The dilutions were prepared independently in each testing vial. The concentration 2 ng/ml was selected as it aligns with the limit of detection for single PAAs in migration experiments, as defined by the European food contact legislation. Hence, the stability of the target analytes at this concentration level holds significant importance. Additionally, the chosen concentration of 10 ng/ml in the sample solution represents a slightly higher concentration that may, in some cases, be encountered in migration experiments.
The choice of time and temperature combinations was based on commonly used migration testing conditions for long-term storage and hot contact, according to Regulation (EU) No 10/2011 (EU 2011). These conditions also account for potential storage scenarios during shipment or storage in the laboratory. For instance, shipment of sample solutions may be necessary when the packaging producer conducts the migration/extraction contact and subsequently ships the solutions to an external analytical laboratory for analysis.
2.3 LC–MS analysis
The chromatographic separation of the PAAs in the ‘PAA mix standard’ was carried out on a Dionex Ultimate 3000 HPLC system from Thermo Fisher Scientific (San Jose, CA, USA) equipped with a quaternary pump, an autosampler and a column oven. As stationary phase a Synergi Polar (150 × 2 mm, 4 µm) column from Phenomenex (Aschaffenburg, Germany) was used with a mobile phase of 0.1% formic acid in ACN (A) and 0.1% formic acid in water (B) with the gradient program shown in Table 2.
The injection volume was 20 μL and column oven temperature was held at 45 °C during the chromatographic run. To separate the TDA-isomers, a SB C3 (250 × 3 mm, 5 µm) from Agilent (Santa Clara, CA, USA) was used, with the gradient program summarized in Table 3.
The LC–MS detection was performed in the multiple reaction monitoring mode (MRM). MRM traces of the analysed PAAs are listed in Table 4. The defined limit of quantification (LOQ) signal to noise ratio was ≥ 10. The LOQs were deduced from the levels of the calibration (0.2–1 ng/ml). For some amines, the LOQ was even lower than 0.2 ng/ml. The LOQs of the different analytes are summarized in Table 4.
The recovery (R in %) was calculated by comparing the analyzed concentration of the stored samples to the spiked concentration, using the following equation:
Standard deviations were determined based on replicates of a single concentration for each time–temperature condition per simulant. In most cases, the standard deviation closely matched the standard deviation of LC–MS analysis for TDA and MDA in our laboratory, which are between 20 – 40% (k = 2). The US-American Food and Drug Administration (FDA) requirements for recovery and standard deviation for standard addition experiments were followed to categorize the obtained recovery values (FDA 2007). PAAs showing recoveries between 70–120% and a standard deviation of < 20% were considered as stable.
3 Results and discussion
3.1 LC–MS method
In this study, a targeted multi-analyte LC–MS/MS method was developed for 28 PAAs of interest (Table 2). The established method effectively separates the 28 PAAs, except for 2 TDA-isomers for which baseline separation was not achieved. To separate these 2 isomers, a different stationary phase with an altered gradient program can be used (Table 3). However, most of the stability analyses were conducted using the presented method with a Synergi Polar column, which does not baseline-separate the TDA-isomers, but uses less solvent. ACN was chosen as organic phase instead of methanol, due to the improved sensitivity achieved using ACN, particularly for 2-Methyl-5-nitroaniline (improved by factor 3). The chosen conditions allowed to achieve a LOQ of < 2 ng/ml for each individual amine in the measurement solution. This LOQ falls below the required detection limit of 2 µg/kg food, as per 15th amendment of Regulation EU 10/2011 (assuming 100 ml simulant and a contact area of 1 dm2 in the migration experiment, and assuming that 1 kg food is packed in a cube with 1 dm side length). Extracted MRM traces for each PAA in 3% acetic acid with a concentration of 1 ng/ml are shown in Fig. S1 (Supplementary Material). The respective chromatograms of the standards in water are depicted in Figure S2 (Supplementary Material).
