Summary
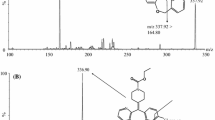
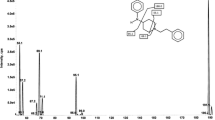
A sensitive and selective liquid chromatography-tandem mass spectrometric (LC-MS) method was developed and validated for the determination of Tramadol in human plasma and urine. The analyte was separated on a Diamonsil C18 column with ammonium acetate( 5mmol·L−1)-methanol(50:50, v:v) adjusted PH by caustic soda at a flow rate of 0.8ml min−1, and analyzed by mass spectrometry is in positive ion mode. The ion mass spectrum of m/z were 264.1 for Tramadol and 248.0 for Tinidazole (I.S.), respectively. The weighted (1/x2) calibration curve was linear over plasma concentration range 1.00–400.00ng/ml and urine concentration range 0.01–16.00 ng/ml, with a correlation coefficient (r) of 0.9995 and 0.9997, respectively. The lower limit of quantification in human plasma was 1.00ng/ml. The inter- and intra-day precisions (CV%) in both plasma and urine were lower than 10%, the mean method accuracies and recoveries from spiked plasma samples at three concentrations ranged from 98.2 to 100.1% and 61.6 to 62.9%, respectively. The developed method was successfully applied to determine Tramadol in human plasma and urine, and provided suitable profiles for clinical pharmacokinetic study of Tramadol.
Similar content being viewed by others
References
Lee CR, McTavish D., Sorkin E.M. (1993): Drugs, 46, 313.
Collart L., Luthy C, Favario-Constantin C, Dayer P., (1993): Schweiz Med., 123, 2241.
Dayer P., Collart L., Desmeules J., (1994): Drugs, 47(Suppll), 3.
Desmeules J.A., Piguet V., Collart L., Dayer P., (1996): Br. J. Clin. Pharmacol., 41, 7.
Driessen B., Reimann W., Giertz H., (1993): Br. J. Pharmacol., 108, 806.
Raffa R.B., Friderichs E., Reimann W., Shank R.P., Codd E.E., Vaught J.L., Jacoby H.I., Selve N., (1993): J. Pharmacol Exp Ther. 267 (1993) 331.
CH. Wilder-Smith, A. Bettiga. Br J Clin Pharmacol., 43, 71.
Driessen B., Reimann W., (1992): Br. J. Pharmacol., 105, 147.
Lintz W, Erlacin S., Frankus, E., Uragg H., (1981): Arzneimittelforschung, 31, 1932.
Subrahmanyam V., Renwick A.B., Walters D.G., Price P.J., Tonelli A.P., Lake B.G., (2001): Drug Metab. Dispos. 29, 1146.
Guidance for industry. Bioanalytical Method Validation, US Department of Health and Human Services, May 2001.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, P., Liang, S., Wang, BJ. et al. Development and validation of a sensitive LC-MS method for the determination of tramadol in human plasma and urine. Eur. J. Drug Metabol. Pharmacokinet. 34, 185–192 (2009). https://doi.org/10.1007/BF03191172
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03191172