Summary
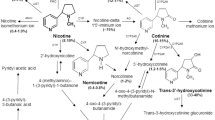
The urinary excretion of nicotine and its metabolites in noninduced and Aroclor-induced male and female rats has been determined following intravenous administration of 2′-[14C]-labeled S-nicotine at a dose of 4.6 μmol/kg. Complete recovery of the administered radioactivity was achieved: 95% in urine and 4% in feces over 96 h and 1% remaining in the body. More than 40 nicotine metabolites were found by radio-HPLC; 19 were identified including thecis/trans-diastereomers of nicotine-N′-oxide and 3′-hydroxycotinine. The urinary metabolite profile and excretion kinetics of nicotine and its metabolites were significantly different between noninduced and Aroclor-induced rats. The major urinary nicotine metabolite in the noninduced rat wascis-nicotine-N′-oxide. In the Aroclor-induced rat, cotinine metabolites were the major metabolites found. Sex differences were found for the urinary nicotine metabolite profile, mainly expressed in the excretion ofcis-nicotine-N′-oxide, 29% in the male and 17% in the female noninduced rat, and the excretion of cotinine, 5% in the male and 12% in the female noninduced rat. High stereoselectivity was found for the formation of thecis/trans-diastereomers of nicotine-N′-oxide as well as of 3′-hydroxycotinine, the stereoselectivity being more pronounced in male rats.
Similar content being viewed by others
References
Gorrod J.W., Jenner P. (1975): The metabolism of tobacco alkaloids. In: Hayes W.J., ed. Essays in Toxicology, Vol. 6. New York: Academic Press, pp. 35–78.
Schievelbein H. (1982): Nicotine, resorption and fate. Pharmacol. Ther., 18, 233–248.
Nakayama H. (1988): Nicotine metabolism in mammals. Drug Metab. Drug Interact., 6, 95–122.
Benowitz N.L., Porchet H., Jacob P. (1990): Pharmacokinetics, metabolism, and pharmacodynamics of nicotine. In: Wonnacott S., Russell M.A.H., Stolerman I.P., eds. Nicotine Psychopharmacology; Molecular, Cellular, and Behavioural Aspects. Oxford: University Press, pp. 112–157.
Kyerematen G.A., Vesell E.S. (1991): Metabolism of nicotine. Drug Metab. Rev., 23, 3–41.
Jenner P., Gorrod J.W., Beckett A.H. (1973): Species variation in the metabolism of R-(+)- and S-(−)-nicotine by α-C- and N-oxidation in vitro. Xenobiotica, 3, 573–580.
Nwosu C.G., Crooks P.A. (1988): Species variation and stereoselectivity in the metabolism of nicotine enantiomers. Xenobiotica, 18, 1361–1372.
Cundy K.C., Crooks P.A., Godin C.S. (1985): Remarkable substrate-inhibitor properties of nicotine enantiomers towards a guinea pig lung aromatic azaheterocycle N-methyltransferase. Biochem. Biophys. Res. Commun., 128, 312–316.
Turner D.M. (1977): The effects of some tobacco smoke constituents on foreign compound metabolism in the cat and the rat. Res. Commun. Chem. Pathol. Pharmacol., 16, 85–100.
McCoy G.D., DeMarco G.J. (1986): Characterization of hamster liver nicotine metabolism. II. Differential effects of ethanol or phenobarbital pretreatment on microsomal N and C oxidation. Biochem. Pharmacol., 35, 4590–4592.
Rüdell U., Foth H., Kahl G.F. (1987): Eightfold induction of nicotine elimination in perfused rat liver by pretreatment with phenobarbital. Biochem. Biophys. Res. Commun., 148, 192–198.
Shinegaga M.K., Trevor A.J., Castagnoli N. (1988): Metabolism-dependent covalent binding of (S)-[5-3H]nicotine to liver and lung microsomal macromolecules. Drug Metab. Dispos., 16, 397–402.
