Abstract
Dosing convenience is a key element in the effective management of any chronic disease, and is particularly important in the long-term management of osteoporosis. Less frequent dosing with any medication may enhance compliance, thereby maximizing the effectiveness of therapy. Animal data support the rationale that once-weekly dosing with alendronate 70 mg (7 times the daily oral treatment dose) could provide similar efficacy to daily dosing with alendronate 10 mg due to its long duration of effect in bone. In addition, dog studies suggest that the potential for esophageal irritation, observed with daily oral bisphosphonates, may be substantially reduced with once-weekly dosing. This dosing regimen would provide patients with increased convenience and would be likely to enhance patient compliance. We compared the efficacy and safety of treatment with oral once-weekly alendronate 70 mg (N=519), twice-weekly alendronate 35 mg (N=369), and daily alendronate 10 mg (N=370) in a one-year, double- blind, multicenter study of postmenopausal women (ages 42 to 95) with osteoporosis (bone mineral density [BMD] of either lumbar spine or femoral neck at least 2.5 SDs below peak premenopausal mean, or prior vertebral or hip fracture). The primary efficacy endpoint was the comparability of increases in lumbar spine BMD, using strict pre-defined equivalence criteria. Secondary endpoints included changes in BMD at the hip and total body and rate of bone turnover, as assessed by biochemical markers. Both of the new regimens fully satisfied the equivalence criteria relative to daily therapy. Mean increases in lumbar spine BMD at 12 months were: 5.1% (95% CI 4.8, 5.4) in the 70 mg once-weekly group, 5.2% (4.9, 5.6) in the 35 mg twice-weekly group, and 5.4% (5.0, 5.8) in the 10 mg daily treatment group. Increases in BMD at the total hip, femoral neck, trochanter, and total body were similar for the three dosing regimens. All three treatment groups similarly reduced biochemical markers of bone resorption (urinary N-telopeptides of type I collagen) and bone formation (serum bone-specific alkaline phosphatase) into the middle of the premenopausal reference range. All treatment regimens were well tolerated with a similar incidence of upper GI adverse experiences. There were fewer serious upper GI adverse experiences and a trend toward a lower incidence of esophageal events in the once-weekly dosing group compared to the daily dosing group. These data are consistent with preclinical animal models, and suggest that once-weekly dosing has the potential for improved upper GI tolerability. Clinical fractures, captured as adverse experiences, were similar among the groups. We conclude that the alendronate 70 mg once-weekly dosing regimen will provide patients with a more convenient, therapeutically equivalent alternative to daily dosing, and may enhance compliance and long-term persistence with therapy.
Similar content being viewed by others
References
Greenberg R.N.: Overview of patient compliance with medication dosing: a literature review. Clin. Ther. 6: 592–599, 1984.
Lau H.S., Beuning K.S., Postma-Lim E., Klein-Beernink L., de Boer A., Porsius A.J.: Non-compliance in elderly people: evaluation of risk factors by longitudinal data analysis. Pharmacy World Sci. 18: 63–68, 1996.
Venturini F., Nichol M.B., Sung J.C., Bailey K.L., Cody M., McCombs J.S.: Compliance with sulfonylureas in a health maintenance organization: a pharmacy record-based study. Ann. Pharmacother. 33: 281–288, 1999.
Liberman U.A., Weiss S.R., Broll J., Minne H.W., Quan H., Bell N.H., Rodriguez-Portales J., Downs R.W. Jr., Dequeker J., Favus M., Seeman E., Recker R.R.., Capizzi T., Santora A.C. II, Lombardi A., Shah R., Hirsch L.J., Karpf D.B.: Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. N. Engl. J. Med. 333: 1437–1443, 1995.
Black D.M., Cummings S.R., Karpf D.B., Cauley J.A., Thompson D.E., Nevitt M.C., Bauer D.C., Genant H.K., Haskell W.L., Marcus R., Ott S.M., Torner J.C., Quandt S.A., Reiss T.F., Ensrud K. E.: Randomized trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Lancet 348: 1535–1541, 1996.
Cummings S.R., Black D.M., Thompson D.E., Applegate W.B., Barrett-Connor E., Musliner T.A., Palermo L., Prineus R., Rubin S.M., Scott J.C., Vogt T., Wallace R., Yates A.J., LaCroix A.Z.: Effect of alendronate on risk of fracture in women with low bone density but without vertebral fractures. JAMA 280: 2077–2082, 1998.
Pols H.A.P., Felsenberg D., Hanley D.A., Stepan J., Munoz-Torres M., Wilkin T.J., Qin-sheng G., Galich A.M., Vandormael K., Yates A.J., Stych B.: Multinational, placebo-controlled, randomized trial of the effects of alendronate on bone density and fracture risk in postmenopausal women with low bone mass: results of the FOSIT study. Osteoporos. Int. 9: 461–468, 1999.