3.2 Stability of PAAs in 3% acetic acid
3.2.1 General results and considerations
To investigate the stability of various PAAs, standard solutions of the ‘PAA mix standard’ were stored in glass vials under different conditions (Table 1). From the analysed and the (expected) spiked concentration, a recovery and standard deviation was calculated for all samples. PAAs showing recoveries between 70–120% and a standard deviation of < 20% were considered as stable. An overview of the recovery of all test conditions in 3% acetic acid is shown in Fig. 1. The numbers in the bars represent the numbers of PAAs with the corresponding recovery range. To simplify this overview, recoveries of both concentrations (2 and 10 ng/ml) were summarized, a detailed listing of the recoveries and standard deviations can be found in Tables S1–8 (Supplementary Material).
The exact mechanism responsible for the low recovery values observed for certain amines and under specific test conditions remains unknown. Several plausible explanations include amine degradation into smaller molecules, volatilization, rearrangement reactions or the potential accumulation of the amines on the glass vessel walls used for analytical testing or storage. The fate of the amines in the stored solutions is subject to further investigations. Additionally, it's noteworthy that the PAAs showing recoveries < 70% under the experimental conditions were also characterized by standard deviations > 20%. This suggests that the underlying mechanism leading to PAA instability is not completely reproducible, and the recovery rates may experience fluctuations.
3.2.2 Test condition: 40 °C, 70 °C and 100 °C for 2 h
In the industry, a testing protocol has been established to assess food regulatory compliance of laminates containing a PU adhesive, with testing conditions of 2 h/70 °C in 3% acetic acid (Störmer et al. 2005). Therefore, we selected these starting conditions. However, it was observed that 7 amines were not stable under these conditions, and their mean recoveries at 2 tested concentrations were: 0% for 4-methoxy-m-phenylenediamine, 20% for 3,3ʹ-dimethoxybenzidine, 24% for 2,4-TDA, 32% for 3,3ʹ-dimethylbenzidine, 54% for 2,6-TDA, 59% for 6-methoxy-m-toluidine, and 66% for benzidine. Furthermore, the recoveries showed a high standard variation (> 20%) across repetitions and different concentrations. This suggests that either the degradation or volatilisation of these amines is not a reproducible process, or is sensitive to variations in the testing procedure. Based on these observations, it is likely that the recovery rates for unstable amines may fluctuate significantly. Consequently, it might not be feasible to adjust the obtained migration values to a specific recovery value.
Lowering the testing temperature to 40 °C for 2 h resulted in an improved recovery of several PAAs that exhibited low recovery rates at 70 °C for 2 h. Specifically, at a testing temperature of 40 °C, 3,3ʹ-dimethoxybenzidine showed a recovery of 29% and 4-methoxy-m-phenylenediamine had a recovery of 0%. The other 5 amines, which previously had low recovery rates at 70 °C, showed recoveries > 70% at 40 °C. This clearly demonstrates the influence of temperature on the stability of PAAs. Therefore, testing at 40 °C could be considered as an alternative “mild” testing condition for these amines.
Increasing the temperature to 100 °C in 3% acetic acid for 2 h resulted in a significant decrease of the recovery values compared to the lower temperatures tested. The following PAAs had recoveries < 40% in 3% acetic acid at 100 °C/2 h: 3,3ʹ-dimethoxybenzidine, 3,3ʹ-dimethylbenzidine, 6-methoxy-m-toluidine, 4,4ʹ-thiodianiline, 4-methoxy-m-phenylendiamine, 2,6-TDA, 4,4ʹ-methylenedi-o-toluidine, o-anisidine, 2-naphthylamine, 3,3ʹ-dichlorobenzidine, and benzidine. Additionally, 6 PAA compounds showed a recovery lower than 70% at a concentration of 2 ng/ml, including 4,4ʹ-methylene-bis-(2-chloro-aniline) (0% at 2 ng/ml), 4,4ʹ-MDA (0% at 2 ng/ml), 4,4ʹ-oxydianiline (57% at 2 ng/ml), 2,4ʹ-MDA (0% at 2 ng/ml), 2,4,5-trimethylaniline (13% at 2 ng/ml), 2,2ʹ-MDA (68% at 2 ng/ml). Further investigation is required to determine if the concentration plays a role in the stability of the PAAs under these testing condition.
