Abstract
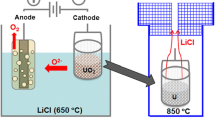
Uranium metal with a purity of about 99.9 pet was produced on a large scale by fused salt electrolysis with a material efficiency of about 90 pet The material efficiency depends mainly on bath composition, electrolysis temperature, electrode smothering, and elutriation during washing of the powder.
Similar content being viewed by others
References
F. H. Driggs and W. C. Lilliendahl: Industrial and Engineering Chemistry, 1930, vol. 22, p. 516.
F. H. Driggs and W. C. Lilliendahl: U. S. Patent No. 1, 861, 625, 1932.
H. D. Smyth: Atomic Energy for Military Purposes, pp. 92–94. Princeton University Press. Princeton, 1946.
W. C. Lilliendahl, D. Wroughton, R. Nagy, and J. W. Marden: U. S. Patent No. 2, 690, 421, 1954.
G. Meister: Metal Progress, 1948, vol. 53, p. 515.
Author information
Authors and Affiliations
Additional information
TP 4604D. Manuscript, Mar. 5, 1956. New York Meeting, February 1956.
Rights and permissions
About this article
Cite this article
Meister, G., Lilliendahl, W.C. Preparation of Uranium Metal by Fused Salt Electrolysis. JOM 9, 1445–1447 (1957). https://doi.org/10.1007/BF03377941
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03377941