Hosking D., Chilvers C.E.D., Christiansen C., Ravn P., Wasnich R., Ross P., McClung M., Balske A., Thompson D., Daley M., Yates A.J.: Prevention of bone loss with alendronate in postmenopausal women under 60 years of age. N. Engl. J. Med. 338: 485–492, 1998.
McClung M., Clemmesen B., Daifotis A., Gilchrist N.L., Eisman J., Weinstein R.S., El Hajj Fuleihan G., Reda C., Yates A.J., Ravn P.: Alendronate prevents postmenopausal bone loss in women without osteoporosis: a double-blind, randomized, controlled trial. Ann. Intern. Med. 128: 253–261, 1998.
Bauer D.C., Black D., Ensrud K., Thompson D., Hochberg M., Nevitt M., Musliner T., Freedholm D.: Upper gastrointestinal tract safety profile of alendronate: the Fracture Intervention Trial. Arch. Intern. Med. 2000 (in press).
Seedor J.G., Quartuccio H.A., Thompson D.D.: The bisphosphonate alendronate (MK-217) inhibits bone loss due to ovariectomy in rats. J. Bone Min. Res. 6: 339–346, 1991.
Balena R., Toolan B.C., Shea M., Markatos A., Myers E.R., Lee S.C., Opas E.E., Seedor J.G., Klein H., Frankenfield D., Quartuccio H.A., Fioravanti C., Clair J., Brown E., Hayes W.C., Rodan G.A.: The effects of 2-year treatment with the aminobisphosphonate alendronate on bone metabolism, bone histomorphometry, and bone strength in ovariectomized nonhuman primates. J. Clin. Invest. 92: 2577–2586, 1993.
Rodan G.A., Seedor J.G., Balena R.: Preclinical pharmacology of alendronate. Osteoporos. Int. Suppl. (3): S7–12, 1993.
Khan S.A., Kanis J.A., Vasikaran S., Kline W.F., Matuszewski B.K., McCloskey E.V., Beneton M.N., Gertz B.J., Sciberras D.G., Holland S.D., Orgee J., Coombes G.M., Rogers S.R., Porras A.G.: Elimination and biochemical responses to intravenous alendronate in postmenopausal osteoporosis. J. Bone Miner. Res. 12: 1700–1707, 1997.
Gertz B.J., Holland S.D., Kline W.F., Matuszewski B.K., Porras A.G.: Clinical pharmacology of alendronate sodium. Osteoporos. Int. Suppl. (3): S13–16, 1993.
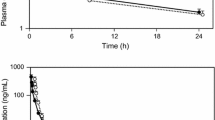
Porras A.G., Holland S.D., Gertz B.J.: Pharmacokinetics of alendronate. Clin. Pharmacokinet. 36: 315–328, 1999.
Lufkin E.G., Argueta R., Whitaker M.D., Cameron A.L., Wong V.H., Egan K.S., O’Fallon W.M., Riggs B.L.: Pamidronate: an unrecognized problem in gastrointestinal tolerability. Osteoporos. Int. 4: 320–322, 1994.
Chestnut C.H., McClung M.R., Ensrud K.E., Bell N.H., Genant H.K., Harris S.T., Singer F.R., Stock J.L., Yood R.A., Delmas P.D., Kher U., Pryor-Tillotson S., Santora A.C.: Alendronate treatment of the postmenopausal osteoporotic woman: effect of multiple dosages on bone mass and bone remodeling. Am. J. Med. 99: 144–152, 1995.
DeGroen P.C., Lubbe D.F., Hirsch L.J., Daifotis A., Stephenson W., Freedholm D., Pryor-Tillotson S., Seleznick M.J., Pinkas H., Wang K.K.: Esophagitis associated with the use of alendronate. N. Engl. J. Med. 335: 1016–1021, 1996.
Siris E.S., Chines A.A., Altman R.D., Brown J.P., Johnston C.C. Jr., Lang R., McClung M.R., Mallette L.E., Miller P.D., Ryan W.G., Singer F.R., Tucci J.R., Eusebio R.A., Bekker P.J.: Risedronate in the treatment of Paget’s disease of bone: an open label, multicenter study. J. Bone Miner. Res. 13: 1032–1037, 1998.
Saunders R.L.: Appearance of a gastric ulcer during diphosphonate therapy in a woman with CRST syndrome. South Med. J. 70: 1327–1329, 1977.
Peter C.P., Handt L.K., Smith S.M.: Esophageal irritation due to alendronate sodium tablets: possible mechanisms. Dig. Dis. Sci. 43: 1998–2002, 1998.
