Abstract
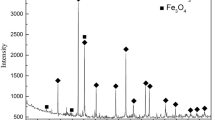
Carbothermic reduction kinetics of ilmenite concentrates catalyzed by sodium chloride and the microwave-absorbing characteristics of the resulting reductive products were investigated; the reduction degree of the reaction was deduced to be \(R = {{4(16y + 56x)(\Delta {W_\Sigma} - {f_{A - P}}W)} \over {7(16y + 56x + 112)}}\). Results show that the activation energy is 112.03 kJ/mol when NaCl is used as catalyst, lowering to 23.11 kJ/mol for the final stage activation energy using sodium silicate as a catalyst. There is a sharp change in the reduction rate constant at temperature ranges of 1,223 K–1,273 K and 1,373 K–1,423 K. Microwave-absorbing characteristics of reductive products were measured by the method of microwave cavity perturbation. It was found that microwave-absorbing characteristics of reductive products obtained at temperatures of 850° C, 1,100° C and 1,150° C show sharp changes. Through X-ray diffraction (XRD) characterization, it was found that the formation of Fe occurs at a temperature of 900° C during the reduction process of ilmenite concentrate. The decrease of FeTiO3 content results in the decrease of microwave-absorbing characteristics, while the increase of Fe content gives rise to the increase of microwave-absorbing characteristics. The decreasing trend is larger than the increasing trend, which is the main reason for the sharp changes in the microwave-absorbing characteristics of reductive products at the temperature range of 850° C to 900° C. When the decreasing trend is smaller than the increasing trend, it becomes the main reason for the sharp changes in microwave-absorbing characteristics of reductive products at the temperature range of 1,100° C to 1,150° C. Kinetics results show that sodium chloride can catalyze the carbothermic reduction of ilmenite, and microwave-absorbing characteristic investigations indicate that microwave heating can be used in the carbothermic reduction of ilmenite.
Similar content being viewed by others
References
Al-Harahsheh, M., and Kingman, S.W., 2004, “Microwave-assisted leaching— a review,” Hydrometallurgy, Vol. 73, No. 3–4, pp. 189–203.
Cutmore, N., Evans, T., Crnokark, D., Middleton, A., and Stoddard, S., 2000, “Microwave technique for analysis of mineral sands,” Minerals Engineering, Vol. 13, No. 7, pp. 729–736.
Dewan, M.A.R., Zhang, G., and Ostrovski, O., 2010, “Carbothermal reduction of a primary ilmenite concentrate in different gas atmospheres,” Metallurgical and Materials Transactions B, Vol. 41B, pp. 182–192.
Dewan, M.A.R., Zhang, G., and Ostrovski, O., 2011, “Carbothermal reduction of ilmenite concentrates and synthetic rutile in different gas atmospheres,” Mineral Processing and Extractive Metallurgy Vol. 120, pp. 111–117.
Guo S.H., Li W., Peng, J.H., Niu, H., Huang, M.Y., Zhang, L.B., and Zhang S.M., Huang M., 2009, “Microwave-absorbing characteristics of mixtures of different carbonaceous reducing agents and oxidized ilmenite,” International Journal of Mineral Processing, Vol.93, No. 3–4, pp. 289–293.
El-Tawil, S.Z., and Morsi, I.M., Francis, A.A., 1993, “Kinetics of solid state reduction of ilmenite ore,” Canadian Metallurgical Quarterly, Vol. 32, No. 4, pp. 281–288.
El-Tawil, S.Z., Morsi, I.M., Yehia, A., and Francis, A.A., 1996, “Alkali reductive roasting of ilmenite ore.,” Canadian Metallurgical Quarterly, Vol. 35, No. 1, pp. 31–37.
Francis A.A., and El-Midany A.A. 2008, “An assessment of the carbothermic reduction of ilmenite ore by statistical design,” Journal of Materials Processing Technology, Vol. 199, No. 1–3, pp. 279–286.
Haque, K.E., 1999, “Microwave energy for mineral treatment processes, a brief review,” International Journal of Mineral Processing, Vol. 57, No. 1, pp. 1–24.
Itoh, S., Suga, T., Takizawa, H., and Nagasaka, T., 2007, “Application of 28 GHz Microwave irradiation to oxidation of ilmenite ore for new rutile extraction process,” ISIJ International, Vol. 47, No. 10), pp. 1416–1421.
Kelly, R.M., and Rowson, N.A., 1995, “Microwave reduction of oxidised ilimenite concentrate,” Minerals Engineering, Vol. 8, No. 11, pp. 1427–1438.
Kingman, S.W., 2006, “Recent developments in microwave processing of minerals,” International Materials Review, Vol. 51, No. 1, pp. 1–12.
Ku, H.S., Siu, F., Siores, E., Bal, J.A.R., and Blicblau, A.S., 2001, “Applications of fixed and variable frequency microwave (VFM) facilities in polymeric materials processing and joining,” Journal of Materials Processing Technology, Vol. 113, pp. 184–188.
