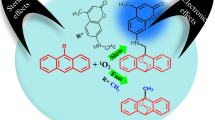
The quantum yield, ΦΔ, of singlet oxygen generation under two-photon excitation has been determined for a fluorene derivative. A photochemical method was developed using 1,3-diphenylisobenzofuran (DPBF), a chemical quencher of 1O2, and 2-(9,9-didecyl-7-nitrofluoren-2-yl)benzothiazole (1) as a two-photon photosensitizer (PS). The photochemical kinetics of the quencher was measured by two different fluorescence methods. Fluorene 1 exhibited relatively high singlet oxygen quantum yield, ΦΔ ≈ 0.4 ± 0.1, and had a two-photon absorption cross-section of 28 ± 5 GM. Thus, 1 may have potential for use as a two-photon PS in the near-IR spectral region for biomedical applications.







Similar content being viewed by others
REFERENCES
B. W. Henderson and T. J. Dougherty (1992). How does photodynamic therapy work? Photochem. Photobiol. 55, 145–147.
M. J. Cook, I. Chambrier, S. J. Cracknell, D. A. Mayes, and D. A. Russell (1995). Octa-alkyl zinc phthalocyanines: Potential photosensitizers for use in the photodynamic therapy of cancer. Photochem. Photobiol. 62, 542–545.
W. Spiller, D. Wohrle, G. Schulz-Ekloff, W. T. Ford, G. Schneider, and J. Stark (1996). Photo-oxidation of sodium sulfide by sulfonated phthalocyanines in oxygen-saturated aqueous solutions containing detergents or latexes. J. Photochem. Photobiol. A 95, 161–173.
J. D. Spikes, J. E. van Lier, and J. C. Bommer (1995). A comparison of the photoproperties of zinc phthalocyanine and zinc naphthalocyanine tetrasulfonates: Model sensitizers for the photodynamic therapy of tumors. J. Photochem. Photobiol. A 91, 193–198.
K. Araki, F. M. Engelmann, I. Mayer, H. E. Toma, M. S. Baptista, H. Maeda, A. Osuka, and H. Furuta (2003). Doubly N-confused porphyrins as efficient sensitizers for singlet oxygen generation. Chem. Lett. 32, 244–245.
J. R. Kanofsky, P. D. Sima, and C. Richter (2003). Singlet-oxygen generation from A2E. Photochem. Photobiol. 77, 235–242.
A. Karotki, M. Kruk, M. Drobizhev, A. Rebane, E. Nickel, and C. W. Spangler (2001). Efficient singlet oxygen generation upon two-photon excitation of new porphyrin with enhanced nonlinear absorption. IEEE J. Sel. Top. Quant. Electron. 7, 971–975.
P. K. Frederiksen, M. Jorgensen, and P. R. Ogilby (2001). Two-photon photosensitized production of singlet oxygen. J. Am. Chem. Soc. 123, 1215–1221.
T. D. Poulsen, P. K. Frederiksen, M. Jorgensen, K. V. Mikkelsen, and P. R. Ogilby (2001). Two-photon singlet oxygen sensitizers: Quantifying, modeling, and optimizing the two-photon absorption cross section. J. Phys. Chem. A 105, 11488–11495.
I. Mac Donald and T. Dougherty (2001). Basic principles of photodynamic therapy. J. Porphyrins Phthalocyanines 5, 105–129.
S. E. Maree, T. Nyokong (2001). Syntheses and photochemical properties of octasubstituted phthalocyaninato zinc complexes. J. Porphyrins Phthalocyanines 5, 782–792.
L. Jiang and Y. He (2001). Photophysics, photochemistry and photobiology of hypocrel-lin photosensitizers. Chin. Sci. Bull. 46, 6–16.
S. Yamaguchi and Y. Sasaki (2001). Spectroscopic determination of very low quantum yield of singlet oxygen formation photosensitized by industrial dyes. J. Photochem. Photobiol. A 142, 47–50.
W. Spiller, H. Kliesch, D. Wohrle, S. Hackbarth, B. Roder, and G. Schnurpfeil (1998). Singlet oxygen quantum yields of different photosensitizers in polar solvents and micellar solutions. J. Porphyrins Phthalocyanines 2, 145–158.
K. D. Belfield, K. S. Schafer, W. Mourad, B. A. Reinhardt (2000). Synthesis of new two-photon absorbing fluorene derivatives via Cu-mediated Ullmann condensations. J. Org. Chem. 65, 4475–4481.
