Summary
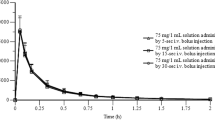
The plasma concentration of the anti-inflammatory drug fenclofenac was investigated in volunteers following single oral doses of 200, 500 and 600 mg, as well as multiple doses of 600mg b.i.d. over five days, using gas chromatography with electron capture detection. The pharmacokinetic parameters derived were independent of dose, and the terminal half-life, t1/2, varied independently of dose between 20 and 38 hours (27.23±1.8 at 600mg). The apparent volume of distribution (Vd area) had similar values at doses of 200, 500 and 600mg of 15.2±2.6, 18.2±1.5 and 14.7±1.7 litres respectively. These small volumes of distribution indicate that fenclofenac distributes mainly into extracellular space. A mean peak plasma concentration of 63.5±4.6 μg/ml developed after 3 to 4 hours following a single 600mg dose whilst a mean steady state plasma concentration (600mg b.i.d.) of 86.9±5.7 μg/ml was achieved within four days, and this decayed with a mean terminal half-life of 25.9±4.2 hours.
Similar content being viewed by others
References
Atkinson, D.C., Godfrey, K.E., Jordan, B.J., Leach, E.C., Meek, B., Nichols, J.D. and Saville, J.F., (1974). 2-(2,4-Dichlorophenoxy) phenylacetic acid (Fenclofenac): one of a novel series of anti-inflammatory compounds with low ulcerogenic potential. J. Pharm. Pharmac.26, 357–359.
Atkinson, D. C. and Cowan, A. (1974). Reversal of yeast induced motor impairment in rats as a test for narcotic and non-narcotic analgesics. J. Pharm. Pharmac.26, 727–729.
Atkinson, D.C. and Leach, E.C. (1976). Anti-inflammatory and related properties of 2-(2,4 dichlorophenoxy) phenylacetic acid (Fenclofenac). Agents in Action,6, 657–666.
Clarke, D.R., Goldberg, A.A.J. and Tudor, R. (1977). The methodology of the fenclofenac clinical research programme and results obtained with particular reference to the therapeutic dose range. Proc. Roy. Soc. Med.,70, 11–15.
Davies, L.L. (1949). Statistical methods in research and production, 2nd ed. London, Oliver and Boyd.
Draper, N.R. and Smith, H. (1966). Applied regression analysis, 1st ed. New York: Wiley.
Gibaldi, M. and Perrier, D. (1975). Drugs and the pharmaceutical sciences V.l. Pharmacokinetics, 1st ed., New York: M. Dekker.
Nguyen Phong Chau, J., (1976). Area-dose relationships in non-linear models. J. Pharmacokin. Biopharm.4, 537–557.
Roncucci, R. and Lambelin, G. (1977). Relevance of pharmacokinetic studies for the evaluation of anti-inflammatory drugs, the example of alclofenac. Drug. Expt. 1. Clin. Res.2, 9–25.
Welling, P.G. (1977). Influence of food and diet on gastrointestinal drug absorption: A Review. J. Pharmacokin. Biopharm.5, 291–335.
Smith, G.L., Gouldbown, R.A., Burt, R.A. and Chatfield, D.H. (1977). Preliminary studies of absorption and excretion of benoxaprofen in man. Br. J. Clin. Pharmac.4, 585–592.
Humphrey, M.J. Personal Communication, (to be published).
Greenway, C.V. and Stark, R.D. (1971). Hepatic Vascular Bed. Physiological Review,51, 23–65.
Wilkinson, G.R. and Shand, D.G. (1975). A physiological approach to hepatic drug clearance. Clin. Pharmac. Ther.18, 377–390.
Swain, M.C. Reckitt and Colman Pharmaceutical Division, Personal Communication.
Gugler, R. and Hartlapp, J. (1978). Clofibrate kinetics after single and multiple doses. Clin. Pharmacol, and Therap.24, 432–438.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Henson, R., Lloyd-Jones, J.G., Nichols, J.D. et al. Pharmacokinetics of fenclofenac following single and multiple doses. European Journal of Drug Metabolism and Pharmacokinetics 5, 217–223 (1980). https://doi.org/10.1007/BF03189467
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03189467