Abstract
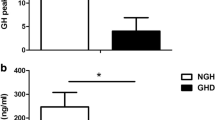
Background and aims: Ghrelin is an orexigenic hormone produced in the stomach and in other organs, exerting a wide range of metabolic functions, including stimulation of GH secretion. Ghrelin secretion is decreased by iv or oral glucose load as well as during euglycemic-hyperinsulinemic clamp and hypoglycemia. We evaluated the circulating ghrelin levels in GH-deficient (GHD) and in GH-sufficient (GHS) patients during GHRH plus arginine test. Materials and methods: The study group comprised 35 patients, including 20 with pituitary tumors, 12 with empty sella, 2 with short stature, and 1 with post-traumatic isolated GH deficiency. According to the results of GHRH plus arginine test, 14 patients were defined as GHD and 21 as GHS. Patients with central hypothyroidism, hypocorticism, and hypogonadism had been on replacement therapy for at least 3 months at the moment of the study. Blood samples were collected every 20 min up to 60 min after GHRH and arginine administration. Results: By definition, GH response to GHRH plus arginine was higher in GHS than GHD group (p<0.0001). Basal serum ghrelin levels were not different in the two groups and did not correlate with body mass index, GH, IGF-I and insulin concentrations. After GHRH plus arginine, serum ghrelin decreased significantly in both groups, with percent decreases ranging 13.3–66.6% in GHD patients (p=0.001) and 7.2–42.2% in GHS patients (p=0.004), with no significant difference in the two groups (p=0.12). Conclusion: Our results show that ghrelin secretion is not modulated by acute GH increase observed in GHS subjects during GHRH plus arginine infusion. The similar decrease of serum ghrelin after GHRH plus arginine stimulation in both GHS and GHD subjects demostrated that there is no negative feedback of GH on ghrelin secretion.
Similar content being viewed by others
References
Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402: 656–60.
Hosoda H, Kojima M, Matsuo H, Kangawa K. Purification and characterization of rat des-Gln14-ghrelin, a second endogenous ligand for the growth hormone secretagogue receptor. J Biol Chem 2000, 275: 21995–2000.
Seoane LM, Tovar S, Baldelli R, et al. Ghrelin elicits a marked stimulatory effect on GH secretion in freely-moving rats. Eur J Endocrinol 2000, 143: R7–9.
Takaya K, Ariyasu H, Kanamoto N, et al. Ghrelin strongly stimulates growth hormone release in humans. J Clin Endocrinol Metab 2000, 85: 4908–11.
Casanueva FF, Dieguez C. Growth hormone secretagogues: physiological role and clinical utility. Trends Endocrinol Metab 1999, 10: 30–8.
Dieguez C, Casanueva FF. Ghrelin: a step forward in the understanding of somatotroph cell function and growth regulation. Eur J Endocrinol 2000, 142: 413–7.
Hataya Y, Akamizu T, Takaya K, et al. A low dose of ghrelin stimulates growth hormone (GH) release synergistically with GH-releasing hormone in humans. J Clin Endocrinol Metab 2001, 86: 4552.
Arvat E, Maccario M, Di Vito L, et al. Endocrine activities of ghrelin, a natural growth hormone secretagogue (GHS), in humans: comparison and interactions with hexarelin, a nonnatural peptidyl GHS, and GH-releasing hormone. J Clin Endocrinol Metab 2001, 86: 1169–74.
Tannenbaum GS, Epelbaum J, Bowers CY. Interrelationship between the novel peptide ghrelin and somatostatin/growth hormone-releasing hormone in regulation of pulsatile growth hormone secretion. Endocrinology 2003, 144: 967–74.
Date Y, Kojima M, Hosoda H, et al. Ghrelin, a novel growth hormone-releasing acylated peptide, is synthesized in a distinct endocrine cell type in the gastrointestinal tracts of rats and humans. Endocrinology 2000, 141: 4255–61.
Wright C, Bartke A. Ghrelin administration decreases insulin secretion in Ames dwarf mice. Proceedings of the 83rd Annual Meeting of the Endocrine Society. 2001, Denver, USA. Abstract P1-98.