McCoy G.D., DeMarco G.J., Koop D.R. (1989): Microsomal nicotine metabolism: A comparison of relative activities of six purified rabbit cytochrome P-450 isozymes. Biochem. Pharmacol., 38, 1185–1188.
Kyerematen G.A., Morgan M., Warner G., Martin L.F., Vesell E.S. (1990): Metabolism of nicotine by hepatocytes. Biochem. Pharmacol., 40, 1747–1756.
Foth H., Looschen H., Neurath H., Kahl G.F. (1991): Nicotine metabolism in isolated perfused lung and liver of phenobarbital- and benzoflavone-treated rats. Arch. Toxicol., 65, 68–72.
Adir J., Wildfeuer W., Miller R.P. (1980): Effect of ethanol pretreatment on the pharmacokinetics of nicotine in rats. J. Pharmacol. Exp. Ther., 212, 274–279.
Foth H., Walther U.I., Kahl G.F. (1990): Increased hepatic nicotine elimination after phenobarbital induction in the conscious rat. Toxicol. Appl. Pharmacol., 105, 382–392.
Ames B.N., McCann J., Yamasaki E. (1975): Methods for detecting carcinogens and mutagens with the salmonella/mammalian-microsome mutagenicity test. Mutat. Res., 31, 347–363.
Demetriou D., Rustemeier K., Voncken P., Schepers G. (1993): HPLC separation of the enantiomers of nicotine and nicotine-like compounds. Chirality, In press.
Lehnhardt F.-J. (1989): A new system for catheterism of the V. cava in rats for longtime infusions (Implantofix-R). Z. Versuchstierkd., 32, 171–178.
Cundy K.C., Crooks P.A. (1984): High-performance liquid chromatographic method for the determination of N-methylated metabolites of nicotine. J. Chromatogr., 306, 291–301.
Demetriou D., Rustemeier K., Voncken P., Schepers G. (1992): Two radiochromatographic HPLC methods for the determination of nicotine metabolites. Med. Sci. Res., 20, 873–875.
Kyerematen G.A., Damiano M.D., Dvorchik B.H., Vesell E.S. (1982): Smoking-induced changes in nicotine disposition: application of a new HPLC assay for nicotine and its metabolites. Clin. Pharmacol., 32, 769–780.
Jenner P., Gorrod J.W., Beckett A.H. (1973): The absorption of nicotine-1′-N-oxide and its reduction in the gastro-intestinal tract in man. Xenobiotica, 3, 341–349.
Dajani R.M., Gorrod J.W., Beckett A.H. (1975): Reduction in vivo of (−)-nicotine-1′-N-oxide by germ-free and conventional rats. Biochem. Pharmacol., 24, 648–650.
Duan M.J., Yu L., Savanapridi C., Jacob III P., Benowitz N.L. (1991): Disposition kinetics and metabolism of nicotine-1′-N-oxide in rabbits. Drug Metab. Dispos., 19, 667–672.
Sepkovic D.W., Haley N.J. (1987): Metabolism of nicotine in smokers and non-smokers. In: Martin W.R., Van Loon G.R., Iwamoto E.T., Davis L., eds. Tobacco Smoking and Nicotine. New York: Plenum Press, pp. 375–388.
Kyerematen G.A., Owens G.F., Chattopadhyay B., deBethizy J.D., Vesell E.S. (1988): Sexual dimorphism of nicotine metabolism and distribution in the rat. Drug Metab. Dispos., 16, 823–828.
Castro A., Monji N., Ali H., Yi J.M., Bowman E.R., McKennis H. (1980): Nicotine antibodies: Comparison of ligand specificities of antibodies produced against two nicotine conjugates. Eur. J. Biochem., 104, 331–340.
Curvall M., Kazemi Vala E., Englund G., Enzell C.R. (1989): Urinary excretion of nicotine and its major metabolites. Paper No. 54 presented at the 43rd Tobacco Chemists’ Research Conference, Richmond VA., USA.
