Summary
The influence of food intake on the bioavailability of omeprazole was studied after single and repeated administrations of enteric coated (EC) granules. 12 healthy males participated in an open crossover trial consisting of 3 study periods. Omeprazole 20mg was given as a single intravenous dose and orally as EC granules once daily for 7 days either immediately before or immediately after breakfast (periods 2 and 3). Plasma concentrations of omeprazole were followed after the intravenous dose and on days 1 and 7 in the other periods.
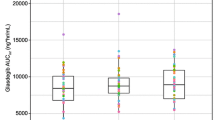
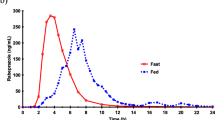
The time for appearance of omeprazole in plasma seemed to be prolonged when the EC granule formulation was given after food intake. The total amount of drug absorbed, however, was not affected. Starting from identical area under the plasma concentration-time curve (AUC) values following the first dose, the AUC increased by approximately 60% during repeated administration when the EC granules were given both before and after breakfast.
Similar content being viewed by others
References
Andersson T, Andrén K, Cederberg C, Lagerström P-O, Lundborg P, et al. Pharmacokinetics and bioavailability of omeprazole after single and repeated oral administration in healthy subjects. British Journal of Clinical Pharmacology 29: 557–563, 1990
Bardhan KD, Bianchi Porro G, Bose K, et al. A comparison of two different doses of omeprazole versus ranitidine in treatment of duodenal ulcers. Journal of Clinical Gastroenterology 8: 408–413, 1986
Fellenius E, Berglindh T, Sachs G. Substituted benzimidazoles inhibit gastric acid secretion by blocking (HK)-ATPase. Nature 290: 159–161, 1981
Grundevik I, Jerndal G, Balmér K, Persson B-A. Fully automated liquid Chromatographic assay of omeprazole and two metabolites by use of gradient elution. Journal of Pharmaceutical and Biomedical Analysis 4: 389–398, 1986
Hetzel DJ, Dent J, Reed WD. Healing and relapse of severe peptic esophagitis after treatment with omeprazole. Gastroenterology 95: 903–912, 1988
Klinkenberg-Knol EC, Jansen JMBJ, Festen HPM, Meuwissen SGM, Lamers CBHW. Double-blind multicentre comparison of omeprazole and ranitidine in the treatment of reflux oesophagitis. Lancet 1: 349–351, 1987
Lind T, Cederberg C, Ekenved G, Haglund U, Olbe L. Effect of omeprazole — a gastric proton pump inhibitor — on pentagastrin stimulated acid secretion in man. Gut 24: 270–276, 1983
Pilbrant Å, Cederberg C. Development of an oral formulation of omeprazole. Scandinavian Journal of Gastroenterology (Suppl. 108): 113–120, 1985
Prichard PJ, Yeomans ND, Mihaly GW, et al. Omeprazole: a study of its inhibition of gastric pH and oral pharmacokinetics after morning or evening dosage. Gastroenterology 88: 64–69, 1985
Walan A, Bader JP, Classen M. Effect of omeprazole and ranitidine on ulcer healing and relapse rates in patients with benign gastric ulcer. New England Journal of Medicine 320: 69–75, 1989
Wallmark B, Lorentzon P, Larsson H. The mechanism of action of omeprazole: a survey of its inhibitory actions in vitro. Scandinavian Journal of Gastroenterology (Suppl. 108): 37–51, 1985
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Andersson, T., Andrén, K., Cederberg, C. et al. Bioavailability of Omeprazole as Enteric Coated (EC) Granules in Conjunction with Food on the First and Seventh Days of Treatment. Drug Invest. 2, 184–188 (1990). https://doi.org/10.1007/BF03259193
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03259193