Ho KKY; 2007 GH Deficiency Consensus Workshop Partecipants. Consensus guidelines for the diagnosis and treatment of adults with GH deficiency II: a statement of the GH Research Society in association with the European Society for Pediatric Endocrinology, Lawson Wilkins Society, European Society of Endocrinology, Japan Endocrine Society and Endocrine Society of Australia. Eur J Endocrinol 2007, 157: 695–700.
Marzullo P, Di Somma C, Pratt KL, et al. Usefulness of different biochemical markers of the insulin-like growth factor (IGF) Family in diagnosing growth hormone excess and deficiency in adults. J Clin Endocrinol Metab 2001, 86: 3001–8.
Hoffman DM, O’Sullivan AJ, Baxter RC, Ho KK. Diagnosis of growth-hormone deficiency in adults. Lancet 1994, 343: 1064–8.
Tschöp M, Flora DB, Mayer JP, Heiman ML. Hypophysectomy prevents ghrelin-induced adiposity and increases gastric ghrelin secretion in rats. Obes Res 2002, 10: 991–9.
Dall R, Kanaley J, Hansen TK, et al. Plasma ghrelin levels during exercise in healthy subjects and in growth hormone-deficient patients. Eur J Endocrinol 2002, 147: 65–70.
Freda PU, Reyes CM, Conwell IM, Sundeen RE, Wardlaw SL. Serum ghrelin levels in acromegaly: effects of surgical and long-acting octreotide therapy. J Clin Endocrinol Metab 2003, 88: 2037–44.
Torsello A, Scibona B, Leo G, et al. Ontogeny and tissue-specific regulation of ghrelin mRNA expression suggest that ghrelin is primarily involved in the control of extraendocrine functions in the rat. Neuroendocrinology 2003, 77: 91–9.
Janssen JA, van der Toorn FM, Hofland LJ, et al. Systemic ghrelin levels in subjects with growth hormone deficiency are not modified by one year of growth hormone replacement therapy. Eur J Endocrinol 2001, 145: 711–6.
Giavoli C, Cappiello V, Corbetta S, et al. Different effects of short- and long-term recombinant hGH administration on ghrelin and adiponectin levels in GH-deficient adults. Clin Endocrinol 2004, 61: 81–7.
Edén Engström B, Burman P, Holstock C, Karlsson FA. Effects of growth hormone (GH) on ghrelin, leptin and adiponectin in GH-deficient patients. J Clin Endocrinol Metab 2003, 88: 5193–8.
Broglio F, Gottero C, Prodam F, et al. Ghrelin secretion is inhibited by glucose load and insulin-induced hypoglycaemia but unaffected by glucagon and arginine in humans. Clin Endocrinol 2004, 61: 503–9.
Christodoulou C, Schally AV, Chatzistamou I, et al. Expression of growth hormone-releasing hormone (GHRH) and splice variant of GHRH receptors in normal mouse tissues. Regul Pept 2006, 136: 105–8.
Seoane LM, Al-Massadi O, Barreiro F, Dieguez C, Casanueva FF. Growth hormone and somatostatin directly inhibit gastric ghrelin secretion. An in vitro organ culture system. J Endocrinol Invest 2007, 30: RC22–5.
Hosoda H, Kojima M, Kangawa K. Biological, physiological, and pharmacological aspects of ghrelin. J Pharmacol Sci 2006, 100: 398–410.
Kawamata T, Inui A, Hosoda H, Kangawa K, Hori T. Perioperative plasma active and total ghrelin levels are reduced in acromegaly when compared with in non functioning pituitary tumors even after normalization of serum GH. Clin Endocrinol (Oxf) 2007, 67: 140–4.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tarantini, B., Ciuoli, C., Checchi, S. et al. Serum ghrelin levels in growth hormone-sufficient and growth hormone-deficient patients during growth hormone-releasing hormone plus arginine test. J Endocrinol Invest 32, 335–337 (2009). https://doi.org/10.1007/BF03345723
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03345723