Curvall M., Kazemi Vala E., Englund G. (1991): Conjugation pathways in nicotine metabolism. In: Adikofer F., Thurau K., eds. Effects of Nicotine on Biological Systems. Basel: Birkhäuser Verlag, pp. 69–75.
Kyerematen G.A., Morgan M.L., Chattopadhyay B., deBethizy J.D., Vesell E.S. (1990): Disposition of nicotine and eight metabolites in smokers and nonsmokers: Identification in smokers of two metabolites that are longer lived than continine. Clin. Pharmacol. Ther., 48, 641–651.
Byrd G.D., Chang K.-M., Greene J.M., deBethizy J.D. (1992): Evidence for urinary excretion of glucuronide conjugates of nicotine, cotinine, andtrans-3′-hydroxycotinine in smokers. Drug Metab. Dispos., 20, 192–197.
Rustemeier K., Demetriou D., Schepers G., Voncken P. (1993): High-performance liquid chromatographic determination of nicotine and its urinary metabolites via their 1,3-diethyl-2-thiobarbituric acid derivatives. J. Chromatogr., 613, 95–103.
Schepers G., Demetriou D., Rustemeier K., Voncken P., Diehl B. (1992): Nicotine phase 2 metabolites in human urine — structure of metabolically formedtrans-3′-hydroxycotinine glucuronide. Med. Sci. Res., 20, 863–865.
Kyerematen G.A., Taylor L.H., deBethizy J.D., Vesell E.S. (1988): Pharmacokinetics of nicotine and 12 metabolites in the rat. Drug Metab. Dispos., 16, 125–129.
Pilotti A., Enzell C.R., McKennis H., Bowman E.R., Dufva E., Holmstedt B. (1976): Studies on the identification of tobacco alkaloids, their mammalian metabolites and related compounds by gas chromatography — mass spectrometry. Beitr. Tabakforsch., 8, 339–349.
Murphy P. (1973): Enzymatic oxidation of nicotine to nicotine Δ1′(5′) iminium ion. J. Biol. Chem., 248, 2796–2800.
Nguyen T.-L., Gruenke L.D., Castagnoli N. (1979): Metabolic oxidation of nicotine to chemically reactive intermediates. J. Med. Chem., 22, 259–263.
Lemoine A., Williams D.E., Cresteil T., Leroux J.-P. (1991): Hormonal regulation of microsomal flavin-containing monooxygenase: Tissue-dependent expression and substrate specificity. Mol. Pharmacol., 40, 211–217.
Ziegler D.M. (1980): Microsomal flavin-containing monooxygenase: Oxygenation of nucleophilic nitrogen and sulfur compounds. In: Jacoby W.B., ed. Enzymatic Basis of Detoxication, Vol. 1. New York: Academic Press, pp. 201–227.
Damani L.A., Pool W.F., Crooks P.A., Kaderlik R.K., Ziegler D.M. (1988): Stereoselectivity in the N′-oxidation of nicotine isomers by flavin-containing monooxygenase. Mol. Pharmacol., 33, 702–705.
Booth J., Boyland E. (1970): The metabolism of nicotine into two optically-active stereoisomers of nicotine-1′-oxide by animal tissues in vitro and by cigarette smokers. Biochem. Pharmacol., 19, 733–742.
Jenner P., Gorrod J.W., Beckett A.H. (1971): Comparative C- and N-oxidation of (+)- and (−)-nicotine by various species. Xenobiotica, 1, 497–498.
Voncken P., Rustemeier K., Schepers G. (1990): Identification ofcis-3′-hydroxycotinine as a urinary nicotine metabolite. Xenobiotica, 20, 1353–1356.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schepers, G., Rustemeier, K., Walk, R.A. et al. Metabolism of S-nicotine in noninduced and aroclor-induced rats. Eur. J. Drug Metab. Pharmacokinet. 18, 187–197 (1993). https://doi.org/10.1007/BF03188795
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03188795