Kucukkaragoz, C.S., and Eric, R.H., 2006, “Solid state reduction of a natural ilmenite,” Minerals Engineering, Vol. 19, pp. 334–337.
Lester, E., Kingman, S., Dodds, C., and Patrick, J., 2006, “The potential for rapid coke making using microwave energy,” Fuel, Vol. 85, No. 14–15, pp. 2057–2063.
Li, W., Peng, J.H., Zhang, L.B., Zhang, Z.B., Li, L., Zhang, S.M., and Guo, S.H., 2008, “Pilot-scale extraction of zinc from the spent catalyst of vinyl acetate synthesis by microwave irradiation,” Hydrometallurgy, Vol. 92, No. (1–2), pp. 79–85.
Li, W., Peng, J.H., Zhang, L.B., Yang, K.B., Xia, H.Y., Zhang, S.M., and Guo, S.H., 2009, “Preparation of activated carbon from coconut shell chars in pilot-scale microwave heating equipment at 60 KW,” Waste Management, Vol. 29, No. 2, pp. 756–760.
Mackey T.S., 1994, “Upgrading ilmenite into a high-grade synthetic rutile,” JOM, Vol. 4, pp. 59–64.
Park, E., and Ostrovski, O., 2004, “Effects of preoxidation of titania-ferrous ore on the ore structure and reduction behavior,” ISIJ International, Vol. 44, No. 1, pp. 74–81.
Pickles, C.A., 2009a, “Microwaves in extractive metallurgy: Part 1 — Review of fundamentals,” Minerals Engineering, Vol. 22, No. 13, pp. 1102–1111.
Pickles, C.A., 2009b, “Microwaves in extractive metallurgy: Part 2 — A review of applications,” Minerals Engineering, Vol. 22, No. 13, pp. 1112–1118.
Roberts, J.M.C., 1971, “Ilmenite upgrading,” Mineralogical Magazine, Vol. 125, No. 6, pp. 548.
Sarker, M.K., Rashid, A.K.M.B., and Kurny, A.S.W., 2006, “Kinetics of leaching of oxidized and reduced ilmenite in dilute hydrochloric acid solutions,” International Journal of Mineral Processing, Vol. 80, No. 2–4, pp. 223–228.
Standish, N., and Worner, H., 1990, “Microwave application in the reduction of metal oxides with carbon,” Journal of Microwave Power and Electromagnetic Energy, Vol. 25, No. 3, pp. 177–180.
Suresh, K.T., Rajakumar, V., and Grieveson, P., 1987, “Kinetics of reduction of ilmenite with graphite at 1000 to 1100 °C,” Metallurgical Materials Transanction B: Process metallurgy and materials processing science, 18B, 713–717.
Tong, Z.F., Bi, S.W., and Yang, Y.H., 2004, “Present situation of study on microwave heating application in metallurgy,” Journal of Materials and Metallurgy, Vol. 3, No. 2, pp. 117–120.
Wang, Y.M., and Yuan, Z.F., 2006, “Reductive kinetics of the reaction between a natural ilmenite and carbon,” International Journal of Mineral Processing, Vol. 81, No. 3, pp.133–140.
Welham, N.J., and Williams, J.S., 1999, “Carbothermic reduction of ilmenite (FeTiO3) and rutile (TiO2),” Metallurgical and Materials Transactions B, 30B, pp. 1075–1081.
Wouterlood, H.J., 1979, “The reduction of ilmenite with carbon,” Journal of Chemical Technology and Biotechnology, Vol. 29, pp. 603–618.
Xia, D.K., and Pickles, C.A., 1997, “Applications of microwave energy in extractive metallurgy—a review,” CIM Bulletin, Vol. 90, No. 1011, pp. 96–107.
Xiong, D.A., 2004, “Slon magnetic separators applied in the ilmenite processing industry,” Physical Separation in Science and Engineering, Vol.13, No. 3–4, pp. 119–126.
Zhang, G.Q., and Ostrovski, O., 2002, “Effect of preoxidation and sintering on properties of ilmenite concentrate,” International Journal of Mineral Processing, Vol. 64, No. 4, pp. 201–218.
Author information
Authors and Affiliations
Corresponding author
Additional information
Paper number MMP-12-019.
Discussion of this peer-reviewed and approved paper is invited and must be submitted to SME Publications Dept. prior to November 30, 2013.
Rights and permissions
About this article
Cite this article
Li, W., Peng, J., Guo, S. et al. Carbothermic reduction kinetics of ilmenite concentrates catalyzed by sodium chloride and microwave-absorbing characteristics of reductive products. Mining, Metallurgy & Exploration 30, 108–116 (2013). https://doi.org/10.1007/BF03402413
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03402413