Bone H.G., Adami S., Rizzoli R., Favus M., Ross P.D., Santora A., Prahalada S., Daifotis A., Orloff J., Yates A.J.: Weekly administration of alendronate: rationale and plan for clinical assessment. Clin. Ther. 22: 15–28, 2000.
Black D.M., Palermo L., Nevitt M.C., Genant H.K, Epstein R., San Valentin R., Cummings S.R.: Comparison of methods for defining prevalent vertebral deformities: the study of osteoporotic fractures. J. Bone Miner. Res. 10: 890–902, 1995.
Faulkner K.G., McClung M.R.: Quality control of DXA instruments in multicenter trials. Osteoporos. Int. 5: 218–227, 1995.
Genant H.K., Wu C.Y., van Kuijk C., Nevitt M.C.: Vertebral fracture assessment using a semiquantitative technique. J. Bone Miner. Res. 8: 1137–1148, 1993.
Devogelaer J.P., Broll H., Correa-Rotter R., Cumming D.C., De Deuxchaisnes C.N., Geusens P., Hosking D., Jaeger P., Kaufman J.M., Leite M., Leon J., Liberman U., Menkes C.J., Meunier P.J., Reid I., Rodriguez J., Romanowicz A., Seeman E., Vermeulen A., Hirsch L.J., Lombardi A., Plezia K., Santora A.C., Yates A.J., Yuan W.: Oral alendronate induces progressive increases in bone mass of the spine, hip, and total body over 3 years in postmenopausal women with osteoporosis. Bone 18: 141–150, 1996.
Adami S., Passeri M., Ortolani S., Broggini M., Carratelli L., Caruso I., Gandolini G., Gnessi L., Laurenzi M., Lombardi A., Norbiato G., Pryor-Tillotson S., Reda C., Romanini L., Subrizi D., Wei L., Yates A.J.: Effects of oral alendronate and intranasal salmon calcitonin on bone mass and biochemical markers of bone turnover in postmenopausal women with osteoporosis. Bone 17: 383–390, 1995.
Jones B., Jarvis P., Lewis J.A., Ebbutt A.F.: Trials to assess equivalence: the importance of rigorous methods. Br. Med. J. 313: 36–39, 1996.
ICH Harmonised Tripartite Guideline. Statistical principles for clinical trials. International Conference on Harmonisation E9 Expert Working Group. Stat Med. 18: 1905–1942, 1999.
Storm T., Thamsborg G., Steiniche T., Genant H.K., Sorensen O.H.: Effect of intermittent cyclical etidronate therapy on bone mass and fracture rate in women with postmenopausal osteoporosis. N. Engl. J. Med. 322: 1265–1271, 1990.
Watts N.B., Harris S.T., Genant H.K., Wasnich R.D., Miller P.D., Jackson R.D., Licata A.A., Ross P., Woodson G.C. III, Yanover M.J., Mysiw W.J., Kohse L., Rao M.B., Steiger P., Richmond B., Chestnut C.H.: Intermittent cyclical etidronate treatment of postmenopausal osteoporosis. N. Engl. J. Med. 323: 73–79, 1990.
Marshall D., Johnell O., Wedel H.: Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. B. M. J. 312: 1254–1259, 1996.
Hochberg M., Ross P.D., Black D., Cummings S.R., Genant H.K., Nevitt M.C., Barrett-Connor E., Musliner T., Thompson D.: Larger increases in bone mineral density during alendronate therapy are associated with a lower risk of new vertebral fractures in women with postmenopausal osteoporosis. Arthritis Rheum. 42: 1246–1254, 1999.
Wasnich RD, Miller PD.: Antifracture efficacy of antiresorptive agents are related to changes in bone density. J. Clin. Endocrinol. Metab. 85: 231–236, 2000.
Garnero P., Sornay-Rendu E., Delmas P.D.: Decreased bone turnover in oral contraceptive users. Bone 16: 499–503, 1995.
Chavassieux P.M., Arlot M.E., Reda C., Wei L., Yates A.J., Meunier P.J.: Histomorphometric assessment of the long-term effects of alendronate on bone quality and remodeling in patients with osteoporosis. J. Clin. Invest. 100: 1475–1480, 1997.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
For affiliations and the complete list of participating investigators and Clinical Centers see Appendix.
Rights and permissions
About this article
Cite this article
Schnitzer, T., Bone, H.G., Crepaldi, G. et al. Therapeutic equivalence of alendronate 70 mg onceweekly and alendronate 10 mg daily in the treatment of osteoporosis. Aging Clin Exp Res 12, 1–12 (2000). https://doi.org/10.1007/BF03339822
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03339822