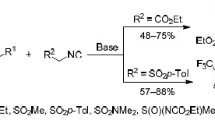
Summary.
The reaction of benzoylacetonitrile, methyl cyanoacetate, and ethyl benzoylacetate with 2-allyloxy- and 2-(3-methyl-2-butenyloxy)-benzaldehyde as well as 2-(3-methyl-2-butenyloxy)-1-naphthaldehyde afforded the corresponding Knoevenagel condensation products. Some of those underwent spontaneous intramolecular cycloaddition to give cis-fused 2H-pyran derivatives as major products. One-pot reactions of 1,3-cyclohexanedione and 5,5-dimethyl-1,3-cyclohexanedione with 2-(3-methyl-2-butenyloxy)-benzaldehyde and 2-(3-methyl-2-butenyloxy)-1-naphthaldehyde resulted in Diels-Alder cycloadducts.
Zusammenfassung.
Reaktion von Benzoylacetonitril, Methylcyanoacetat and Ethylbenzoylacetat mit 2-Allyloxy- und 2-(3-Methyl-2-butenyloxy)-benzaldehyd sowie mit 2-(3-Methyl-2-butenyloxy)-1-naphthaldehyd ergab das entsprechende Knoevenagel-Derivat. Einige dieser letzteren Produkte unterlagen einer spontanen intramolekularen Cycloaddition zu cis-verknüpfen 2H-Pyranderivaten als Hauptprodukt. Eintopfreaktionen von 1,3-Cyclohexandion und 5,5-Dimethyl-1,3-cyclohexandion mit 2-(3-Methyl-2-butenyloxy)-benzaldehyd und 2-(3-Methyl-2-butenyloxy)-1-naphthaldehyd resultierten in Diels-Alder-Cycloaddukten.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received November 2, 1998. Accepted November 19, 1998
Rights and permissions
About this article
Cite this article
Bogdanowicz-Szwed, K., Pałasz, A. Intramolecular Hetero-Diels-Alder Reactions of Functionalized α, β-Unsaturated Carbonyl Compounds: Polycyclic 2H-Pyran Derivatives. Monatshefte fuer Chemie 130, 795–807 (1999). https://doi.org/10.1007/PL00010261
Published:
Issue Date:
DOI: https://doi.org/10.1007/PL00010261