The experiments demonstrated that reducing the temperature leads to improved recovery values for PAAs. Furthermore, it was evident that conducting tests at high-temperature contact conditions in 3% acetic acid yields very low recoveries. Consequently, migration testing at 100 °C in this medium is not recommended. When testing for this application, it is essential to explore the feasibility of employing alternative conditions. Additionally, the experiments revealed a degree of dependency of the recovery rates on the concentration of the PAAs in the 3% acetic acid solutions.
3.2.3 Test condition: 20 °C, 40 °C, and 60 °C for 10 days
In addition to the 2 h tests, stability experiments were extended to a period of 10 days in accordance to the testing conditions for migration experiments specified by the European Plastics Regulation (EU) No 10/2011. The regulation describes specific accelerated testing conditions at elevated temperature based on the respective food storage conditions. It includes a migration contact of 10 days at 20 °C for long term food contact at frozen conditions, 10 days at 40 °C for refrigerated conditions, and 10 days at 60 °C for room temperature storage exceeding 6 months. Furthermore, conditions of 10 days/20 °C could simulate storage, such as during shipping from the packaging producer to an external laboratory for analysis.
In this study, a PAA standard mix in 3% acetic acid was stored at 20 °C, 40 °C, and 60 °C for 10 days. The number of PAAs with recoveries > 70% decreased as the temperature increased. The following 4 PAAs exhibited recoveries below 70% at both concentrations in 3% acetic acid after 10 days at 20 °C: 45% for 3,3ʹ-dimethoxybenzidine, 33% for 4-methoxy-m-phenylenediamine, 20% for 2,6-TDA, and 17% for 2,4-TDA. Furthermore, 5 amines displayed lower recoveries than 70% at the lower concentration (2 ng/ml) tested. These PAAs consistently showed low recoveries across various testing conditions.
At 40 °C storage temperature for 10 d, the following PAAs showed recoveries < 70%: 65% for 4,4ʹ-methylene-bis-(2-chloro-aniline), 15% for 3,3ʹ-dimethoxybenzidine, 28% for 3,3ʹ-dimethylbenzidine, 12% for 6-methoxy-m-toluidine, 63% for 4,4ʹ-thiodianiline, 0% for 4-methoxy-m-phenylendiamine, 66% for 2,2ʹ-MDA, 0% for 2,6-TDA, 67% for o-anisidine, 56% for benzidine, and 0% for 2,4-TDA. Rising the temperature to 60 °C for 10 days increased the number of PAAs displaying a recovery < 70% even further: 32% for 4,4ʹ-methylene-bis-(2-chloro-aniline), 59% for 4,4ʹ-methylendianiline, 42% for 4,4ʹ-oxydianiline, 0% for 3,3ʹ-dimethoxybenzidine, 5% for 3,3ʹ-dimethylbenzidine, 4% for 6-methoxy-m-toluidine, 40% for 2,4ʹ-MDA, 26% for 4,4ʹ-thiodianiline, 0% for 4-methoxy-m-phenylendiamine, 18% for 2,2ʹ-MDA, 0% for 2,6-TDA, 61% for 4,4ʹ-methylendi-o-toluidine, 36% for o-anisidine, 42% for 2-naphthylamine, 47% for 3,3ʹ-dichlorbenzidine, 18% for benzidine, and 1% for 2,4-TDA.
The experiments once again highlighted the impact of temperature on the stability of PAAs, consistent with the observations from stability testing with a 2-h contact time. Lower temperatures resulted in improved recoveries. Notably, it was demonstrated that under very typical migration testing conditions of 10 days 20 °C, 40 °C or 60 °C to mimic long-term storage, PAAs exhibited stability issues in 3% acetic acid. Comparing the results of the 2-h and 10-day contact times at 40 °C, it can be concluded that the contact time itself is another influential factor, as a higher number of PAAs exhibited recoveries > 70% at the shorter contact time.
