Summary
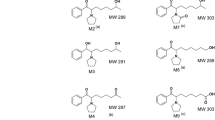
After intraperitoneal administration of procyclidine, eight metabolites were isolated from rat urine. They were identified as l-(4-oxocyclohexyl)-1-phenyl-3-(1-pyrrolidinyl)-1-propanol, 1-(cis-4-hydroxycyclohexyl)-1-phenyl-3-(1-pyrrolidinyl)-1-propanol, 1-(trans-4-hydrocyclohexyl)-1-phenyl-3-(1-pyrrolidinyl)-1-propanol, (1R*,3R*,4S*,7R*)- and ( 1R*,3R*,4S*, 7S*)-1-(cis-3,cis-4-dihydroxy-cyclohexyl)-1-phenyl-3-(1-pyrrolidinyl)-1-propanol, (1R*,3R*,4R*,7R*)- and (1R*,3R*,4R*, 7S*)-1-(cis-3,trans-4-dihydroxycyclo-hexyl)-l-phenyl-3-(l-pyrrolidinyl)-1-propanol, and one of both (1R*, 3S*, 4R*,7R*)- or (1R*, 3S*, 4R*, 7S*)-l-(trans-3, trans-4-dihydroxycyclohexyl)-1-phenyl-3-(l-pyrrolidinyl)-1-propanol by comparative TLC, GLC-MS andl3C-NMR spectroscopy.
Similar content being viewed by others
References
Schwab R.S. and Chafetz M.E. (1955): Kemadrin in the Treatment of Parkinsonism, Neurology,5, 213–211.
Mindham R.H.S., Lamb P. and Bradley R. (1977): A comparison of Piribedil, Procyclidine and Placebo in the control of Phenothiazine-induced Parkinsonism, Brit. J. Psychiat,130, 581–585.
Paeme G. and Vercruysse A., unpublished.
Paeme G., Grimée R. and Vercruysse A. (1982): Carbon-13 Chemical Shifts of Three Oxo-, Six Monohydroxy-, and Six Dihydroxycyclohexyl Derivatives of Procyclidine, Bull. Soc. Chim. Belg.,91, 641–651.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Paeme, G., Grimee, R. & Vercruysse, A. Isolation and identification of eight procyclidine metabolites from rat urine. European Journal of Drug Metabolism and Pharmacokinetics 9, 103–108 (1984). https://doi.org/10.1007/BF03189612
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03189612