Abstract
OBJECTIVE
Glucocorticoids (GC) are the treatment of choice for moderate-to-severe and active Graves’ orbitopathy (GO), but optimal treatment is still undefined. The aim of the present study was to analyze the efficacy and tolerability of combined parenteral GC pulse therapy followed by oral GC in the interpulse period.
DESIGN
The study included 50 patients (48 ± 10 years; 37 female) with untreated, active and moderate-to-severe GO. Patients received 500mg of methylprednisolone in 500ml of physiologic saline. Infusion was repeated after 48h and then followed by tapering doses of oral prednisone and the cycle repeated each month for the next 5 months. The cumulative dose was 10.2g. Ophthalmic assessment was performed before and 6 months after start of treatment. Side effects of GC therapy were evaluated and recorded each month.
RESULTS
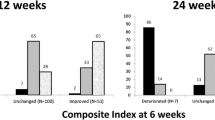
GC showed the greatest effectiveness on soft tissue changes (incorporated in the CAS). Median CAS values decreased from 4.5 to 2 (p>0.001). Improvement was demonstrated in 37 patients (74%), there was no change in 13 patients (26%) and none of the patients presented with deterioration of inflammatory status. Diplopia improved in 21 patients (42%), was unchanged in 28 patients (56%) and deteriorated in 1 patient (2%). Improvement in visual acuity occurred in 36% of patients. At 6 months, 33/50 patients (66%) demonstrated overall treatment response. Response to GC therapy was influenced by CAS, TSHRAb and smoking behavior. The only independent parameter associated with positive treatment response was CAS ≥4 (p<0.001). Side effects occurred in 35/50 patients (70%) and the vast majority of them were mild to moderate. During the 6-months follow-up period, 2/33 patients (6%) had relapsing GO.
CONCLUSION
With appropriate selection of patients and careful monitoring during and after treatment, combined parenteral and oral GC therapy is effective and safe.
Similar content being viewed by others
References
Bahn RS, 2010 Graves ophthalmopathy. N Engl J Med 362: 726–738.
Burch HB, Wartofsky L, 1993 Graves ophthalmopathy: current concepts regarding pathogenesis and treatment. Endocr Rev 14: 747–793.
Bartalena L, Pinchera A, Marcocci C, 2000 Management of Graves ophthalmopathy: reality and perspectives. Endocr Rev 21: 168–199.
Bartalena L, Baldeschi L, Dickinson AJ, et al, 2008 Consensus statement of the European group on Graves orbitopathy (EUGOGO) on management of Graves orbitopathy. Thyroid 18: 336–346.
Zang S, Ponto KA, Kahaly GJ, 2011 Intravenous glucocorticoids for Graves orbitopathy: Efficacy and morbidity. J Clin Endocrinol Metab 96: 320–332.
Bartalena L, Krassas GE, Wiersinga W, 2012 Efficiacy of three different cumulative doses of intravenous methylprednisolone for moderate to severe and active Graves’ orbitopathy. J Clin Endocrinol Metab 97: 4454–4463.
Kendall-Taylor P, Crombie AL, Stephenson AM, et al, 1988 Intravenous methylprednisolone in the treatment of Graves ophthalmopathy. Br Med J 297: 1574–1578.
Dickinson AJ, Perros P, 2001 Controversies in the clinical evaluation of active thyroid associated orbitopathy: use of a detailed protocol with compartice photographs for objective assessment. Clin Endocrinol (Oxf) 55: 283–303.
Bahn RS, Gorman C, 1987 Choice of therapy and criteria for assessing treatment outcome in thyroid-associated ophthalmopathy. Endocrinol Metab Clin N Am 16: 391–407.
Mourits MP, Prummel MF, Wiersinga WM, Koornneef L, 1997 Clinical activity score as a guide in the management of patients with Graves ophthalmopathy. Clin Endocrinol (Oxf) 47: 9–14.
Bartalena L, Marcocci C, Bogazzi F, et al, 1998 Relation between therapy for hyperhyroidism and the course of Graves ophthalmopathy. N Engl J Med 338: 73–78.
Terwee CB, Dekker FW, Mourits MP, et al, 2001 Interpretation and validity of changes in scores on the Graves’ ophalmopathy quality of life questionnaire (GO-QoL) after different treatment. Clin Endocrinol (Oxf) 54: 391–398.
Kendall-Taylor P, Crombie AL, Stephenson AM, et al, 1998 Intravenous methylprednisolone in the tretment of Graves’ ophthalmopathy. Br Med J 297: 1574–1578.
Kaupinen-Makelin R, Karma A, Leinomen E, et al, 2002 High dose intravenous methylprednisolone puis therapy versus oral prednisone for thyroid associated ophthalmopathy. Ophthalmol Scand 80: 316–321.
Marcocci C, Bartalena L, Tanda LM, et al, 2001 Comparison of the effectiveness and tolerability of intravenous or oral glucocorticoids associated with orbital radiotherapy in the management of severe Graves’ ophthalmopathy: results of a prospective, single-blind, randomized study. J Clin Endocrinol Metab 86: 3562–3567.
Kahaly GJ, Pitz S, Hommel G, Dittmar M, 2005 Randomized, single blind trial of intravenous versus oral steroid monotherapy in Graves’ orbitopathy. J Clin Endocrinol Metab 90: 5234–5240.
Utiger RD, 1998 Effects of smoking on thyroid function. Eur J Enocrinol 138: 368–369.
Hegedius L, Brix TH, Vestergaard P, 2004 Relationship between cigaretes smoking and Graves ophthalmopathy. J Endocrinol Invest 27: 265–271.
Le Moli R, Baldeschi I, Saeed P, Regensburg N, Mourits MP, Wiersinga WM, 2007 Determinants of liver damage associated with intravenous methylprednisolone pulse therapy in Graves ophthalmopathy. Thyroid 17: 357–362.
Laurberg P, Berman DC, Bülow Pedersen I, Andersen S, Carlé A, 2012 Incidence and clinical presentation of moderete to severe Graves’ Orbitopathy in a Danish population before and after iodine fortification of salt. J Clin Endocrinol Metab 97: 2325–2332.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beleslin, B.N., Ciric, J., Zarkovic, M. et al. Efficacy and safety of combined parenteral and oral steroid therapy in Graves’ orbitopathy. Hormones 13, 222–228 (2014). https://doi.org/10.1007/BF03401336
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03401336