Summary
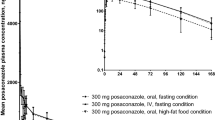
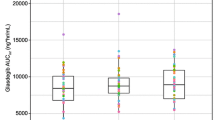
This randomized, six-treatment, six-period, six sequence, single dose, crossover pharmacokinetic study assessed the effect of different types of food on the bioavailability of 500-mg cefaclor extended release tablet in 23 healthy male volunteers. A single dose of cefaclor extended release 500-mg tablet was administered at six occasions: after overnight fasting, after two vegetarian (high-fat and low-fat), two non-vegetarian (high-fat and low-fat) and rice diets. Serial blood samples were collected up to 12 h after dose. Serum cefaclor concentrations were determined by a validated HPLC method. An almost equivalent increase in both Cmax and AUC was observed with both high-fat non-vegetarian and low-fat vegetarian breakfasts. However, when MIC90 values, a pharmacodynamic end-point were compared, the low-fat vegetarian diet fared better than the high-fat non-vegetarian diet. The results obtained favor low-fat vegetarian diet (breakfast) to be taken with cefaclor extended release tablet to achieve maximum benefit in terms of clinical efficacy.
Similar content being viewed by others
References
Sourgens H., Derendorf H., Schifferer H. (1997): Pharmacokinetic profile of cefaclor. Int. J. Clin. Pharm. Ther., 35, 374–380.
Hodges G.R., Liu C., Hinthron D.R., Harms J.L., Dworzack D.L. (1978): Pharmacological Evaluation of Cefaclor in volunteers. Antimicrob. Agents Chemother., 14, 454–456.
Lode H., Fassbender M., Schaberg T. (1994): Comparative pharmacokinetics of the new oral cephalosporins. Durgs, 47 (Suppl. 3), 10–19.
Nix D.E., Symonds W.T., Wilton J.H., Reidenberg P., Teal M.A., O’Connor G. (1994): Comparative pharmacokinetics of oral Ceftibuten, Cefixime, cefaclor and cefuroxime axetil in normal adults. Clin. Pharmacol. Ther., 55, 192–195.
Welling P.G., Dean S., Selen A., Kendall M.J., Wise R. (1979): The pharmacokinetics of the oral cephalosporins cefaclor, cephradine and cephalexin. Int. J. Clin. Pharmacol. Biopharm., 17, 397–400.
Glynne A., Goulborn A., Ryder R. (1978): A human pharmacology study of cefaclor. Antimicrob. Agents Chemother., 4, 343–348.
Haginaka J., Yamaoka K., Kagagama T., Nishimura Y., Uno T. (1979): Evaluation of food ingestion on bioavailability of cephalexin by moment analysis. Chem. Pharm. Bull., 27, 3156–3159.
Lode H., Stahlmann R., Koeppe P. (1979): Comparative pharmacokinetics of cephalexin, cefaclor, Cefadroxil, and CGP 9000. Antimicrob. Agents Chemother., 16, 1–6.
Barbhaiya R.H., Gleason C.R., Shyu W.C., Pittman K.A. (1990): Comparison of the effects of food on the pharmacokinetics of cefprozil and cefaclor. Antimicrob. Agents Chemother., 34, 1210–1213.
McCracken G.H. Jr., Ginsburg C.M., Clahsen J.C., Thomas M.L. (1978): Pharmacologic evaluation of orally administered antibiotics in infants and children: effect of feeding on bioavailability. Pediatrics, 62, 738–743.
Hodges G.R., Liu C., Hinthorn D.R., Harms J.L., Dworzack D.L. (1978): Pharmacological evaluation of cefaclor in volunteers. Antimicrob. Agents Chemother., 14, 454–456.
Finn A., Straughn A., Meyer M. (1987): Effect of dose and food on the bioavailability of cefuroxime axetil. Biopharm. Drug Dispos., 8, 519–526.
Williams P.E., Harding S.M. (1984): The absolute bioavailability of oral cefuroxime axetil in male and female volunteers after fasting and after food. J. Antimicrob. Chemother., 13, 191–196.
Karim S., Ahmed T., Monif T., Saha N., Sharma P.L. (2003): The effect of four different types of food on the bioavailability of cefaclor. Eur. J. Drug Metab. Pharmacokinet., 28, 185–190.
Welling P.G. (1996): Effects of Food on drug absorption. Ann. Rev. Nut., 16, 383–415.
James N.C., Donn K.H., Collins J.J., Davis I.M., Lloyd T.L., Hart R.W., Powell J.R. (1991): Pharmacokinetics of cefuroxime axetil andcefaclor: relationship of concentrations in serum to MICs for common respiratory pathogens. Antimicrob. Agents Chemother., 35, 1860–1863.
Jacobs M.R., Bajaksouzian S., Zilles A., Lin G., Pankuch G.A., Appelbaum P.C. (1999): Susceptibilities of Streptococcus pneumonia and Haemophilus influenzae to 10 oral antimicrobial agents based on pharmacodynamic parameter 1997 U.S. surveillance study. Antimicrob. Agents Chemother., 43, 1901–1908.
Oguma T., Yamada H., Sawaki M., Narita N. (1991). Pharmacokinetic analysis of the effects of different foods on absorption of cefaclor. Antimicrob. Agents Chemother., 35, 1729–1735.
Welling P.G., Tse F.L.S. (1982): The influence of food on the absorption of antimicrobial agents. J. Antimicrob. Chemother., 9, 7–27.
Welling P.G. (1989): Effects of food on drug absorption. Pharmacol. Ther., 43, 425–441.
Gai M.N., Isla A., Andonaegui M.T. (1997): Evaluation of the effect of 3 different diets on the bioavailability of 2 sustained release theophylline matrix tablets. Int. J. Clin. Pharmacol. Ther., 35, 565–571.
Rosen A., Macheras P. (1985): Effect of protein on the absorption of Phenytoin through everted gut preparations. J. Pharm. Pharmacol., 37, 154–158.
Williams L. Hill Jr D.P., Davis J.A., Lowenthal D.T. (1996): The influence of food on the absorption and metabolism of drugs: an update. Eur. J. Drug Metab. Pharmacokinet., 21, 201–211.
Zhi J., Rakhit A., Patel I.H. (1995): Effects of dietary fat on drug absorption. Clin. Pharmacol. Ther., 58, 487–491.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Khan, B.A.H., Ahmed, T., Karim, S. et al. Comparative effect of different types of food on the bioavailability of cefaclor extended release tablet. European Journal of Drug Metabolism and Pharmacokinetics 29, 125–132 (2004). https://doi.org/10.1007/BF03190587
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190587