K. D. Belfield, C. C. Corredor, A. R. Morales, M. A. Dessources, and F. E. Hernandez (2005). Synthesis and characterization of new fluorene-based singlet oxygen sensitizers. J. fluoresc. 15(6).
J. R. Lakowicz (1999). Principles of Fluorescence Spectroscopy, Chapter 2, Kluwer Academic/Plenum, New York, Boston, Dordrecht, London, Moscow.
C. S. Foote (1979). In H. H. Wasserman and R. W. Murray (Eds.), Singlet Oxygen, Academic Press, New York, San Francisco, London, pp. 139–171.
F. Wilkinson, D. J. McGarvey, and A. F. Olea (1993). Factors governing the efficiency of singlet oxygen production during oxygen quenching of singlet and triplet states of anthracene derivatives in cyclohexane solution. J. Am. Chem. Soc. 115, 12144–12151.
F. Wilkinson, D. J. McGarvey, and A. F. Olea (1994). Excited triplet state interactions with molecular oxygen: influence of charge transfer on the bimolecular quenching rate constants and the yields of singlet oxygen [O × 2(1Δg)] for substituted naphthalenes in various solvents. J. Phys. Chem. 98, 3762–3769.
R. D. Scurlock, B. J. Wang, and P. R. Ogilby (1996). Chemical reactivity of singlet Σ oxygen (b1Σg+) in solution. J. Am. Chem. Soc. 118, 388–392.
F. Wilkinson and A. A. Abdel-Shafi (1999). Mechanism of quenching of triplet states by molecular oxygen: Biphenyl derivatives in different solvents. J. Phys. Chem. A 103, 5425–5435.
A. A. Abdel-Shafi and F. Wilkinson (2000). Charge transfer effects on the efficiency of singlet oxygen production following oxygen quenching of excited singlet and triplet states of aromatic hydrocarbons in acetonitrile. J. Phys. Chem. A 104, 5747–5757.
C. Schweitzer and R. Schmidt (2003). Physical mechanisms of generation and deactivation of singlet oxygen. Chem. Rev. 103, 1685–1757.
K. D. Belfield, M. V. Bondar, Y. Liu, and O. V. Przhonska (2003). Photophysical and photochemical properties of 5,7-dimethoxycoumarin under one- and two-photon excitation. J. Phys. Org. Chem. 16, 69–78.
M. Sheik-Bahae, A. A. Said, T. H. Wei, D. J. Hagan, and E. W. Van Stryland (1990). Sensitive measurement of optical nonlinearities using a single beam. IEEE J. Quant. Electron. 26, 760–769.
J. R. Lakowicz (1999). Principles of Fluorescence Spectroscopy, Kluwer Academic/Plenum, New York, Boston, Dordrecht, London, Moscow, Chapter 13.
K. J. Schafer, J. M. Hales, M. Balu, K. D. Belfield, E. W. Van Stryland, and D. J. Hagan (2004). Two-photon absorption cross-sections of common photoinitiators. J. Photochem. Photobiol. A 162, 497–502.
M. Rumi, J. E. Ehrlich, A. A. Heikal, J. W. Perry, S. Barlow, Z. Hu, D. McCord-Maughon, T. C. Parker, H. Rolckel, S. Thayumanavan, S. R. Marder, D. Beljonne, and J.-L. Bredas (2000). Structure-property relationships for two-photon absorbing chromophores: Bis-donor diphenylpolyene and bis(styryl)benzene derivatives. J. Am. Chem. Soc. 122, 9500–9510.
ACKNOWLEDGMENTS
We wish to acknowledge the Civilian Research and Development Foundation (UK-C2-2574-MO-04), the donors of The Petroleum Research Fund of the American Chemical Society, the Research Corporation Cottrell College Science program, the National Research Council COBASE award, the Florida Hospital Gala Endowed Program for Oncologic Research, the National Science Foundation, and the University of Central Florida Presidential Initiative for Major Research Equipment for partial support of this work. The authors also wish to thank Dr. Joel M. Hales and Ms. Mihaela Balu for assistance in two-photon absorption measurements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Belfield, K.D., Bondar, M.V. & Przhonska, O.V. Singlet Oxygen Quantum Yield Determination for a Fluorene-Based Two-Photon Photosensitizer. J Fluoresc 16, 111–117 (2006). https://doi.org/10.1007/PL00021939
Published:
Issue Date:
DOI: https://doi.org/10.1007/PL00021939