3.2.4 Test condition: – 20 °C and 4 °C for 10 days
The stability of PAAs mix standard in 3% acetic acid was also assessed at temperatures of 4 °C and – 20 °C for a period of 10 days to mimic the storage conditions of standards during sample shipment or between laboratory measurements. At – 20 °C, the majority of the analyzed PAAs remained stable, with recoveries falling within the range of 90–120%. However, 3,3ʹ-dimethoxybenzidine and 4-methoxy-m-phenylenediamine were exceptions, with recoveries < 40% at – 20 °C (29% and 21%, respectively).
At 4 °C, the same 2 PAAs also displayed reduced recoveries, with 3,3ʹ-dimethoxybenzidine at 62% and 0% for 4-methoxy-m-phenylendiamine. This indicates that the majority of standard solutions can be stored at – 20 °C and 4 °C for a certain period (10 days), without the need to prepare new standards. This is an important consideration in laboratory work. As for shipping of already prepared samples, both tested temperatures are applicable. However, it is important to keep in mind that the 2 PAAs 3,3ʹ-dimethoxybenzidine and 4-methoxy-m-phenylendiamine still exhibit stability issues under the investigated conditions in 3% acetic acid. This is particularly important for standard preparation and storage. As previously concluded from the experiments carried out at higher temperatures, these amines are not stable under current testing conditions, suggesting that additional shipping might not significantly impact their concentration in a sample that migrates or extracts.
3.3 Stability of PAAs in water
The stability of the PAA mix standard in water was tested under the following conditions: 40 °C/10 days, 70 °C/2 h and 100 °C/2 h. The recovery rates were between 80–120% except for 4-methoxy-m-phenylendiamine at 40 °C/10 days (45%, mean value of both concentrations). The recovery values for all PAAs, as obtained from the experiments in water, are summarized in Tables S9–S12 (Supplementary Material). When comparing the recovery values in water to those in 3% acetic acid, water proved to be more favourable than 3% acetic acid. This also applies at the higher temperature of 100 °C.
4 Conclusion
When storing a PAA mix standard at different time and temperature conditions in 3% acetic acid, recoveries < 70% were found for some analytes compared to the originally spiked concentration. In this study, PAAs with low recovery rates were referred to as "unstable". At a 2 h testing time, 7 PAAs were unstable at 70 °C, 13 PAAs at 100 °C, and 2 PAAs at 40 °C. After 10 days testing, 3 PAA were unstable at 20 °C, 6 PAA at 40 °C and 17 amines at a temperature of 60 °C. Regarding their stability, 7 of the investigated PAAs were identified to be the most “critical” substances (Table S13 (Supplementary Material).
The data presented in this study have underscored the significant influence of both time and storage temperature on the recovery of PAAs. Moreover, the choice of food simulant or solvent is also a crucial factor. In this context, the following key observations should be highlighted:
-
3,3ʹ-dimethoxybenzidine and 3,3ʹ-dimethylbenzidine showed low recoveries at 70 °C and 40 °C, but were more stable at 20 °C/10 d.
-
Examining PAAs such as 6-methoxy-m-toluidine, 2,6-TDA, benzidine, and 2,4-TDA revealed that both temperature and time play significant roles in their stability. For instance, 6-methoxy-m-toluidine showed a better recovery at the lower temperature of 40 °C compared to 70 °C when tested for 2 h, but the mean recovery for both concentrations dropped to < 20% at 40 °C after a 10-day storage period. In contrast, when stored at 20 °C for 10 d, the amine displayed a mean recovery over both concentrations of > 70%.
-
4-Methoxy-m-phenylenediamine was the only unstable amine under any tested contact conditions in 3% acetic acid.
-
At 4 °C and – 20 °C, most amines were stable during the testing period of 10 d. Hence, concerning laboratory work, solutions can be stored at these temperatures for some time, potentially eliminating the need to create fresh standards every time.
-
The recoveries at 40 °C/10 days, 70 °C/2 h and 100 °C/2 h in water was in the range of 80–120% for all investigated amines except for 4-methoxy-m-phenylendiamine at 40 °C/10 days, which is in contrast to the low recoveries in 3% acetic acid. With respect to migration testing, this indicates that using water as an alternative to 3% acetic acid could overcome most of the observed stability issues in 3% acetic acid.
The time and temperature combinations of 10 days/40 °C and 10 days/60 °C, established by legislation as standard conditions for accelerated testing according to EU Regulation for food contact plastics (2011), revealed significant stability issues. This raises the question about whether a food regulatory compliance assessment for PAAs can be reliable based on migration testing under these conditions in 3% acetic acid. The instability of PAAs in 3% acetic acid over a period of 10 days at 40 °C or 60 °C may lead to an underestimation of migration. Consequently, food contact articles may be evaluated as compliant, even though migration of PAAs into real foodstuffs cannot be ruled out.
Finally, it is important to note that the stability tests were conducted using pure standard solutions without additional components. Therefore, it possible that the results may not directly translate to a 3% acetic acid solution in conjunction with a food contact material and other migrating components or a real food contact scenario. It is possible that components from the sample matrix could influence the stability of PAAs. Furthermore, the extent to which results in acidic solutions can mirror the processes in complex real food matrices is not fully understood. For example, amines are known to react with fatty acids and fatty acid esters in oils (Paseiro-Cerrato et al. 2014). However, our experiments performed do provide valuable indications of stability issues of PAAs under certain contact conditions applied for compliance testing, and the potential for underestimating their migration into food. Through our investigation and findings, we also want to raise the awareness on this topic.
References
Aznar M, Canellas E, Nerín C (2009) Quantitative determination of 22 primary aromatic amines by cation-exchange solid-phase extraction and liquid chromatography-mass spectrometry. J Chromatogr A 1216(27):5176–5181. https://doi.org/10.1016/j.chroma.2009.04.096
BfR – German Federal Institute for Risk Assessment (2013) Primary aromatic amines from printed food contact materials such as napkins or bakery bags. Opinion No 021/2014 of 24 July 2013
Campanella G, Ghaani M, Quetti G, Farris S (2015) On the origin of primary aromatic amines in food packaging materials. Trends Food Sci Technol 46:137–143. https://doi.org/10.1016/j.tifs.2015.09.002
Commission Regulation (EU) No 10/2011 of 14 January 2011 on plastic materials and articles intended to come into contact with food. http://data.europa.eu/eli/reg/2011/10/oj. Accessed 9 June 2023
Commission Regulation (EU) 2020/1245 of 2 September 2020 amending and correcting Regulation (EU) No 10/2011 on plastic materials and articles intended to come into contact with food. https://eur-lex.europa.eu/eli/reg/2020/1245/oj. Accessed 09 June 2023
Devreux V, Combet S, Clabaux E, Emmanuelle G, Estelle D (2020) From pigments to coloured napkins: comparative analyses of primary aromatic amines in cold water extracts of printed tissues by LC-HRMS and LC-MS/MS. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 37(11):1985–2010. https://doi.org/10.1080/19440049.2020.1802068
European Commission Joint Research Centre (JRC) (2016) Technical guidelines for compliance testing – Draft for stakeholder consultation. JRC Science and policy Reports
FDA-Food and Drug Administration (2007) Guidance for Industry: Preparation of Premarket Submissions for Food Contact Substances (Chemistry Recommendations). https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-preparation-premarket-submissions-food-contact-substances-chemistry. Accessed 9 June 2023
Hoppe M, Schmid P, Gmeiner M, Stärker C (2022) In: Poster Presentation at the 7th international Symposium on Food Packaging, 3rd to 6th May, 2022: Stability of primary aromatic amines under different test conditions
Industrievereinigung für Lebensmitteltechnologie und Verpackung e.V. (IVLV), Hoppe M, Schmid P, Stärker C (2022): Untersuchung der Stabilität von primären aromatischen Aminen unter verschiedenen Lager- und Migrationsbedingungen. Project report. https://www.ivlv.org/project/stabilitaet-paa/. Accessed 9 June 2023
Mortensen SK, Trier XT, Foverskov A, Petersen JH (2005) Specific determination of 20 primary aromatic amines in aqueous food simulants by liquid chromatography-electrospray ionization-tandem mass spectrometry. J Chromatogr A 1091(1–2):40–50. https://doi.org/10.1016/j.chroma.2005.07.026
Paseiro-Cerrato R, Rodríguez-Bernaldo de Quirós A, Sendón R, Bustos J, Sánchez JJ, López-Hernández J, Paseiro-Losada P (2014) Instability of an aromatic amine in fatty food and fatty food simulant: characterisation of reaction products and prediction of their toxicity. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 32(1):100–109. https://doi.org/10.1080/19440049.2014.975751
Pezo D, Fedeli M, Bosetti O, Nerín C (2012) Aromatic amines from polyurethane adhesives in food packaging: the challenge of identification and pattern recognition using Quadrupole-Time of Flight-Mass SpectrometryE. Anal Chim Acta 756:49–59. https://doi.org/10.1016/j.aca.2012.10.031
Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), establishing a European Chemicals Agency, amending Directive 1999/45/EC and repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC. http://data.europa.eu/eli/reg/2006/1907/2014-04-10. Accessed 9 June 2023
Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006. http://data.europa.eu/eli/reg/2008/1272/oj. Accessed 09 June 2023
Störmer A, Rüter M, Brandl F, Brandsch J (2005) Früherkennung und Sicherung der Konformität isocyanathaltiger, funktioneller Schichten. Abschlussbericht BMBF-Projekt Nr. 033 0347, Deutsches Bundesministerium für Bildung und Forschung
Szabó BS, Jakab P, Hegedűs J, Kirchkeszner C, Petrovics N, Nyiri Z, Bodai Z, Rikker T, Eke Z (2021) Determination of 24 primary aromatic amines in aqueous food simulants by combining solid phase extraction and salting-out assisted liquid–liquid extraction with liquid chromatography tandem mass spectrometry. Microchem J 164:105927. https://doi.org/10.1016/j.microc.2021.105927
Szabó BS, Petrovics N, Kirchkeszner C, Nyiri Z, Bodai Z, Eke Z (2022) Stability study of primary aromatic amines in aqueous food simulants under storage conditions of food contact material migration studies. Food Packag Shelf Life 33(1):100909. https://doi.org/10.1016/j.fpsl.2022.100909
Yavuz O, Valzacchi S, Hoekstra E, Simoneau C (2016) Determination of primary aromatic amines in cold water extract of coloured paper napkin samples by liquid chromatography-tandem mass spectrometry. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 33(6):1072–1079. https://doi.org/10.1016/j.toxlet.2013.05.211
Acknowledgements
The authors gratefully acknowledge the financial support by Industrievereinigung für Lebensmitteltechnologie und Verpackungen e. V. (IVLV) and would like to thank Elba Pajares Alva and Carina Stärker for the analytical work and input to this project.
Funding
Open Access funding enabled and organized by Projekt DEAL. This research program has been implemented with support provided from the Industrievereinigung Lebensmitteltechnologie und Verpackung IVLV, financed under project No. 023/202016.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hoppe, M., Gmeiner, M. & Schmid, P. Examining primary aromatic amines' stability in aqueous food simulants: effects of 3% acetic acid and water test conditions. J Consum Prot Food Saf (2024). https://doi.org/10.1007/s00003-024-01494-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00003-024-01494-9